Sample Analysis
Nasal Cytology
Nasal cytology can be performed by pressing a microscope slide directly onto the nose of an animal or by obtaining material with a cotton swab and spreading it onto a slide. It is non‐invasive and inexpensive; however, there are substantial limitations to the amount of information obtained from this test. Its primary usefulness is in the diagnosis of nasal fungal infection with cryptococcosis (see Chapter 4). If eosinophils are noted in a dog with cough and nasal discharge, the possibility of eosinophilic lung disease should be considered (see Chapter 6). Accuracy in the diagnosis of nasal aspergillosis or neoplasia is poor unless an endoscopically obtained sample from a fungal plaque (De Lorenzi et al. 2006) or from a mass lesion is examined cytologically.
Bacterial culture of nasal discharge is not recommended. In fact, since most infections in the nose are secondary rather than primary, the value of culturing a deep nasal flush or culturette sample can also be questioned because of the difficulty in distinguishing normal flora from pathogens or opportunistic invaders. Interpretation of bacterial cultures is more difficult in animals that have been on multiple courses of antibiotic trials because this tends to favor the development of resistant species. Despite this, bacterial culture from a deep nasal swab or flush can provide some direction for antibiotic therapy in cats with chronic rhinosinusitis. The easiest way to obtain a sample from the nose of a cat is to direct a sterile cytology brush (Cytosoft™, Medical Packaging Co., Panorama City, CA) into the ventral portion of the nasal cavity and gently twirl the brush to collect cells and secretions. A deep nasal flush can be performed by inserting an 8 French red rubber catheter halfway into the ventral portion of the rostral nasal cavity, occluding the soft palate digitally, and instilling and aspirating 1–2 ml of sterile non‐bacteriostatic saline. Culture for aerobic bacteria and Mycoplasma spp. is recommended.
A study on the utility of fungal cultures in the diagnosis of canine nasal aspergillosis reported moderate sensitivity (77%) but high specificity (100%; Pomrantz et al. 2007). In that study, the material submitted for culture was from a visualized fungal plaque and therefore a low number of false‐positive values would be expected. In cases of cryptococcosis, culture and speciation can be helpful in determining the epidemiology of the infection, although subclinical colonization of the nasal cavity has been reported in 7–14% of dogs and cats (Malik et al. 1997).
Histopathology provides the diagnosis of aspergillosis in cases in which a plaque lesion has been sampled by direct visualization; however, a random sampling of the nasal mucosa or a blindly collected sample may not contain fungal elements, leading to a false‐negative biopsy sample. Neoplasia can be difficult to confirm with a biopsy, because a rim of necrosis and inflammation often surrounds neoplastic cells. In some instances, neoplastic cells can be detected in histopathology from a mass in the nasopharynx, but they are not evident in a sample from within the nasal cavity. This seems particularly common in feline nasal/nasopharyngeal lymphoma.
Normal tracheal wash cytology is composed of ciliated columnar to cuboidal epithelial cells and rare macrophages or inflammatory cells. Normal BAL cytology contains primarily alveolar macrophages (65–75%), with 5–10% neutrophils, lymphocytes, and eosinophils; although in a cat, 18–25% eosinophils can be normal ( Figure 2.25). Normal macrophages can exhibit variable appearances depending upon stimuli present within the lung, such as infection or inflammation. Activated macrophages contain intracellular inclusions, can appear foamy, and may be engulfing granulocytes or particulate matter in comparison to quiescent macrophages. Normal BAL cell counts have been reported for the dog and cat and are approximately 300–400 cells/μl. Higher cell counts and/or an altered percentage of cells present within the lavage fluid are indicative of lower respiratory tract pathology. Little progress has been made in standardizing the procedures for BAL, for handling fluid after collection, or for interpreting cell counts. Inflammatory cells can be trapped in mucus and straining a sample will artificially lower cell numbers. While hypercellular BAL fluid could be considered suggestive of an inflammatory process, not all laboratories report a total nucleated cell count and some do not supply a differential cell count. Whenever possible, these numbers should be requested from the clinical pathologist to aid in interpretation.
