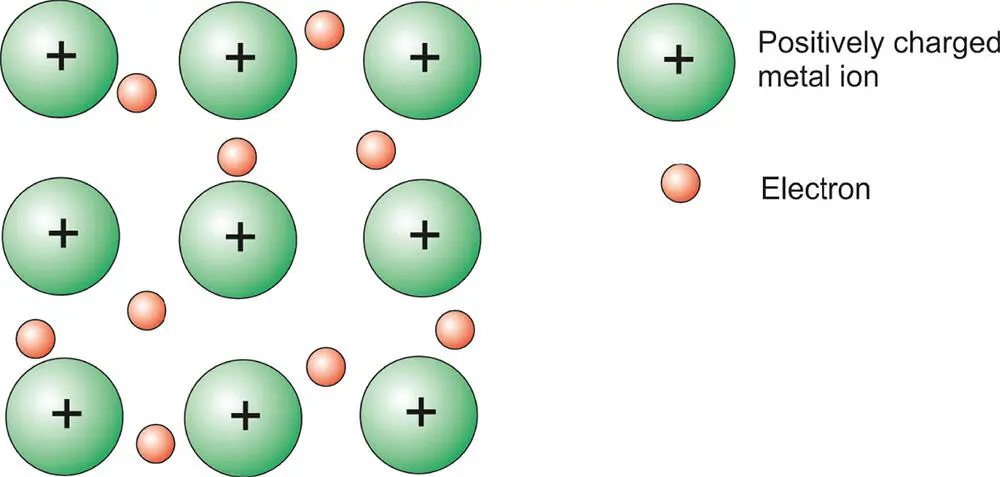Source: Reproduced with permission of Public-domain-photos.com; Jon Sullivan.
Metallic bonding is the type of bonding found in metals such as sodium, copper, magnesium, and iron. Consider sodium. We have already seen how it can form ionic bonds, such as in NaCl. We saw that it has one spare electron which it can transfer to an atom such as chlorine to become a sodium ion and achieve a stable noble gas configuration. Another way it could lose this electron is for the electron to dissociate from the atom and form a “sea” of electrons around other sodium ions that are behaving in an identical way. The positively charged sodium ions do not fly apart from electrostatic repulsion because of this intervening sea of negatively charged electrons. This is depicted in Figure 3.10. In this way, the atoms, by delocalizing the electrons (which is essentially like losing them), achieve a stable noble gas configuration.
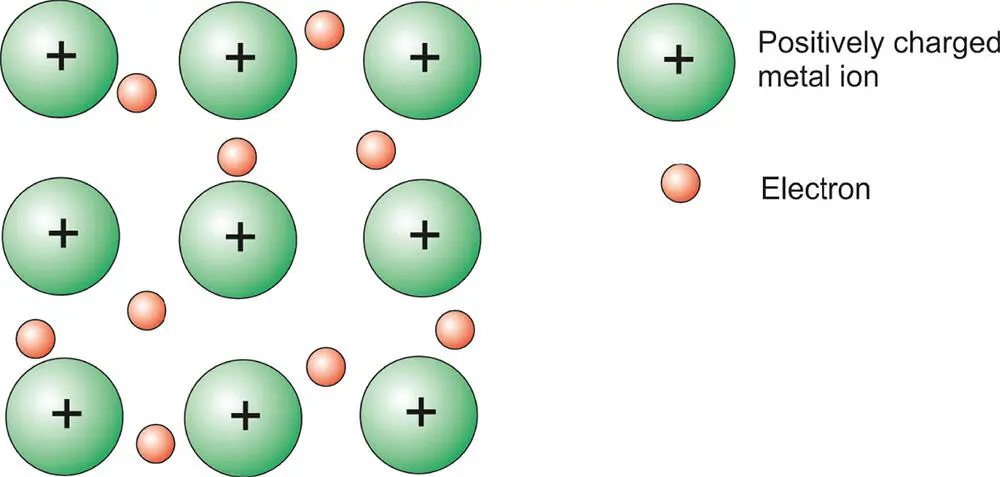
Figure 3.10 Metallic bonding showing a “sea” of delocalized electrons around positively charged metal ions.
