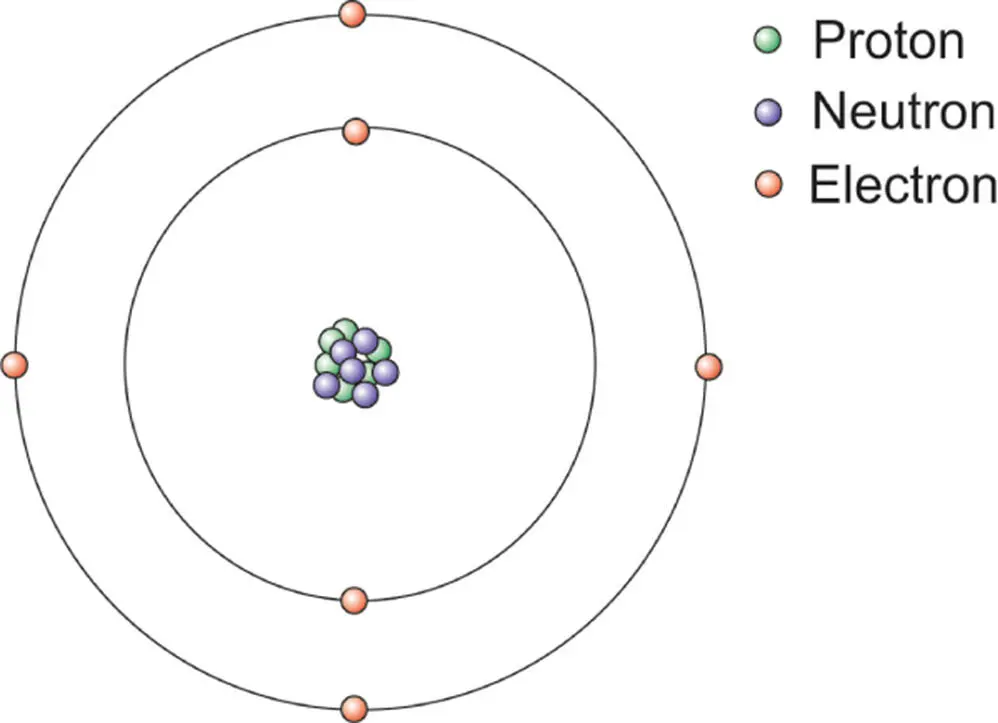
Figure 3.1 A very simplified depiction of the structure of a typical atom showing the nucleus and electrons. Note that this is not to scale. The electrons are in orbitals about 10 000 times further out than the diameter of the nucleus. Electrons occupy energy levels, depicted here as circles.
In the nucleus of any given atom, the total number of protons is given by Z . This is also called the atomic number. The number of protons defines the element. So, for instance, in the biologically important atom carbon, the number of protons is 6. The element has an atomic number of 6. This defines it as the element carbon.
The nucleus also contains neutrons (a total number N ). For any given atom, the atomic mass is the total number of protons and neutrons. In all stable elements, there are at least an equal number of neutrons as protons, and in many elements there are more neutrons. An exception is hydrogen, which contains just one proton. Returning to carbon, most carbon atoms have 6 neutrons, so with its 6 protons this gives it an atomic mass numberof 12. Protons, because they are positively charged, tend to repel each other. You might think that this would cause the nucleus to fall apart. However, the nucleus is held together by the strong nuclear force that operates over very small distances and overwhelms the electrostatic repulsion, explaining why the ball of positively charged protons in the atomic nucleus remains together.
There is a caveat to the simple picture described above. Not all atoms of the same element have the same number of neutrons. The value of N can vary. Atoms that have the same number of protons but different numbers of neutrons are called isotopes.
As we will see later, isotopes turn out to be very important tools for astrobiologists. Life has a tendency to prefer lighter isotopes since they are ever so slightly more reactive than heavier isotopes of the same element. This means that living things are often enriched in light isotopes of elements. This isotopic fractionationcan be used as a signature of life in ancient Earth rocks and it is a means by which one might try to detect life elsewhere.
Later in the textbook, we return to isotopes to consider in more detail how they can be used to search for life. In the meantime, it is worth pointing out that this is another example of how a fundamental understanding of matter is necessary to understand not just biology, but even the technologies one might use to search for life elsewhere.
As carbon is so important in terrestrial biology, it is the first useful example of isotopes. The element has three important isotopes depicted in schematic form in Figure 3.2. There are actually 15 known isotopes of carbon, but these three are of importance in biological processes. Isotopes are often designated by showing the atomic mass number as a superscript on the left-hand side of the element. The three main isotopes of carbon are:
Carbon 12 (12C): 6 neutrons, 6 protons (a stable isotope, which makes up 98.9% of the carbon on Earth).
