John T. Moore - Biochemistry For Dummies
Здесь есть возможность читать онлайн «John T. Moore - Biochemistry For Dummies» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Biochemistry For Dummies
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
Biochemistry For Dummies: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Biochemistry For Dummies»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
In
you’ll explore the carbons, proteins, and cellular systems that make up the biochemical processes that create and sustain life of all kinds. Perfect for students majoring in biology, chemistry, pre-med, health-services, and other science-related fields, this book tracks a typical college-level biochemistry class. It simplifies and clarifies the subject with easy-to-follow diagrams and real-world examples. You’ll also get:
Explorations of cell biology, carbohydrates, proteins, lipids, and other fundamental building blocks of life Discussions of the basic structures common to all living organisms Treatments of the microscopic details of life that make us all tick If you’re looking for a hand with some of the trickier parts of biochemistry—or you just need an accessible overview of the subject—check out
today!

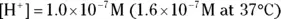 . The hydroxide ion concentration is the same as the hydrogen ion concentration because they’re formed in equal amounts during the autoionization reaction. Keep in mind that
. The hydroxide ion concentration is the same as the hydrogen ion concentration because they’re formed in equal amounts during the autoionization reaction. Keep in mind that 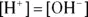 only in pure water.
only in pure water. M is a concentration term, the molarity. Molarity is the number of moles of solute per liter of solution.
M is a concentration term, the molarity. Molarity is the number of moles of solute per liter of solution.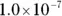 , isn’t always convenient. Thankfully, you have a way of simplifying the representation of the hydrogen ion concentration: the pH. You can calculate the pH for any solution by using the following equation:
, isn’t always convenient. Thankfully, you have a way of simplifying the representation of the hydrogen ion concentration: the pH. You can calculate the pH for any solution by using the following equation: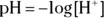
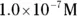 , the pH calculation would be
, the pH calculation would be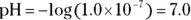
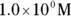
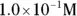
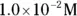
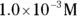
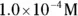
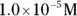
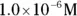
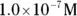
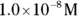
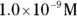
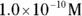
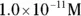
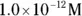
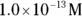
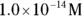
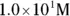 solution of hydrochloric acid is
solution of hydrochloric acid is 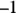 . John loves to ask questions based on this topic to his advanced chemistry students! The
. John loves to ask questions based on this topic to his advanced chemistry students! The 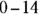 scale is a convenient part of the pH scale for most real-world solutions — especially ones found in biochemistry. Most biological systems have a pH near 7, although significant deviations may exist (for example, the pH in your stomach is close to 1).
scale is a convenient part of the pH scale for most real-world solutions — especially ones found in biochemistry. Most biological systems have a pH near 7, although significant deviations may exist (for example, the pH in your stomach is close to 1).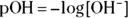 . You can calculate the hydroxide ion concentration from the hydrogen ion concentration and the K w(equilibrium constant) relationship:
. You can calculate the hydroxide ion concentration from the hydrogen ion concentration and the K w(equilibrium constant) relationship: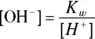
 A useful shortcut to get from pH to pOH is the following relationship:
A useful shortcut to get from pH to pOH is the following relationship: 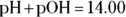 for any aqueous solution
for any aqueous solution 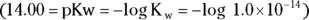 .
.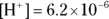 , its pH would be
, its pH would be