Ionic bonds are typically very strong. We can consider the stability of these bonds from an energetic (thermodynamic) point of view. Let's consider sodium fluoride, NaF. The energy required to break an ionic chemical bond in this structure is about 3 × 10 −19J. We can also calculate the typical thermal energy in a bond at a specified temperature. This is the thermal energy that would be in the bond when it is in equilibrium with a given environment at a particular temperature. This can be approximated by ∼k B T . k Bis the Boltzmann constant, which has a value of 1.381 × 10 −23JK −1, and T is the temperature, measured in Kelvin. At room temperature (about 300 K), the value of k B T is 4.1 × 10 −21J. The ionic bond energy is therefore about 75 times the thermal energy in the bond. In other words, the thermal energy in NaF at room temperature will not cause the ions to break apart. You need to put in a lot more energy to break apart the ionic bonds, explaining why this compound is stable at room temperature. This is the case for many ionic bonds.
3.6.1 Ionic Bonds and Life
Ionic bonds are important for life because they can also play a role in holding different parts of molecules together.
In Figure 3.5, you can see a typical protein chain. A protein is made up of individual amino acidsstrung together in a long chain. Some of these amino acids are charged (we will come back to amino acids and discuss their features in more detail in Chapter 4).
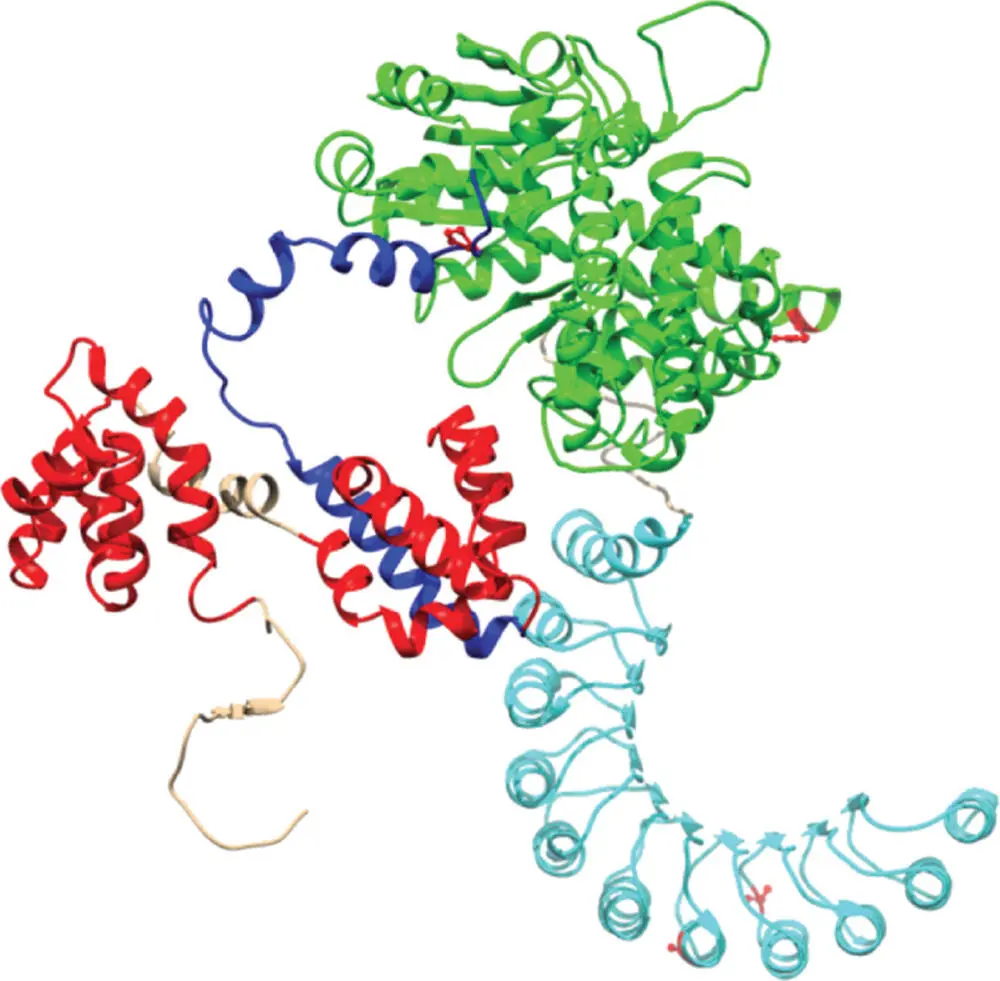
Figure 3.5 A typical protein. The colored ribbons and lines depict the chains made of amino acids strung together, a chain that is itself folded together into a complex three-dimensional molecule. This one (called NOD2) is involved in the human immune system.
