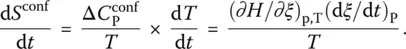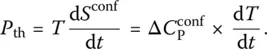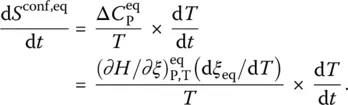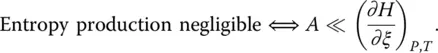From the configurational Gibbs free energy calculated for the lattice‐hole model, one readily simulates with the definition ( 3) of the affinity its variations upon vitrification (cooling) and structural recovery (heating) [19]. Thermodynamic data measured on o ‐terphenyl may be used to simulate the corresponding affinities during temperature ramps ( Figure 5): cooling at 0.3 K/min from an initial temperature of 255 K is followed by heating at the same rate, and then by further cooling at 0.5 K/min preceding final heating at 20 K/min. That the supercooled liquid begins to lose internal equilibrium from 248 K is indicated by the departure at this temperature of the affinity curve from the zero line, which represents the maximum (equilibrium) value of the affinity during cooling. The affinity then linearly decreases with temperature below 240 K in the glassy state, with higher values for slower cooling as a result of lower glass transition temperatures. Upon heating, the affinity begins to increase linearly according to the same line pathway before crossing the equilibrium line. It then exhibits a peak whose position shifts toward higher temperatures and whose magnitude and width increase with the heating rate in ways such that the configurational heat capacity and the other thermodynamic coefficients can be simulated [19].

Figure 5 Simulated affinities of o ‐terphenyl in the glass transition range upon cooling and heating as calculated from the lattice‐hole model. Solid circle for −0.3 K/min and solid square for −0.5 K/min; empty circle for +0.3 K/min and empty square for +20 K/min. The horizontal line represents equilibrium ( A = 0). Inset: entropy production rates calculated from the previous affinities. Solid circle upon cooling and empty circle upon heating.
The entropy production can also be calculated from Eq. (4)(inset in Figure 5). In agreement with previous results [18], it shows a single peak upon cooling but two peaks upon heating. With respect to the experimental data, the advantage of the calculation is thus to distinguish clearly two contributions to the entropy produced when heat is brought to the material. The first peak is associated with a decrease of the configurational energy of the system, which is taking place because of the delay introduced by the relaxation time, even though heat is being supplied. As to the second peak, it is in contrast associated with the configurational energy necessary to recover internal equilibrium in the supercooled liquid state.
Here the wording “is associated” instead of “represents” is necessary because the entropy produced and configurational entropy changes necessarily differ as a result of the irreversible nature of the glass transition. Whereas the entropy production is the product of the thermodynamic force and flux (see Eq. (4)), the variation of the configurational entropy is written as, see Eqs. ( 8) and ( 13):
( 17) 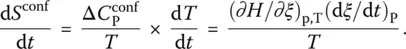
The rate of entropy production thus reflects the spontaneous or irreversible microscopic processes that take place within the system during relaxation. As dictated by the Second Law of thermodynamics, it is always positive whether upon cooling or heating ( Figure 5, inset). Physically, it can be thought of the heat irreversibly generated by friction at a microscopic scale. The resulting thermal power P i= Tσ i, where σ iis the entropy creation in Eq. (4), is produced much too quickly to be compensated instantaneously by an exchange of heat with the surrounding heat bath. Under this circumstance, this is why an effective or fictive temperature can be defined. This surrounding heat bath is sometimes called the phonon bath since it is characterized by fast or vibrational modes.
On the contrary, the change in configurational entropy is a reversible process related to the heat exchanged with the surrounding heat bath whose relevant thermal power is:
(18) 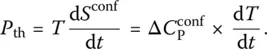
Because the configurational entropy becomes constant upon vitrification, its variations have vanished (i.e. the configurational heat capacity) below the glass transition range. Above this range, in the supercooled liquid state, they of course differ from zero as indicated by
(19) 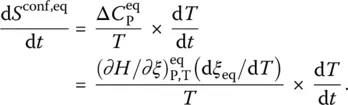
In the transition range, the variations of the configurational entropy of the system are consequently positive or negative upon heating and cooling, respectively. As already evaluated long ago either theoretically [9] or experimentally [20, 21], the entropy produced is generally negligible with respect to the configurational entropy changes. The integration of the heat capacity curves measured by calorimetry is thus a pertinent way to access to the absolute value of the residual entropy at 0 K [3]. As seen from a direct comparison of Eqs. ( 4) and ( 17), arriving at this conclusion is tantamount to neglecting the affinity with respect to the enthalpy of advancement of the configurational change at every temperature:
(20) 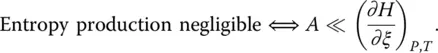
Relaxation times that depend only on temperature and pressure have been considered. Nevertheless, the complexity of microscopic structures in glasses implies the existence of a distribution of relaxation times. Relaxation processes then also depend on the instantaneous state of the system itself and, thus, on its history as, for instance, described by the Tool–Narayanaswamy–Moynihan model ( Chapter 3.7, [22, 23]). A consequence is that some nontrivial relaxation processes can take place well below the glass transition range. Hence, it is interesting to study such a time‐dependent process termed physical aging , which has a practical relevance through its possible effects on glass properties.
The process can be illustrated with DSC scans for PVAc ( Figures 2and 6). If the sample is cooled down at some rate to 297 K, i.e. about 10 K below the calorimetric T g, and its temperature is kept constant for a time interval t a, then its enthalpy (and entropy) relax to their lowest equilibrium values for the temperature (and pressure) considered. Experimentally, physical aging manifests itself as differences between the areas of DSC scans upon heating recorded for samples annealed (72 hours at 297 K) and not annealed. For the experiment without annealing, the lowest temperature of 278 K has been directly reached (see cooling curve in Figure 6). The amount of enthalpy relaxed during aging is equal to the difference between the light gray and dark gray areas in Figure 6. Of course, for a given annealing temperature, such a difference increases with the aging duration. As recently carried out on polymeric glass‐formers annealed at relatively low aging temperatures for a very long time, calorimetry (DSC) can bring to light two different timescales for glass equilibration, revealing the complexity and richness of relaxation processes well below T g[24].
Читать дальше