Brain scans of the affected Pakistani children were normal, as were biopsies of the sural nerve. Unlike the Norrbotten syndrome patients we discussed in chapter 3, these kids had normal numbers of the various sensory fiber types, ranging from the speedy A-alpha fibers to the poky C-fibers. Analysis of their DNA revealed that all six of the children carried mutations in the same gene, SCN9A, which directs production of a voltage-sensitive sodium channel that is essential for the propagation of electrical signals in neurons. However, SCN9A’s expression is almost entirely limited to those neurons that convey pain information from the skin and the viscera. (Other neurons utilize different sodium-channel genes.) Consequently, the neurons that convey pain signals to the spinal cord and brain are present, but they are electrically silent. That’s why the children’s sural nerve biopsies look normal, while their loss of pain sensation is profound. When kidney cells were used to artificially express mutant SCN9A DNA from these patients, no sodium channel function could be seen at all, only a flat line of electrical current (figure 6.1). At present there is no way to restore this current and hence no effective therapy for congenital insensitivity to pain.
The particular mutations in the SCN9A gene that give rise to congenital insensitivity to pain are of a type called loss-of-function mutations: They cause the gene to fail to produce a working protein, resulting in a flat-lined sodium current in pain-sensing neurons. These mutations are inherited in a recessive fashion, so that an individual must have two copies of the SCN9A gene with the relevant mutation, one from each parent, in order to have the disease. This explains why it is much more common in families that practice cousin-marriage. Other types of gene mutation are called gain-of-function mutations. In the case of the SCN9A gene, gain-of-function mutations produce voltage-sensitive sodium channels with aberrant properties. An individual needs to inherit only a single mutant gain-of-function gene to produce a terrible effect on pain perception.
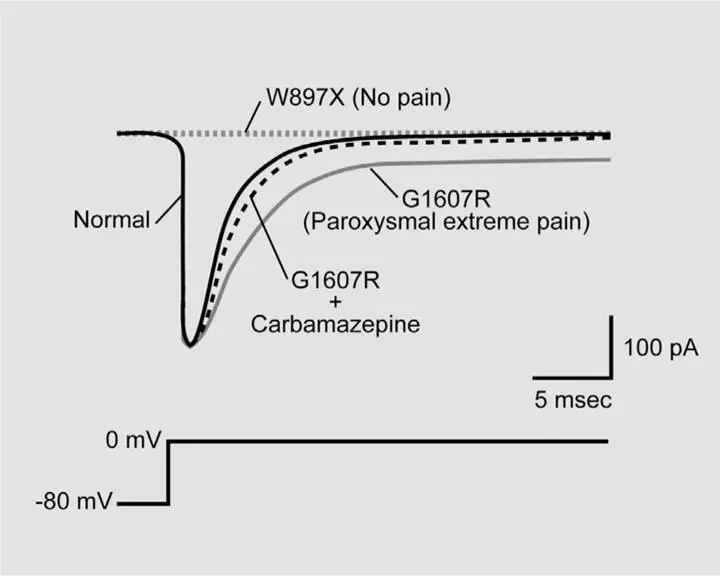
Figure 6.1Mutations of the SCN9A gene can produce dramatic alterations of pain perception and voltage-sensitive sodium current. Here, different versions of the SCN9A gene have been artificially introduced into kidney cells. Sodium currents are evoked by using an electrical circuit to rapidly shift the voltage across the cell membrane from -80 millivolts to 0 millivolts, roughly mimicking what occurs when a neuron fires an electrical spike to send information along its axon. Normal SCN9A produces a typical sodium current: a brief inward flux of positive current that completely terminates within a few milliseconds. SCN9A from a patient with congenital insensitivity to pain harbors a mutation called W897X, which renders the channel completely nonfunctional, leading to no current at all. SCN9A from a patient with paroxysmal extreme pain disorder bears the mutation G1607R and results in sodium current that activates normally but inactivates slowly and incompletely, leaving a long tail of persistent current. The drug carbamazepine can partially reverse this effect and is useful in reducing the symptoms of paroxysmal extreme pain disorder. There are several different mutations that can give rise to congenital insensitivity to pain, but all produce a flat-line current. Likewise, there are several different mutations that can give rise to paroxysmal extreme pain disorder, but all produce sodium current that inactivates incompletely.