Source: Reproduced with permission of LPKozlowski, https://commons.wikimedia.org/wiki/File:Nod2_protein.png.
Some amino acids have a negatively charged side group, and some a positively charged side group. These two different types of amino acids can therefore come together to form an ionic bond. For example, the negative charge in an aspartic acid, which is one type of amino acid, is attracted to the positive charge in a lysine, another type of amino acid, to form an ionic bond, which keeps the chains together. Figure 3.6 illustrates this bonding.
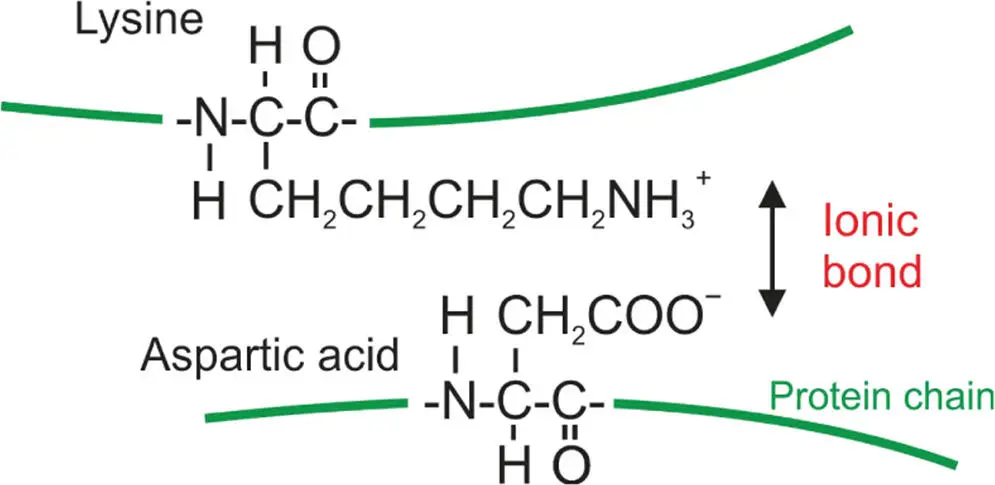
Figure 3.6 An ionic bond in a protein formed between two amino acids, the positively charged amino acid lysine, and the negatively charged amino acid aspartic acid. Hydrogen bonding is also involved giving rise to both ionic and hydrogen bonding across the link – usually called a “salt bridge.”
This bonding is important in proteins that have functions to perform in cells, for example proteins that are enzymesor biological catalysts, that play a role in accelerating chemical reactions in life. These ionic bonds help the proteins keep their three-dimensional shape, which is necessary if they are to perform their functions correctly. From a very colloquial perspective, you can think of ionic bonds as “bolts” that can help hold together the three-dimensional structure of biological molecules, such as proteins. It is important to note that in proteins, as well as this ionic interaction, hydrogen bonding (which is discussed later in this chapter) also plays a role in linking the two amino acids together. It is this combination of ionic bonds and hydrogen bonds that forms the complete link. This combination of ionic and hydrogen bond interaction is referred to by biochemists as a “salt bridge.”
In ionic bonding, the central feature of the bond is the transfer of an electron from one atom to another to generate two ions. In covalent bonds, an electron is shared . Covalent bonds take place between atoms that are generally close to each other in the Periodic Table and have a small difference in electronegativity(the tendency of an atom to attract electrons). Similarly to ionic bonds, the sharing of an electron allows the noble gas electron configuration to be attained by both atoms.
The simplest example of a covalent bond is found in the hydrogen molecule, shown in Figure 3.7. To return to the electron configurations that we discussed earlier, a hydrogen atom has an electron configuration of 1s 1: one electron in the first shell, the s subshell. It would like to have two in this subshell so that the subshell is full. In the hydrogen molecule, the sharing of each electron between two hydrogen atoms allows for two electrons in this subshell.
