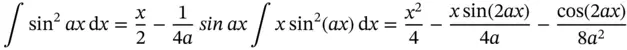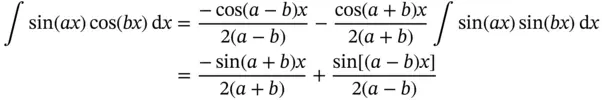1 ΔE = hc/λ or λ = hc/ΔE(E2.4.3)
Certain quantum dot structures can also be modeled by a 2D particle in a box. Quantum dots may be manufactured by creating small circular or square semiconductor deposits on a substrate that is an electric insulator. The electrons of the semiconductor spots are free to move over the entire size of the dot, and the energy levels of the free electrons follow a 2D PiB model [3]. Consequently, the color of electronic transitions can be tuned by varying the size of the quantum dot.
Closely related to these 2‐dimensional quantum dots are 3‐dimensional nanoparticles, such as spheres of metallic or semiconductor materials. The free electrons on the surface of such spheres can assume wave patterns known as “spherical harmonic functions” that are the solutions for particle on a sphere. Similar functions will be discussed in Chapter 7during the treatment of the hydrogen atom wavefunctions. Again, the optical properties of such nanoparticles can be tuned by adjusting the size of the nanoparticle. This is shown in Figure 2.7.
2.5.3 Quantum Cascade Lasers
Finally, an example of a commercial application of the particle in a box will be discussed, namely, that of a solid‐state infrared laser known as the quantum cascade laser (QCL) [4]. In QCLs, the “box” is constructed by creating a semiconductor “superlattice” potential functions that mimics a PiB with finite potential energy barriers. Furthermore, the bottom of the energy well is not flat, but at a slant, as shown in Figure 2.8a. Both the barriers and the slant of the bottom of the energy well can be achieved in the fabrication process by vaporizing different composition of semiconductor materials (doping).
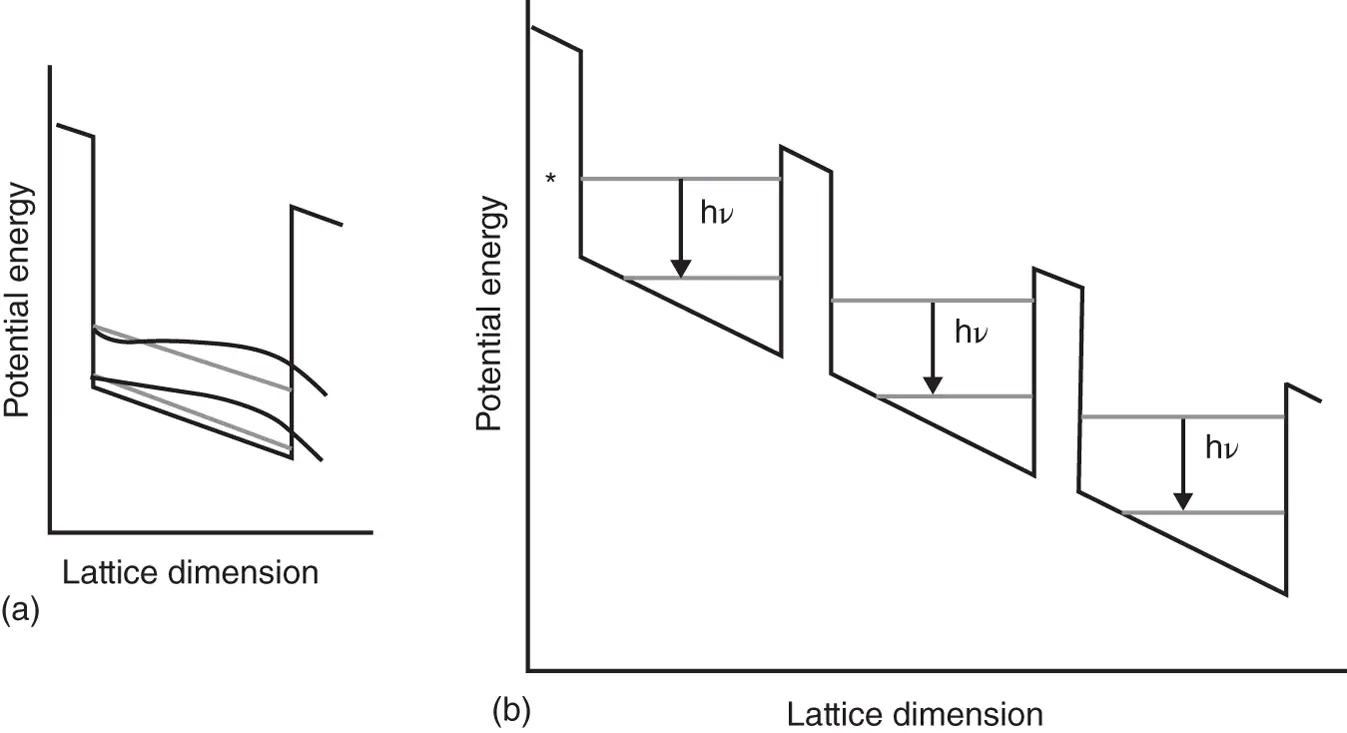
Figure 2.8 (a) An individual energy well with finite barrier height and a sloping energy bottom with the two lowest energy states and wavefunctions. (b) The superlattice structure in a quantum cascade laser modeled by successive PiB potential energy levels.