Metals form strong bonds. Again, using the reasoning discussed earlier, consider potassium. Its energy per bond is ∼5 × 10 −19J, which is equivalent to approximately 125 times the thermal energy in the bond at room temperature. Many metals are therefore stable at room temperature (e.g. iron and silver).
Interestingly, metallic bonding is not relevant for life. Life certainly does make use of metal ions in many diverse ways that will crop up throughout this book. Metal ions are particularly prominent in enzymes, in which they play a role in mediating the catalysis of chemical reactions as cofactors. These are found in many situations in cells where electron transfer is needed, such as iron–sulfur clusters used in energy-gathering processes. Examples of these ions include copper, iron, and magnesium. However, in all these examples, the metal ions are generally present as individual ions, not as bulk metallic deposits. So where are all the metal structures in life? You might like to read the Discussion Point.
Discussion Point: Why Doesn't Terrestrial Biochemistry Use Metal Structures?
Although terrestrial biochemistry makes extensive use of metal ions, such as iron and copper as cofactors in enzymes, life does not fabricate metal structures. This is surprising given the vast use of metals and metallic structures in human engineering. Why aren't bones made from steel for example, or horns in animals made from titanium? There are a number of possible reasons that may not be mutually exclusive: (i) It may be energetically too expensive. Most metals are not pure in the environment and considerable energy is used in human manufacturing processes to purify them. Locked up in sulfides, carbonates, and other compounds, they are difficult to purify biologically. (ii) Pure metals have large densities. For example, compare the density of steels (typically ∼8000 kg m −3) to calcium phosphate bone (typically ∼3800 kg m −3) and one can see that metal structures would come with a considerable mass burden. (iii) Metals are generally at low abundance in the environment. It should be pointed out that this argument holds for some metals such as vanadium, but it clearly does not for metals such as iron and aluminum, which constitute 8.1% and 5.0% of Earth's crust, respectively. The preceding three arguments assume that nature could have attempted to use metals but for various reasons rejected them. However, we have no evidence of extensive experiments in metal construction in biology. It could be that pathways to other high-strength compounds are evolutionarily simpler than pathways that involve the extraction and purification of metals from compounds naturally found in the environment. What do you think? Would alien life be any different?
3.9 Van der Waals Interactions
We have shown how atoms and molecules can be held together by ionic and covalent bonding. There are two other types of bonding that play a crucial role in holding matter together and take prominent roles in life. These two bonding types are generally involved in bonding between molecules. These bonds are generally much weaker than ionic or covalent bonds. They are van der Waals interactions and hydrogen bonding. We first examine van der Waals interactions.
Sometimes also called van der Waals forces, these van der Waals interactionsare in the order of a hundred times less strong than covalent bonds. Nevertheless, they are essential in biological systems.
There are three categories of van der Waals forces.
3.9.1 Dipole–Dipole (Keesom) Forces
Molecules have a charge distribution that is never quite even (i.e. it has a directionality or anisotropy), which results from the non-even distribution of the electrons and their negative charge. This uneven electron charge distribution results in a small permanent electric dipoleacross the length of the molecule.
In Figure 3.11, you can see this schematically illustrated using hydrogen chloride. Chlorine has a higher electronegativity than hydrogen. In other words, it tends to pull electrons toward it. As it tugs hydrogen's electron toward it, it gets a very slight negative charge. This also means that the hydrogen atom develops a slight positive charge (seen from the point of view of the hydrogen atom, the positive charge of its proton is not quite cancelled out by the negative charge of its electron, which is now more associated with the chlorine atom). The result is that the hydrogen atom has a very slight net positive charge. Hence, the molecule now has a polarity or dipole. Now, if two of these molecules meet up, the slightly negatively charged chlorine atom will be attracted to the positively charged hydrogen on the other molecule; they attract one another like small bar magnets (Figure 3.11).
