As a child, I experienced many minor injuries that were equally or even more painful, from skinned knees to a bumped head. But those came as surprises; because they weren’t anticipated, they didn’t seem such a major concern. The brief and minor pain of the injection, however, was amplified enormously by my mounting fearful expectations and my memories of the previous traumatic injections.
These accounts of heroic combat and pediatric melodrama demonstrate how cognitive and emotional factors can dull or heighten pain perception. Can we understand this cognitive and emotional modulation of pain perception in terms of the distributed network of pain-processing centers in the brain? In general, the answer is yes, but many of the details remain to be worked out. One key insight is that the brain can send signals down to the pain-transmitting neurons in the dorsal horn of the spinal cord that can say either, “Speak up and say it louder!” or, “Shut up! Dial down the pain information!” The truly amazing fact is that the brain is exerting control over the information that it receives . It is not just taking in all the data and then biasing its perceptions and responses based upon the present emotional or cognitive state; rather, through these descending nerve fibers, it is controlling which sensory information will be received from the spinal cord. This is a weird and counterintuitive state of affairs. The brain actively and subconsciously suppresses or enhances pain information on a moment-to-moment basis. It spins the media, so to speak. This realization that in many cases we have access only to self-censored information is somewhat disconcerting to those of us who like to feel that we have access to unfiltered reality to guide our rational thoughts. 21
As we have seen, information from both the sensory-discriminative and the affective-emotional regions of the brain’s pain-processing network converges in several locations, including the anterior cingulate cortex, the insula, the prefrontal cortex, and the amygdala. These regions then send signals to a structure in the brain stem called the periaqueductal gray (figure 6.6), which in turn excites structures farther down in the brain stem called the locus coeruleus and the rostroventral medulla. 22It’s these latter regions that finally send axons down to the dorsal horn of the spinal cord, where they form synapses that can either suppress or boost signaling from peripheral pain-sensing fibers. There are some cells in the rostroventral medulla, called on-cells, that increase firing, and others, called off-cells, that decrease their firing in response to pain. Increases in on-cell activity boost transmission of pain signals in the spinal dorsal horn and enhance pain perception, while increases in off-cell activity have the opposite effect. 23It is this circuit that enables the brain to dial the incoming pain information up or down.
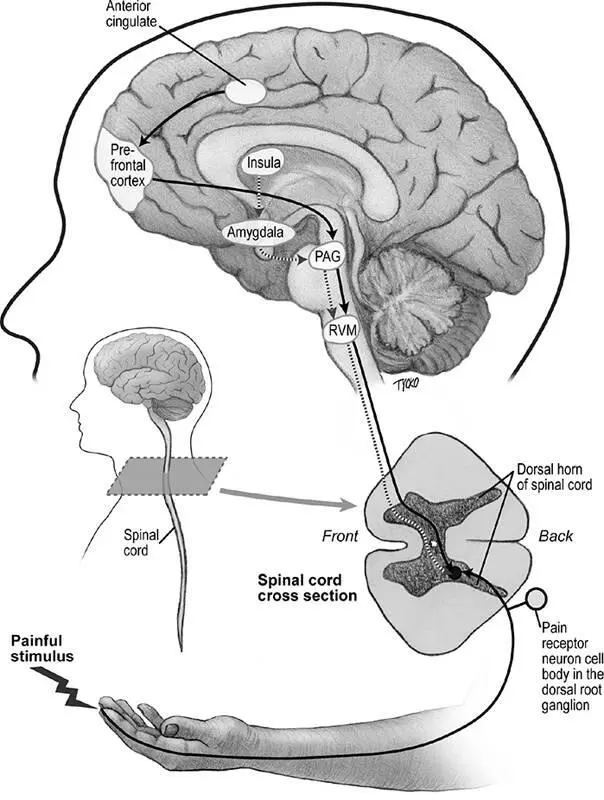
Figure 6.6Descending pathways, running from the brain to the spinal cord, have an important role in the cognitive and emotional modulation of pain. The key way stations for this descending information in the brain stem are the periaqueductal gray region (PAG) and the rostroventral medulla (RVM). This diagram is a simplified representation. In particular, the synaptic actions of several neurotransmitters released in the spinal dorsal horn by descending fibers are varied. © 2013 Joan M. K. Tycko
The pain-controlling properties of the opium poppy and its derivatives, like morphine, have been known since at least 3400 BC in Sumeria (present-day southern Iraq). The Sumerians called the poppy Hul Gil (“joy plant”) and soon exported it to Assyria and Egypt, from which it spread widely. Morphine works by mimicking the action of the brain’s own morphinelike molecules, the endorphins and enkephalins. The receptors for these opioids are widespread in the body and in the nervous system, but a particular type, called the mu-opioid receptor, is concentrated in the descending pain pathways, in the periaqueductal gray, the rostroventral medulla, and the superficial