Carbon 13 (13C): 7 neutrons, 6 protons (a stable isotope, which makes up 1.1% of the carbon on Earth).
Carbon 14 (14C): 8 neutrons, 6 protons (an unstable isotope, accounting for one part per trillion!).
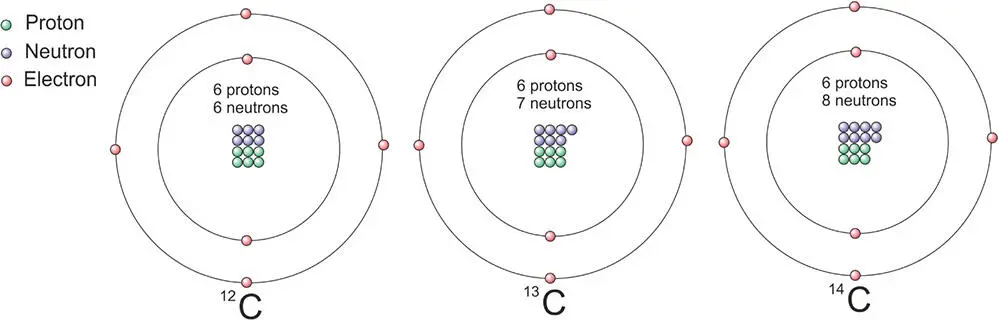
Figure 3.2 Schematic diagram of the three isotopes of carbon. The number of neutrons varies.
Carbon 12 ( 12C) is the most common form of carbon and constitutes more than 98% of carbon in living matter. 13C is rarer but, like 12C, it is a long-lived stable isotope. By contrast, 14C, with two additional neutrons, is not stable. It is called a radioactive isotope or radioisotope. One of the neutrons decays into a proton with the release of an electron and the atom becomes nitrogen 14 ( 14N). It transforms into a different element because it has now gained a proton which, you'll recall, defines the type of element. This decay has a half-lifeof 5730 years. In other words, after 5730 years, half of a sample of 14C will have decayed.
The unstable nature of 14C means that it is a small proportion of carbon isotopes but, despite this, it turns out to be enormously useful. As it decays with a known rate, it can be used to determine the age of ancient material from living things that contain carbon (“carbon dating”) such as bones. Living things constantly take up new 14C when they are alive, but once they are dead, they no longer actively take up carbon through metabolism. The 14C in the now dead organic matter begins to radioactively decay, allowing us to back-track and work out when that decay must have started and thus how old the object is. Later in the book, we see in more detail how radioisotopes can be used to put absolute dates on the fossil and geological record of Earth and other planetary bodies.
3.4 Electrons, Atoms, and Ions
An atomic nucleus is surrounded by electrons, and they orbit at a great distance from the nucleus relative to the diameter of the nucleus itself. A typical atomic nucleus has a diameter of ∼10 −14m, whereas a whole atom has a radius of 2 × 10 −10to 5 × 10 −10m. Atoms have the same number of negatively charged electrons as the positively charged protons so that overall, an atom has no charge. It is neutral.
Electrons have something of a split personality. They exhibit particle-like properties and the behavior of waves, like light. Therefore, to consider electrons orbiting the nucleus in the same sense that a planet orbits a star is not quite technically correct. It is sufficient as a general description of an atom, and for simplicity they are often shown as tiny particles orbiting a nucleus, just as they are in the figures in this chapter. The dual wave and particle-like properties of electrons mean that they actually occupy fuzzy domains around the nucleus determined by probability distributions that, to add confusion, are called “orbitals.” The shapes of orbitals are determined by this wave – particle duality. The structure of electron orbitals will not be considered in detail in this book, but it is worth briefly spending some time pointing out their main features as they explain why atoms react at all and why they bond in particular ways. This is essential for grasping the fundamental atomic and molecular structure of life.
Each orbital can only take a maximum of two electrons. This is called the Pauli exclusion principle, which is rooted in quantum mechanics. Again, we do not need to explore the reasons for this in detail here, but we can take it as a fact to progress the discussion. As we move through higher atomic number elements in the Periodic Table, the electrons are stacked into additional orbitals, two by two.
The orbitals themselves are collected together into subshells that are given letter designations (s, p, d, f, g). Technically, these are electrons that share the same “angular momentum quantum number,” or the same orbital shape. These subshells themselves are put together to form shells with number designations: 1, 2, 3, etc. Technically, shells are electrons that share the same “principal quantum number.” In a crude way, you can think of shells as the “layers” of electrons as you move out from the inner layers of electrons (lower shell numbers) to the outer layers (higher shell numbers). Figure 3.3 shows this somewhat confusing nomenclature more clearly. If you want to understand these quantum constraints better, the further reading section provides suggestions. Here, the purpose is to draw out some key points.
Читать дальше














