7 %(ME) = 16.6; %(EE) = 16.6; %(PE) = 33.4; %(BE) = 33.4.
8 By adding N‐methylserotonin before extraction, we do not have to take into consideration any potential loss of product due to the various manipulations. We suppose that the extraction yield is the same for these two compounds, which are very similar;kS/NMS = 1.002;serotonin mass: 45 ng/ml.
1 1The symbols used follow IUPAC recommendations – Pure and Applied Chemistry, 65(4), 819 (1993).
Chapter 2 Gas Chromatography
Gas chromatography (GC) separates compounds that may be vaporized without decomposition when heated. To do so, analytes, in contact with the gaseous mobile phase, are brought to high temperatures. The same goes for the stationary phase caught in the column. GC can be paired with many types of detection, especially mass spectrometry, which often helps in positive identification of analytes. This versatile and very sensitive technique is known for its quick optimization of analytical conditions, and thanks to current advances, such as high‐speed or multidimensional gas chromatography, it is a very attractive and essential resource when studying volatile compounds.
Representa GC device
Choosethe carrier gas
Choosethe column
Comparethe stationary phases
Knowthe injection methods
Listthe main detectors
Optimizea separation
Addressnew orientations of micro GC and fast GC
Explainretention indexes and constants of stationary phases
2.1 COMPONENTS OF A GC INSTALLATION
A gas chromatograph is composed of three components within a single surround. These components include the injector, the column, and the detector associated with a temperature‐controlled oven that enables the column to attain high temperatures ( Figure 2.1). The mobile phase that transports the analytes through the column is a gas referred to as the carrier gas . The carrier gas flow, which is precisely controlled, enables great reproducibility of the retention times.
Analysis starts when a very small quantity of sample is introduced in either liquid or gas form into the injector, which has the dual function of vaporizing the sample and mixing it with the gas flow at the head of the column. The column is usually a narrow‐bore tube that coils around itself with a length that can vary from 1 m to over 100 m, depending upon the type and the contents of the stationary phase. The column, which can serve for thousands of successive injections, is housed in a temperature‐controlled oven. At the end of the column, the mobile phase (carrier gas) passes through a detector before it exits to the atmosphere. Some gas chromatograph models of reduced size have their own electrical supply, enabling them to operate in the field ( Figure 2.1).
In GC, there are four operational parameters for a given stationary phase: L , length of the column; u , velocity of the mobile phase (which affects the theoretical efficiency N ); T , temperature of the column; and β , phase ratio, which affects the retention factor k . The settings of the chromatograph enable modifications in terms of T and u , and therefore both the efficiency of the column and the retention factors can be adjusted as well.

Figure 2.1 Operational diagram of a gas chromatograph and practical uses. a, b) Versatile analytical chromatogram
(Source: GC‐2010 model from Shimadzu).
The instrument represented is equipped with a sample holder (carousel), an injector, an automatic sampler, and a mass spectrometry detection system (GC‐MS). c) Portable model for analyses made in the field (volatile organic compounds, toxic industrial materials)
(Source: Hapsite ER, information courtesy of Inficon).
2.2 CARRIER GAS AND FLOW REGULATION
The mobile phase is a gas (helium, hydrogen or nitrogen), either drawn from a commercially available gas cylinder or obtained, in the case of hydrogen or nitrogen, from an on‐site generator (water electrolysis for H 2and air separation for N 2), dedicated to the installation. To eliminate all harmful traces of water vapour and oxygen from polar stationary phases and detectors, a double filter, for drying and reducing, is placed before the injector.
The nature of the carrier gas has no significant influence upon the values of the partition coefficients K of the solutes between the stationary and mobile phases, owing to an almost total absence of interaction between the gas and the solutes. Temperature is the only significant modification factor. By contrast, the viscosity and speed of the carrier gas, related to its flow rate or pressure at the head of the column, have an effect on the analytes’ dispersion in the stationary phase and on their diffusion in the mobile phase (see the Van Deemter equation, Chapter 1, Section 1.10). These two factors have a direct impact on column efficiency N ( Figure 2.2).