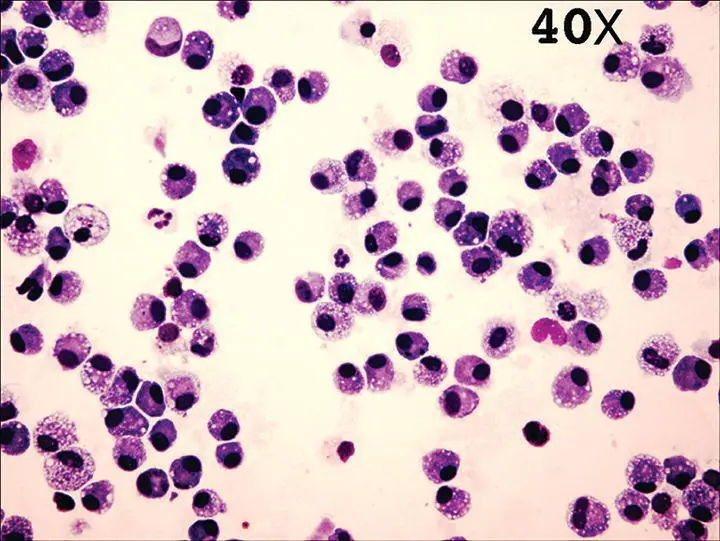
Figure 2.25 Cytocentrifuge of a normal bronchoalveolar lavage sample (40×) reveals primarily macrophages (65–75%), with the remaining percentage made up of neutrophils, lymphocytes, and eosinophils.
Suppurative inflammation is characterized by > 10% neutrophils and can represent pure inflammation, as in chronic bronchitis in the dog or cat, or can be an indicator of infection, as with pneumonia. The finding of degenerate neutrophils containing intracellular bacteria is a reliable indicator of bacterial pneumonia. Pyogranulomatous inflammation (activated macrophages and increased neutrophils) can be an indicator of fungal disease, and samples should be closely screened for cytologic evidence of fungal organisms (see Chapter 6). This type of inflammation can also be seen with very chronic bacterial pneumonia or bronchiectasis.
Eosinophilic inflammation is characteristic of some forms of feline asthma and can also be found with airway parasitism due to Aelurostrongylus , Capillaria , Crenosoma , or Paragonimus , in heartworm disease, or with larval migration of gastrointestinal parasites ( Toxocara ). Airway eosinophilia is a prominent feature of eosinophilic bronchopneumopathy in dogs.
Hemorrhagic inflammation can be found with rodenticide intoxication (vitamin K antagonists), heartworm or Paragonimus infection, foreign body, or trauma. Neoplasia and thromboembolic disease can also result in pulmonary hemorrhage. Evidence of previous airway hemorrhage (indicated by macrophages ingesting red blood cells or hemosiderin‐laden macrophages) can be found in some dogs with congestive heart failure, lung neoplasia, or with exercise‐induced pulmonary hemorrhage.
In rare instances, malignant cells can exfoliate into the airways with pulmonary carcinoma (primary or metastatic) or in pulmonary involvement with lymphoma and will be detected in BAL fluid. Characteristics of malignancy are similar to those in other tissues, including loss of contact inhibition, variation in cell size or nuclear size and shape, increased nuclear‐to‐cytoplasmic ratio, basophilia, multi‐nucleate cells, or frequent cells undergoing mitosis. Neoplastic transformation can be difficult to confirm because dysplastic changes associated with severe inflammation can mimic neoplastic atypia. In addition, some lung tumors have a necrotic center or can become infected, which can complicate interpretation of abnormal‐appearing cells or the presence of bacteria. Review of several samples can be required to confirm the presence of an underlying neoplasm. If a mass lesion is noted in the airway lumen during bronchoscopic examination, an endoscopic biopsy sample should be obtained.
Positive bacterial and mycoplasmal cultures must be interpreted in conjunction with cytology. The presence of squamous cells or Simonsiella bacteria is suggestive of upper airway contamination. In these cases, it is not uncommon to observe growth of two to four types of aerobic bacteria (usually oral flora) with or without Mycoplasma . Because approximately one‐third of healthy dogs and almost three‐fourths of healthy cats can have positive tracheal cultures ( Table 2.5), strict attention should be paid to upper airway contamination of BAL samples and appropriate interpretation of BAL cultures. Quantitative bacterial cultures provide evidence for bacterial infection when > 1.7 × 10 3bacterial colony forming units per milliliter of fluid grow on culture. In addition, detection of more than two intracellular bacteria in any of 50 examined high‐power fields is a reliable indicator of lower respiratory tract infection (Peeters et al. 2000), although approximately one‐quarter of cases lack intracellular bacteria (Johnson et al. 2013). Lower respiratory tract infections are often characterized by culture of several species of bacteria, including anaerobes (Johnson et al. 2013).
Читать дальше