layers of the spinal dorsal horn. 24Microinjecting morphine into the periaqueductal gray region is sufficient to produce a strong analgesia, mediated by exciting off-cells and inhibiting on-cells in the rostroventral medulla. The pain-blunting effect of the periaqueductal gray region is so strong that carefully controlled electrical stimulation of this region can replace chemical anesthetic agents during surgery. Some people suffering from pain that does not respond well to drugs have had electrodes implanted in their periaqueductal gray area to enable them to self-stimulate their brains with a handheld device for pain relief. Pain relief via periaqueductal gray self-stimulation can be blocked by drugs that interfere with mu-opioid receptors, implicating endorphins/enkephalins in the analgesic effect.
Armed with this knowledge of descending pain modulation, let’s return to our examples of Private Turner, who, in the heat of battle, didn’t realize that he had been shot, and your youthful narrator, crying out at the minor pain of a routine injection. Clearly, the behavior in both cases was in part a matter of attentional focus. Private Turner was focused on saving his buddies while under fire; his thoughts were not on his own body. In the laboratory, when subjects are distracted from a pain stimulus (such as by being asked to respond to questions or perform a mental task), their rating of the pain intensity decreases. This response is associated with decreases in activity in the primary somatosensory cortex and the insula. Conversely, when subjects are instructed to focus on their pain, their ratings of pain intensity rise, reflecting increased activity in these same brain regions. Importantly, the emotional component of pain perception, indicated by ratings of pain unpleasantness, is largely unaltered by focused attention. 25
In the confines of the laboratory, we can design experiments that selectively engage the attentional and emotional mechanisms. However, in the real world, these are not so easily separated. Private Turner was distracted by the ongoing firefight and his efforts to save his fellow soldiers, but this diversion was not emotionally neutral. Rather, it was fraught with emotions: fear, compassion, pride, and his deep bonds with his fallen comrades. In the doctor’s office, as I waited for the injection, my focus on the impending prick of the needle was not emotionally neutral either, but laced with dread, seeded by frightening memories.
Negative emotions enhance perception of pain, but they do so in a way that is perceptually and anatomically different from attentional effects. Negative emotions boost pain-evoked activity in the anterior cingulate cortex and increase the ratings of pain unpleasantness, but not their intensity. The end result is that both the sensory-discriminative and the affective-emotional aspects of pain perception are subject to cognitive and emotional modulation. And in the real world, the cognitive and the emotional are highly interdependent. For example, pain unpleasantness, when endured persistently, engages the prefrontal cortex, a region involved in rumination and reflection about the future implications of persistent pain: How long will this pain endure? Will the pain start again? Is it under my control or not? How safe am I? These prefrontal cortex-based ruminative processes contributed to my youthful anxiety about the impending injection and exerted their effects, in part, through descending pathways to the spinal cord. In many people, this sets up a vicious cycle, a positive feedback loop in which rumination boosts pain unpleasantness, which in turn triggers additional anxiety and rumination. This is why tranquilizers (like the benzodiazepines) can be useful in treating pain, particularly chronic pain. Even if these drugs don’t affect pain perception directly, they reduce anxiety and therefore help to attenuate pain unpleasantness and break the positive feedback loop. On average, people suffering from mood disorders are at significantly greater risk for developing chronic pain. 26
Читать дальше













