(1.11) 
the light entering the sample has the intensity
(1.12) 
This light is attenuated, according to the Lambert–Beer law (1.4). Furthermore, before exiting the sample, the light is reflected again on the exit surface. It is found that the transmitted light for single reflection path is given by
(1.13) 
and the absorbance estimation is
(1.14) 
This result shows that, due to the reflection effect, an absorption different from zero is experimentally observed even in the absence of absorbing centers, that is when N = 0. Taking the more accurate multiple reflections effect between the two surfaces with refractive index mismatch between the sample and the medium, it is found that [4]:
(1.15) 
where p is the reflection factor. When this factor, or the refractive index dependence on the wavelength, is not known, the “parasitic” effect of reflection cannot be estimated. A technical solution is to take the measurement of the same material using two different thicknesses, if possible. In fact, considering two samples of thickness L 1and L 2, respectively, we obtain:
(1.16) 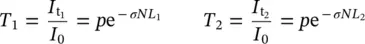
(1.17) 
This way, it is shown that the two measurements enable to find the experimentally relevant features related to the absorbing centers: the cross section and the concentration, or the absorption coefficient.
To conclude this paragraph, in Figure 1.2, a typical absorption spectrum is reported with the absorbance in the vertical axis and the wavelength (in nanometer) in the horizontal axis.
It can be observed that the amount of absorbance (or optical density) changes by changing the wavelength, with a profile depending on the specific features of the investigated material. To carry out a meaningful interpretation of the spectrum, taking in due account the spectral profile and the electronic state distribution, the wavelength axis has to be changed into an axis of energy, E (usually reported in electronvolt, eV; 1 eV = 1.602 ⋅ 10 −19J). To achieve this aim, it is useful to refer to the Planck–Einstein relation [9]: E = hν , where h is the Planck’s constant (6.626 ⋅ 10 −34J⋅s = 4.136 ⋅ 10 −15eV⋅s). To convert the axis from wavelength to energy, one can use the formula:
(1.18) 
and the conversion equation
(1.19) 
that enables to correlate a value of energy with the wavelength and vice versa. Another useful quantity in spectroscopy is the wavenumber, 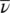 . This is defined by (wavelength) −1, 1/ λ , and, using Eq. (1.18), it is shown that
. This is defined by (wavelength) −1, 1/ λ , and, using Eq. (1.18), it is shown that
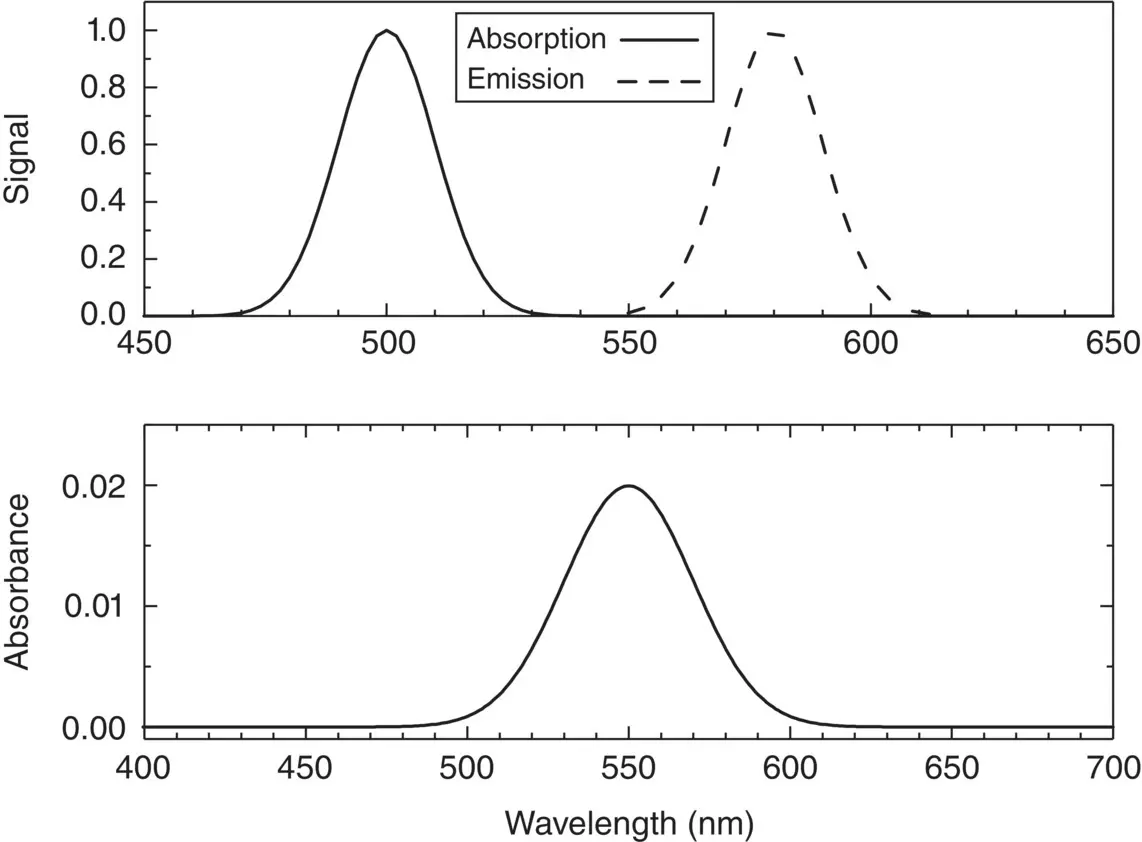
Figure 1.2Bottom: Typical absorption spectrum registered as a function of wavelength. Top: Representative experimental absorption (continuous line) and emission (dashed line) spectra registered as a function of wavelength.
