25 Chapter 19: Potentiometric and Ionometric Methods 19.1 MEASUREMENT CELLS 19.2 THE PH ELECTRODE 19.3 ION‐SELECTIVE ELECTRODES (ISE) 19.4 QUANTIFICATIONS METHODS 19.5 APPLICATIONS KEY POINTS OF THE CHAPTER PROBLEMS SOLUTIONS
26 Chapter 20: Voltammetric Methods 20.1 THE VOLTAMMETRIC METHOD 20.2 THE DROPPING MERCURY ELECTRODE 20.3 DIRECT CURRENT POLAROGRAPHY (DCP) 20.4 DIFFUSION CURRENT 20.5 PULSE POLAROGRAPHY 20.6 ALTERNATING CURRENT POLAROGRAPHY (ACP) 20.7 STRIPPING VOLTAMMETRY 20.8 COULOMETRIC MEASUREMENTS 20.9 COULOMETRIC TITRATION OF WATER CONTENT 20.10 VOLTAMMETRIC DETECTION IN HPLC AND HPCE 20.11 AMPEROMETRIC SENSORS KEY POINTS OF THE CHAPTER PROBLEMS SOLUTIONS
27 Chapter 21: Sample Preparation 21.1 THE NEED FOR SAMPLE PRETREATMENT 21.2 SOLID PHASE EXTRACTION (SPE) 21.3 IMMUNO‐AFFINITY EXTRACTION 21.4 MICRO‐EXTRACTION PROCESSES 21.5 GAS EXTRACTION ON A CARTRIDGE OR A DISC 21.6 HEADSPACE 21.7 SUPERCRITICAL PHASE EXTRACTION (SPE) 21.8 MICROWAVE REACTORS 21.9 ON‐LINE ANALYSERS KEY POINTS OF THE CHAPTER
28 Chapter 22: Basic Statistical Parameters 22.1 CENTRAL VALUE, ACCURACY, AND RELIABILITY OF A SET OF MEASUREMENTS 22.2 VARIANCE AND STANDARD DEVIATION 22.3 RANDOM OR INDETERMINATE ERRORS 22.4 CONFIDENCE INTERVAL OF THE MEAN 22.5 COMPARISON OF RESULTS – PARAMETRIC TESTS 22.6 REJECTION CRITERIA USING THE Q QUOTIENT (OR DIXON TEST) 22.7 CALIBRATION CURVES 22.8 ROBUST METHODS OR NONPARAMETRIC TESTS 22.9 OPTIMIZATION WITH THE ONE‐FACTOR‐AT‐A‐TIME (OFAT) METHOD PROBLEMS SOLUTIONS
29 Appendix Table of Some Useful Constants
30 Bibliography
31 Index
32 End User License Agreement
1 Chapter 2 Table 2.1 Comparison of conventional, fast and ultra‐fast GC set‐ups. Table 2.2 McReynolds constants (∆ I ) for several stationary phases normalize...
2 Chapter 4Table 4.1 Equivalent conductivities of ions at infinite dilution in water a...
3 Chapter 7Table 7.1 Permeation range (Da) of three gels for various standard compound...
4 Chapter 9Table 9.1 Characteristic chromophores of some nitrogen‐containing groups.
5 Chapter 10Table 10.1 Correlation chart in the mid‐infrared region between functional ...Table 10.2 Comparison of absorption (IR) and scattering (Raman) intensities ...
6 Chapter 11Table 11.1 Position of the Raman peaks, calculated for four commonly used s...
7 Chapter 12Table 12.1 Several radioisotope X‐ray sources.
8 Chapter 13Table 13.1 Ratio of N e /N 0for several elements at various temperatures.Table 13.2 Upper temperature limits for some gas mixtures.
9 Chapter 15Table 15.1 Values of γ for some nuclei.Table 15.2 Values in MHz of some resonance frequencies for 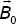 = 1T.Table 15.3 Influence of electronegativity χ of the halogen on δ (T...Table 15.4 Pascal’s triangle and its application to NMR for I = ½.Table 15.5 NMR chemical shifts of the main types of protons in organic molecules.Table 15.6 13C NMR correlation table of organic functions.
= 1T.Table 15.3 Influence of electronegativity χ of the halogen on δ (T...Table 15.4 Pascal’s triangle and its application to NMR for I = ½.Table 15.5 NMR chemical shifts of the main types of protons in organic molecules.Table 15.6 13C NMR correlation table of organic functions.
10 Chapter 16Table 16.1 Atomic mass of the isotopes of the main elements commonly encount...
11 Chapter 17Table 17.1 Some characteristics of commonly used radioisotopes.
12 Chapter 19Table 19.1 Values of the ideal slopes at 298 K for a few ions.
13 Chapter 22Table 22.1 Examples of results from an analysis grouping the various parame...Table 22.2 Values of the Student’s two‐sided confidence coefficient t (calcu...Table 22.3 Summary of the F threshold values established for a confidence le...Table 22.4 Tabular summary of critical values for Q (Dixon’s test)Table P22.1
1 Chapter 1 Figure 1.1 A basic experiment in chromatography. (a) The necessary ingredien... Figure 1.2 The principle of analysis by chromatography. The chromatogram, th... Figure 1.3 Chromatographic elution curve. Example of a graph of Eq. (1.1). Figure 1.4 Characteristics of an ideal chromatographic peak. Meaning of the ... Figure 1.5 Distribution isotherms. (a) The ideal situation corresponding to ... Figure 1.6 Dispersion of a solute in a column. Left, a graph corresponding t... Figure 1.7 Retention factors and separation factor (or selectivity factor) b... Figure 1.8 Resolution factor. A simulation of chromatographic peaks by juxta... Figure 1.9 Effect of column length on the resolution. Chromatograms obtained... Figure 1.10 Van Deemter curve in gas chromatography with the domains of para... Figure 1.11 The chromatographer’s compromise triangle between resolution, sp... Figure 1.12 Analysis by the external standard method. The precision of this ... Figure 1.13 Method of analysis by internal standard. Figure 1.14 Analysis by internal normalization method.