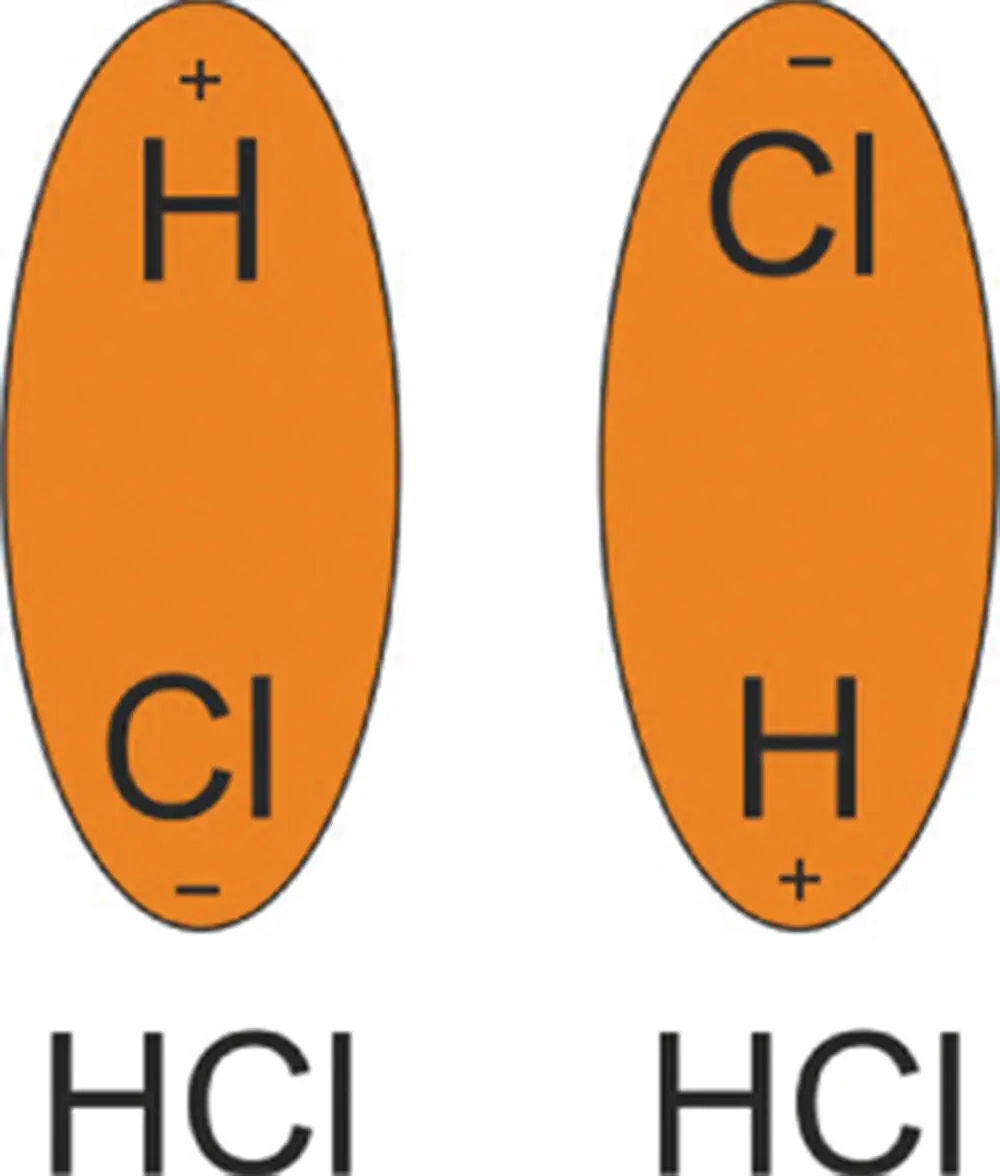
Figure 3.11 The dipoles of two HCl molecules involved in Keesom interactions. They attract each other like tiny bar magnets.