(1.20) 
The wavenumber is usually reported in units of cm −1. Combining Eqs. (1.19)and (1.20), it is found that
(1.21) 
Concluding this paragraph, it is worth mentioning that the absorption phenomenon is one of the basic processes of the radiation–matter interaction and it is extended in a wide range of energy of the electromagnetic spectrum. The underling physical process is related to the specific atomic or molecular species absorbing the energy from the electromagnetic wave [8, 9]. The frequency range of interest for this chapter includes the visible (Vis) radiation and goes from the near infrared (NIR) to the ultraviolet (UV). In particular, the visible range in vacuum extends in frequency from about 3.8⋅10 14to 7.5⋅10 14Hz, in wavelength from 800 to 400 nm, and in energy from 1.6 to 3.1 eV [1].
1.1.2 Emission: Fluorescence and Phosphorescence
The absorption of light at a given wavelength λ by a sample is physically associated to an energy transfer from the radiation electromagnetic field to the electrons of the matter constituting the sample. In this phenomenon, the electrons are typically promoted from one energy level (ground state) to another level of higher energy (excited state). In the process, the electron system is put out of thermal equilibrium; so, each electron spontaneously tends to return to its initial energy level, releasing the acquired energy. In an ideal experiment, a stationary state can be observed in which by continuously illuminating the sample with a radiation at λ , in a given spatial direction, another radiation is emitted by the sample isotropically in space with wavelength λ′ > λ . This phenomenon is known as photoluminescence and can sometimes be observed also by naked eye, illuminating a sample with high‐energy photons, typically in the UV part of the spectrum, and revealing an emission in the visible range (an example of this effect is observed in the luminescence emergency panels, emitting light when they are in the dark after being illuminated by electric lamps or sunlight). It is worth underlining that typically, as shown in Figure 1.2, the absorbed radiation wavelength (energy) and the emitted wavelength (energy) are related by
(1.22) 
due to some internal processes subtracting energy to the electrons when they return to their ground state. The difference between the photon energy at which the absorption maximum amplitude occurs and the energy where the emission maximum amplitude occurs is called Stokes shift [2, 5].
Читать дальше






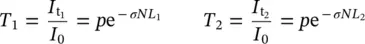



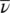 . This is defined by (wavelength) −1, 1/ λ , and, using Eq. (1.18), it is shown that
. This is defined by (wavelength) −1, 1/ λ , and, using Eq. (1.18), it is shown that