2 Chapter 2 Figure 2.1 Operational diagram of a gas chromatograph and practical uses. a,... Figure 2.2 Efficiency as a function of the nature and linear velocity of the... Figure 2.3 Microsyringe for GC and principle of an injection loop installed ... Figure 2.4 Direct vaporization injector used for packed columns. The typical... Figure 2.5 Injectors. Above left, injection chamber with splitter (vent 2 re... Figure 2.6 PTV injector, with programmable temperature and cold on‐column in... Figure 2.7 Capillary columns. Representation of a 50 m long commercial capil... Figure 2.8 Polarity scale of stationary phases in GC and summary of the thre... Figure 2.9 Example of a separation with a chiral phase which contains grafte... Figure 2.10 Chiral stationary phase in GC. Among the three chiral vectors en... Figure 2.11 Gas analyses. Left, one of the earliest ever chromatograms, obta... Figure 2.12 FID detector (a) and NPD detector (b). The make‐up gas (or filli... Figure 2.13 Thermal conductivity detector. Left, layout demonstrating the du... Figure 2.14 (a) Electron capture detector (ECD) and (b) photo‐ionization det... Figure 2.15 Three detectors connected in series. At the outlet of a capillar... Figure 2.16 ‘Ultra‐fast’ chromatogram. Left, separation of several aromatic ... Figure 2.17 Kovats straight line graph. Left, isothermal chromatogram for a ... Figure 2.18 Kovats retention index (I = 100n x) on a column in isothermal mod...
3 Chapter 3 Figure 3.1 Diagram of a modular HPLC instrument. The vertical assembly of th... Figure 3.2 Simplified diagram of a dual‐headed reciprocating pump. From the ... Figure 3.3 Example of a high‐pressure gradient system. The pumps are called ... Figure 3.4 Injection valve for HPLC and an assortment of loops. Left, rear v... Figure 3.5 Injection with a loop. (a) Loading of the loop. In this step, the... Figure 3.6 Standard column and guard column for HPLC. View of the ZORBAX ®... Figure 3.7 Silica gels for HPLC. Solid‐core phases lead to notable improveme... Figure 3.8 Nature of the stationary phase and efficiency. HETP decreases wit... Figure 3.9 The phenomena of adsorption and partition. Adsorption is an inter... Figure 3.10 Evolution of liquid chromatography performance. The four Van Dee... Figure 3.11 Formation of organosilanes grafted at the interface of silica ge... Figure 3.12 Examples of chiral stationary phases used in HPLC. Separation of... Figure 3.13 Strength of solvents used as mobile phases. By mixing several so... Figure 3.14 Relative polarities of several organic compound families along w... Figure 3.15 The separation of sugars on an amine phase. The order of elution... Figure 3.16 Ion pairing effects on a separation with an RP‐18 type column. A... Figure 3.17 Separation on the principle of hydrophobic interaction. The chro... Figure 3.18 Separation using the hydrophilic interaction principle (HILIC). ... Figure 3.19 Photometric detection. Principle of a photometric detector along... Figure 3.20 Chromatograms of a sample containing two compounds A and B whose... Figure 3.21 The diode array detector. Unlike the monochromatic detector, the... Figure 3.22 Three‐dimensional representation, i = f(t, λ), of a chromatograp... Figure 3.23 Fluorometric detector. 1) Diagram of a fluorometric detector. 2)... Figure 3.24 Diagram of a differential refractive index detector. Optical pat... Figure 3.25 Evaporative light scattering detector. In three successive stage...Figure 3.26 Polarimeter detection. Diagram representing the main components ...Figure 3.27 Chromatograms of a separation. The mobile phase is a binary wate...Figure 3.28 Effect of column temperature on a separation. Example showing th...Figure 3.29 Fast chromatography. This example shows that the choice of colum...Figure 3.30 Comparison of flow rates in columns with different internal diam...Figure 3.31 Columns for U‐HPLC. Nanochromatography. Capillary columns, 75 μm...
Читать дальше

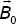 = 1T.Table 15.3 Influence of electronegativity χ of the halogen on δ (T...Table 15.4 Pascal’s triangle and its application to NMR for I = ½.Table 15.5 NMR chemical shifts of the main types of protons in organic molecules.Table 15.6 13C NMR correlation table of organic functions.
= 1T.Table 15.3 Influence of electronegativity χ of the halogen on δ (T...Table 15.4 Pascal’s triangle and its application to NMR for I = ½.Table 15.5 NMR chemical shifts of the main types of protons in organic molecules.Table 15.6 13C NMR correlation table of organic functions.

![Евгений Матерёв - Музеи… или вдохновляющая музыка The Chemical Brothers [litres самиздат]](/books/437288/evgenij-materev-muzei-ili-vdohnovlyayuchaya-muzyka-th-thumb.webp)