3.9.2 Dipole-Induced Dipole (Debye; Pronounced Deh-beye) Forces
For the second category, van der Waals interactions can be induced in a molecule that has no charge. In this type of interaction, one of the molecules has a charge (HCl), but the other does not, for example as shown in Figure 3.12 with neon. Nevertheless, a charge can be induced in neon by the presence of the charge in HCl, which influences the electron distribution in the neighboring atom or molecule. As a result, the molecules are attracted.
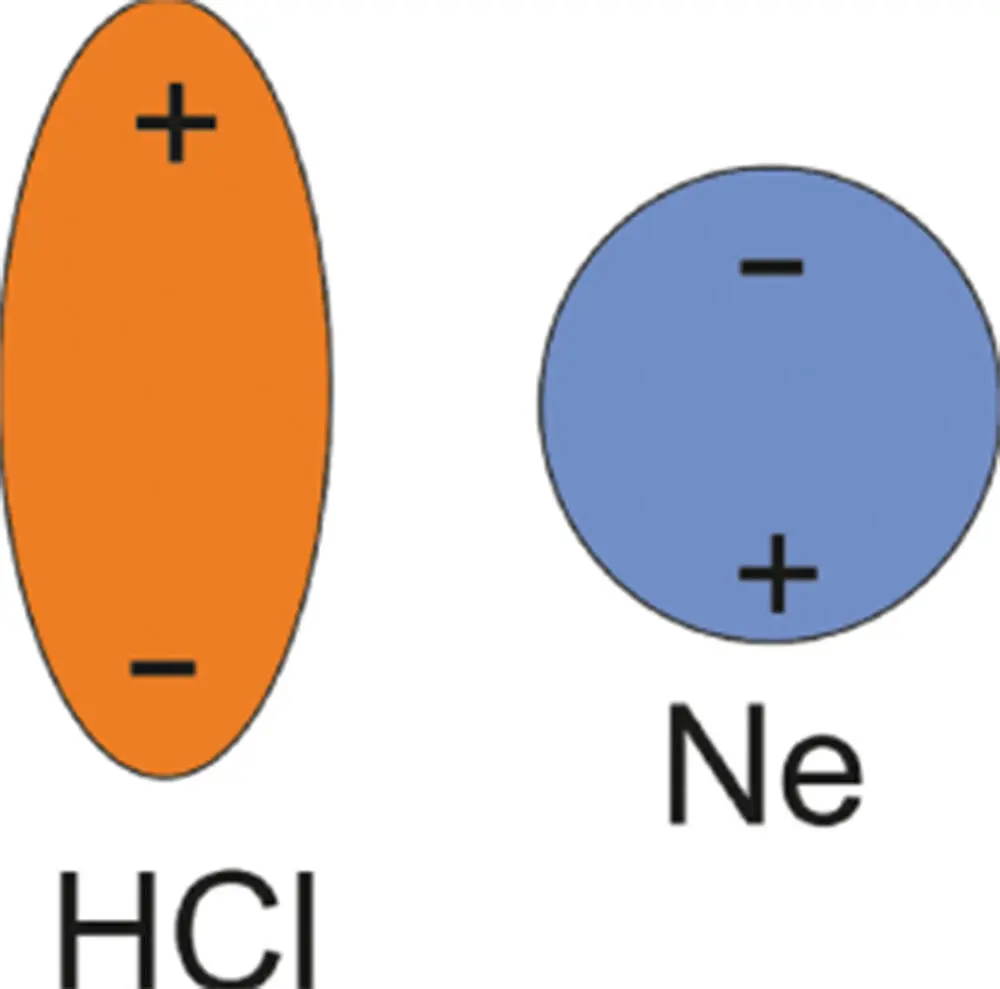
Figure 3.12 An induced dipole in the otherwise uncharged neon is an example of a Debye interaction.
Yet another type of van der Waals interaction is one in which neutral atoms can attract one another. The electron distribution, even in neutral atoms and molecules, is never quite evenly spaced out. A charge anisotropy will exist on account of the electrons in orbit around the atom, which, at a snapshot in time, will always be slightly unevenly distributed around the atom (shown greatly exaggerated in Figure 3.13).
Читать дальше