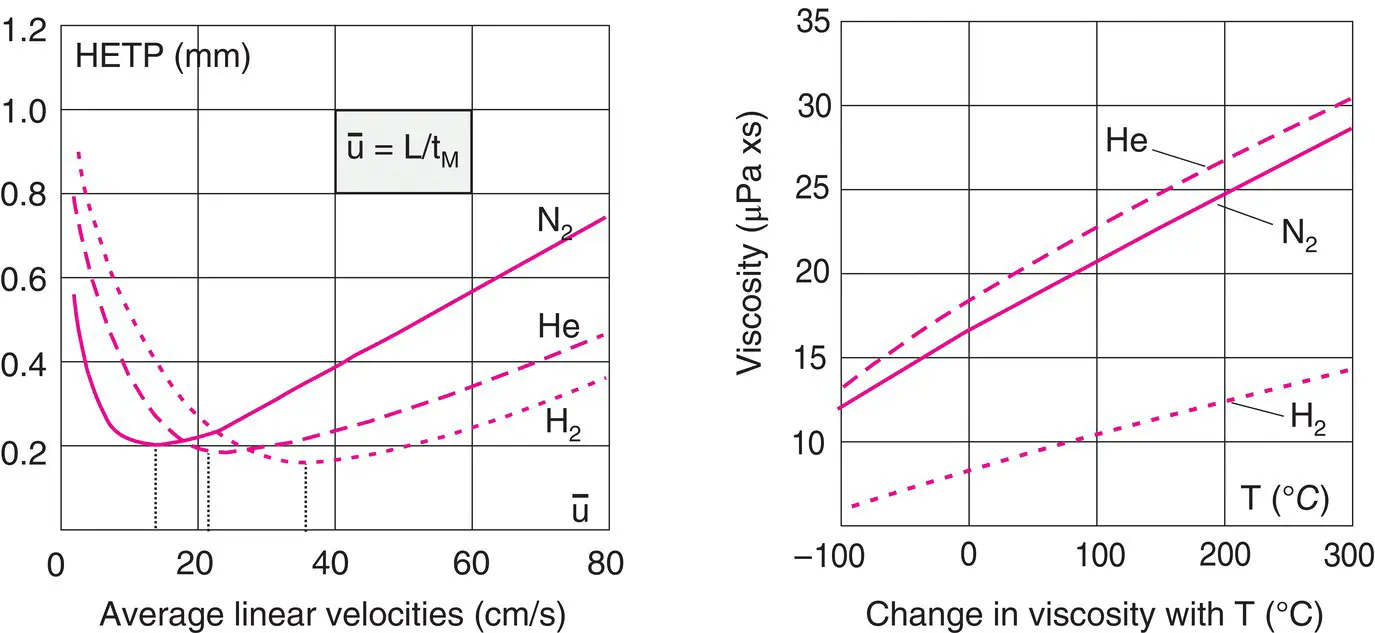
Figure 2.2 Efficiency as a function of the nature and linear velocity of the carrier gas. The Van Deemter curves show the relationship between HETP and linear velocity of the carrier gas for a given compound. Comparison of the viscosities of these three gases. Note the increase in the viscosity of these gases with temperature.
The pressure at the head of the column (several tens to hundreds of kPa – a few tenths to a few bar) is stabilized by electronic pressure control ( EPC ), so that the flow rate remains constant at its optimal value. This device is valuable because if the analysis is performed with ascending temperature programming (temperature gradient), the viscosity of the stationary phase and, by consequence, the pressure drop in the column, increase with time. By controlling the pressure, we conserve a constant and optimal speed of the carrier gas. The result is a faster analysis with the same efficiency.
The comparison of the three Van Deemter curves shows that the minimum of each is obtained for various linear velocities of the carrier gas: low for nitrogen, higher for helium and even higher for hydrogen. This means that the latter gas reduces the analysis time. For hydrogen also, the growth of the curve after the peak is less quick than for the other two gases, which gives more latitude in choosing the carrier gas speed without impacting the column’s efficiency ( Figure 2.2). The three curves plotting the viscosity of these gases versus temperature T again show that hydrogen has a lower viscosity than the other two carrier gases. Lower viscosity means a lower pressure drop and an increased column life.
2.3 INJECTION CHAMBER
2.3.1 Sample Introduction
The sample to be analysed is never introduced into the chromatogram as is, whether it is a liquid or a solid, but rather in a highly diluted solution. We use either a microsyringe (or loop injector) or a device such as a headspace sampler for volatile compounds, which both concentrates the sample and introduces it into the chromatograph.
The most common injection method is where a microsyringe is used ( Figure 2.3) to inject a very small quantity of sample in solution (1 μl or less) through a rubber septum sealing the injection chamber. Some very elaborate septa have been developed ( Figure 2.4) for microsyringes, which are an integral part of the automatic injectors found in most current instruments ( Figure 2.1).
Читать дальше





![Евгений Матерёв - Музеи… или вдохновляющая музыка The Chemical Brothers [litres самиздат]](/books/437288/evgenij-materev-muzei-ili-vdohnovlyayuchaya-muzyka-th-thumb.webp)







