This term is a consequence of entropy, which states that a system will tend spontaneously towards the greatest degree of disorder, just as a drop of ink diffuses into a glass of water into which it has fallen. Consequently, if the flow rate is too slow, the compounds undergoing separation will mix faster than they will migrate. This is why one must never interrupt a chromatographic analysis, even temporarily, once it is underway, as this could negatively impact efficiency.
Liquid (stationary phase) term C = C G+ C L
Term C , which is related to the resistance to mass transfer of the solute between the two phases, becomes dominant when the flow rate is too high for an equilibrium to be attained. Local turbulence within the mobile phase and concentration gradients slow the equilibrium process ( C S⇔ C M). The diffusion of solute between the two phases is not instantaneous, and thus the solute will be carried along out of equilibrium. No simple formula exists which takes into account the different factors involved in term C . The parameter C Gis dependent upon the diffusion coefficient of the solute in the gaseous mobile phase, while the term C Ldepends upon the diffusion coefficient in the liquid stationary phase.
In practice, the values for the coefficients A , B , and C can be obtained by making several measurements of efficiency for the same compound undergoing chromatography at different flow rates, since flow rate and average linear speed are related. Next, the hyperbolic function that best satisfies the experimental values can be calculated, preferably by using the multiple linear regression method.
In 1958, Golay proposed a modified relationship reserved for capillary columns used in gas chromatography:
(1.39) 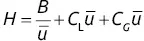
Equation ( 1.39) leads to the minimum value of HETP for a column of radius r , if the retention factor of the test compound is known. The coating efficiency can then be calculated as the ratio between the value found and that deduced from the efficiency ( H = L / N ) obtained from the chromatogram, multiplied by 100.
(1.40) 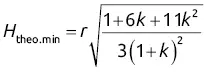
Another, more recent equation (1977), the Knox equation, is applicable to various types of liquid chromatography and includes the adjusted height h (see Eq. (1.21)):
(1.41) 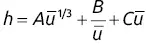
1.11 OPTIMIZATION OF A CHROMATOGRAPHIC ANALYSIS
The utility of analytical chromatography depends on its ability to separate analytes, whether to identify or quantify them. Particularly in the latter case, peak areas must be defined with precision. Since many parameters affect the quality of results, there are a certain number of requirements regarding the resolution between solute peaks, the speed of analysis (elution time, then the time needed to return to the initial conditions of analysis if different from the final conditions), and even the column’s capacity (to avoid saturation). To do so, we often use software to simulate the separation by adjusting various physico‐chemical factors such as temperature, flow rate, and composition of the mobile phase.
Due to the various parameters interacting together, the chromatographer must always find a compromise between resolution , speed , and capacity , which we often represent with the image of a triangle whose corners correspond to these three opposing parameters ( Figure 1.11). In any optimization, the goal is to ensure sufficient separation of the compound(s) of interest in a minimum amount of time. The resolution and the elution time are thus the two most important dependent variables to consider. However, chromatography is a slow type of analysis, since we have to wait for the elution of the most retained compounds and then for the column to return to the initial conditions before we can start the next analysis. If the resolution is very good, then optimization consists in saving time, for example by choosing a shorter column. Remember that resolution varies according to the square root of column length (see parameter N in Equation ( 1.35) and Figure 1.9).
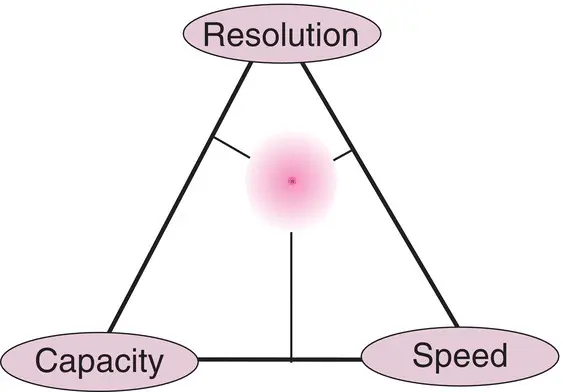
Figure 1.11 The chromatographer’s compromise triangle between resolution, speed, and capacity. Any chromatographic analysis is subject to three contradictory criteria. If we favour one of these three criteria, the two others are necessarily disadvantaged. When we look for good selectivity, we must fall close to the resolution corner of this triangle. The shaded area indicates the domain corresponding to analytical chromatography based principally upon the five parameters K , N , k , α , and R .
Often, in a chromatographic assay, we are only interested in one or two compounds present in the sample. If we have a selective detector adapted to the analytes in question, we can facilitate the optimization stage, since the resulting chromatogram will be greatly simplified. In other cases, however, we try to separate the greatest possible number of compounds from the initial mixture.
1.12 CLASSIFICATION OF CHROMATOGRAPHIC TECHNIQUES
Chromatographic techniques can be classified according to various criteria: the physical nature of the phases, the process used, or physico‐chemical phenomena occurring during separation, etc. The following classification has been established by consideration of the physical nature of the two phases involved.
1.12.1 Liquid Chromatography (LC)
This type, in which the mobile phase is a liquid, is the oldest known form of chromatography, with the earliest applications involving preparative methods of separation (for which it is still used today). Depending on the nature of the stationary phase (nature of interactions determining the separation of solutes), we can distinguish between liquid/solid chromatography (LSC), liquid/liquid chromatography (LLC), ion chromatography (IC), and size exclusion chromatography (SEC) (also known as gel permeation or gel filtration chromatography depending on the nature of the stationary phase).
Liquid/solid chromatography (LSC)
The stationary phase is a permeable solid medium to which the solute molecules adhere through the dual effect of physisorption and chemisorption. The physico‐chemical parameter involved here is the adsorption coefficient . It can be considered as the ancestor of LSC. We can include thin‐layer chromatography, which differs from LSC in the distribution of the stationary phase but which proceeds from the same separation principle.
Liquid/liquid chromatography (LLC)
The stationary phase and the mobile phase are both liquids. The stationary phase is most often a polymer fixed on an inert support, generally by grafting active species with covalent bonds, ensuring a long lifespan for these columns. The separation relies upon the partition coefficient of the solutes between the two phases (a phenomenon comparable to a liquid–liquid extraction in a separatory funnel, a well‐known process in laboratories). This category also includes centrifugal partition chromatography, which is used in preparative chromatography.
Читать дальше

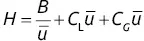
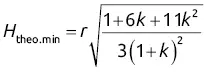
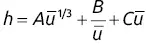



![Евгений Матерёв - Музеи… или вдохновляющая музыка The Chemical Brothers [litres самиздат]](/books/437288/evgenij-materev-muzei-ili-vdohnovlyayuchaya-muzyka-th-thumb.webp)