It usually starts soon after birth, often with the first bowel movement: A baby startles, and its face gradually assumes a look of sheer terror. Screaming inconsolably, the newborn clings to an adult. Her body stiffens and flushes strongly, and her face contorts into a grimace. These attacks, which last for minutes and can occur several times a day, are often triggered by some trivial act involving the mouth or the anus: feeding, wiping, or inserting a rectal thermometer. Obviously, babies cannot communicate, but as these children grow, they describe pain that frequently begins in the area of the anus, the jaw, or the eye and then spreads. At once burning, stabbing, and diffuse, the pain is described by adult sufferers as the worst agony imaginable. All the mothers who have this condition reported that it is far worse than going through labor. Most of them said that if they knew their fetus was affected with this condition, they would have aborted it, as they could not bear the prospect of their child enduring such pain attacks. 3
This condition has been named paroxysmal extreme pain disorder, and it, too, results from a gain-of-function mutation in the SCN9A gene. Figure 6.1 shows sodium current from mutant SCN9A derived from a patient with the disorder. When the cell is depolarized, the sodium current activates normally but inactivates slowly and incompletely. The result is that the pain-sensing neurons are now like machine guns with a hair-trigger: Stimuli that might make normal neurons fire one or two electrical spikes now make them fire a sustained burst. Because of this aberrant electrical signaling, even an innocuous stimulus can trigger a bout of extreme pain. Fortunately, there is a drug called carbamazepine that can promote the inactivation of voltage-sensitive sodium channels, including those derived from the SCN9A gene. In some patients carbamazepine can produce complete relief, and in many others it can reduce the frequency and severity of the pain attacks. 4
Even without carbamazepine treatment, however, sufferers of paroxysmal extreme pain disorder typically live full lives. Most have children and careers and a normal life span. In a sense this is counterintuitive: If you were forced to choose between two mutant forms of SCN9A, one that would render you painless but would almost ensure that you died young, or another that would give you random attacks of mind-numbing pain for your entire normal life span, which would you prefer?

Imagine that you’re walking around the house without shoes and you slam your toes into a heavy wooden chair leg. The pain doesn’t come all at once. First, there’s a sharp pain that’s localized to the specific toes that you struck, which diminishes quickly. Then you can sing “In-a-gadda-da-vida, baby” before the second wave of throbbing and diffuse pain arrives. The first wave of pain is carried to the spinal cord by a mixture of medium-diameter myelinated A-delta fibers that can transmit electrical spikes at a speed of about 70 miles per hour and large-diameter myelinated A-beta fibers at 150 miles per hour. The second wave of pain is conveyed by small-diameter C-fibers that transmit signals much more slowly, at about 2 miles per hour. All areas of the skin (and most parts of the viscera) are innervated by both fast and slow pain fibers (figure 6.2). The time difference between the first and second waves of pain is most noticeable for locations that are distant from the brain, like the toes. By comparison, facial pain also has fast and slow components, but the gap between when the two are sensed is much smaller, and so the two waves of pain are not as clearly distinguished. Of course this lag time for the second wave of pain is even more pronounced in larger animals. For a ninety-foot-long dinosaur (like a diplodocus) who whacked her tail on a floating log, the first wave of pain would have arrived in about one second, while the second wave would have taken a full minute to reach the brain and be perceived. 5
The initial wave of pain is fast, precise, and discriminative; it provides information related to immediate threat and guides a withdrawal response. Often you’ve already initiated the withdrawal response and let an expletive fly by the time the second wave of pain arrives. Imagine grabbing a hot pot handle: The first wave of pain causes you to release the handle immediately, and you are already waving your hand in the air to soothe the first pain as the second wave of pain is felt. The second wave of pain is slow to start, slow to end, and is poorly localized. It can have an aching, burning, or throbbing quality. Secondary pain demands sustained attention and motivates behaviors that reduce further injury and promote recovery (like favoring an injured leg while walking).
Читать дальше