The slant of the energy well bottom has the effect of distorting the PiB wavefunctions as shown exaggerated for the two lowest energy states in Figure 2.8a. (The computation of wavefunctions in the presence of a sloping baseline will be discussed in Appendix 2, perturbation methods.) The distortion causes the amplitude of the wavefunction to shift toward lower potential energy with the consequence that an electron in the ground state of the well has a higher probability of tunneling through the barrier, due to their higher amplitude at the right side of the well. The “superlattice” is formed by having a large number of these energy wells arranged as shown in Figure 2.8b.
During the operation of the QCL, electrons are injected, via an electric current, at a potential energy marked by the * symbol in Figure 2.8binto a highly excited energy state and undergo a transition as indicated by the leftmost down arrow. During this transition, an (infrared) photon is emitted. Subsequently, the electron in the ground state may tunnel through the finite‐height barrier and arrives in the next quantum well and undergoes another transition. The emission and tunneling processes are repeated as many times as there are quantum wells in the superstructure. The term “cascade” in QCL is due to the fact that one electron can undergo many consecutive emission processes in the superlattice structure. By placing the superlattice crystal into an optic cavity, stimulated emission (see Chapter 3) from the excited states into the ground states of each well can be achieved.
1 1 Levine, I. (1983). Quantum Chemistry. Boston: Allyn & Bacon.
2 2 Shoemaker, D.P., Garland, C.W., and Nibler, J.W. (1996). Experiments in Physical Chemistry, 6e. New York: McGraw‐Hill.
3 3 Banin, U. et al. (1999). Identification of atomic‐like electronic states in indium arsenide nanocrystal quantum dots. Nature 400 (6744): 542–544.
4 4 Faist, J. et al. (1994). Quantum Cascade Laser. Science 264 (5158): 553–556.
The following trigonometric integral relationships are needed for these problems:
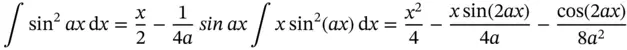
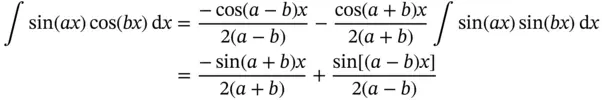
1 Show that the function f(x) = cos(bx) is an eigenfunction of the operator d2/dx2. What is the eigenvalue?
2 Show that the function e−x2/2 is an eigenfunction of the operator (d2/dx2) − x2. What is the eigenvalue?
3 Show that the function e−4ix is an eigenfunction of the operator d2/dx2. What is the eigenvalue?
4 What is the probability P of finding a ground‐state PiB in the center third of the box? What is P for the same range for a classical particle?
5 For the PiB in the ground state, determine the expectation values of x and px.
6 What is the expectation value of the kinetic energy operator T for the ground‐state PiB?
7 What is the probability P of finding a particle in the first excited state in the left half of the box with length L within the PiB approximation?
8 Consider an electron in a one‐dimensional box with a length of 0.1 nm.Calculate the energy of the 1st, 2nd, and 3rd energy levels for this electron.Calculate the wavelength of a photon required to promote the electron from the 2nd to the 3rd energy level.
9 Describe in your own words why the particle‐in‐a‐box model results in quantized energy levels.
10 What is quantum mechanical tunneling?
11 Calculate the commutator [Tx, px] where Tx is the kinetic energy operator in the x‐direction and px is the momentum operator in the x‐direction. Can the kinetic energy and the momentum be determined simultaneously in a quantum mechanical system?
12 Show by analytical integration that ψ1(x) and ψ2(x) are orthogonal for the one‐dimensional PiB.
13 Using a graphics program such as Excel, plot the first and second wavefunctions for a particle in a box. Show by graphical integration that these functions are orthogonal.
Конец ознакомительного фрагмента.
Текст предоставлен ООО «ЛитРес».
Прочитайте эту книгу целиком, купив полную легальную версию на ЛитРес.
Безопасно оплатить книгу можно банковской картой Visa, MasterCard, Maestro, со счета мобильного телефона, с платежного терминала, в салоне МТС или Связной, через PayPal, WebMoney, Яндекс.Деньги, QIWI Кошелек, бонусными картами или другим удобным Вам способом.