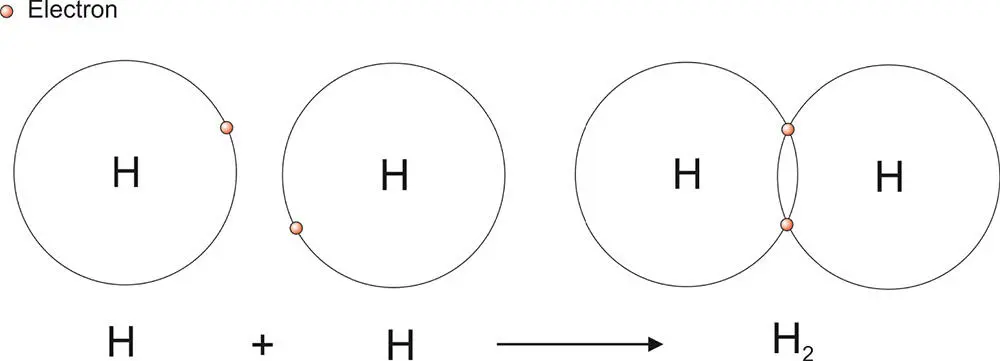
Figure 3.7 The covalent bond in the hydrogen molecule. The two electrons are shared between both hydrogen atoms.
Covalent bonds are very strong. An example is diamond, a covalent network of carbon. Taking the same logic that we used for ionic bonds, the energy per bond is ∼6 × 10 −19J. That's equivalent to 150 times the thermal energy at room temperature. In other words, the thermal energy in the bonds of diamond is much lower than the energy needed to break carbon–carbon bonds. Diamond is very stable at room temperature.
3.7.1 Covalent Bonds and Life
Covalent bonds play a central role in biology because they are the bonds that hold atoms together in the vast array of molecules from which life is constructed.
Covalent bonds are also found in specific situations where biologically important molecular structure is required. In analogy to the role of ionic bonds in holding charged amino acids together in protein chains, covalent bonds can form between sulfur-containing amino acids (cysteine and methionine). The sulfur atoms within the amino acids join together and form a disulfide bridge. These bonds anchor the structure of the protein, making sure that its three-dimensional shape is maintained. Figure 3.8 shows a schematic example of disulfide bridges made of covalent bonds holding a protein chain together in a loop.
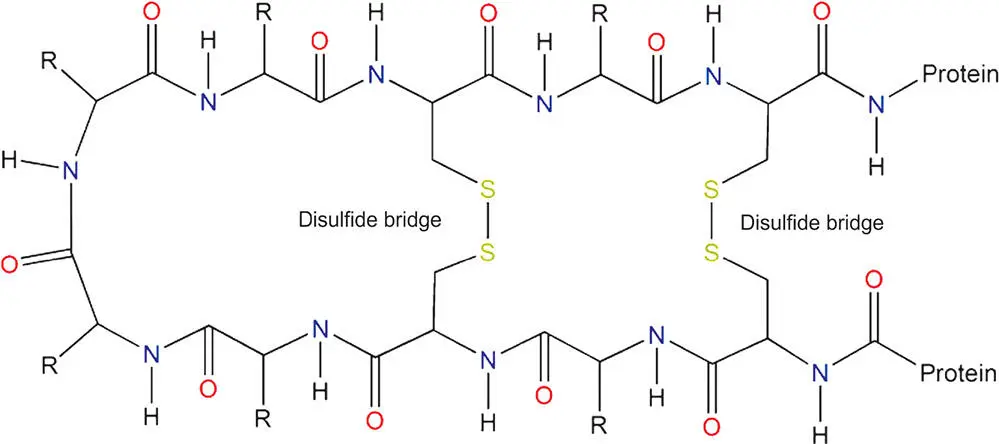
Figure 3.8 As well as holding atoms together in molecules, covalent bonds link within molecules to provide structure. Here covalent bonds within two disulfide bridges hold a protein chain made of amino acids into a loop. The covalent bonds in this structure, as in many similar diagrams, are shown as solid black lines.
As disulfide bridges can add rigidity to a protein, life can use them to enhance the stability of proteins. For example, some microbes, such as those that live in hot springs in Yellowstone National Park (Figure 3.9), must cope with high temperatures, often well over 60 °C. Some proteins have been found to contain extra covalent bonds to enhance their thermostability. We return to examine adaptations to extreme environments later in the book. For now, the point to learn is that changes in bonding in molecules can be used by life as one of a repertoire of adaptations to extreme physical conditions.

Figure 3.9 An enhanced number of covalent bonds in proteins is used in some microbes to stabilize proteins against high temperatures. Enhanced stability of biological molecules is needed in such environments, for example in these volcanic pools in Yellowstone National Park, USA. In this image, the microbes live within the browns and yellows in the spring, colors caused by microbial pigments and minerals such as iron.
Читать дальше

















