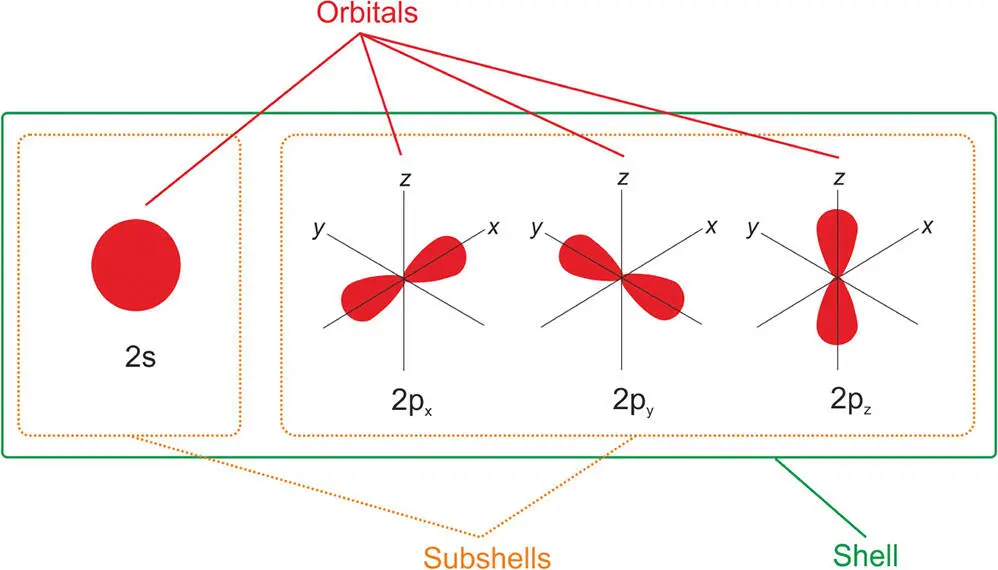
Figure 3.3 A diagram showing the second shell in an atom and the nomenclature used to describe the various electron locations.
From the point of view of atomic structure, and the consequences for molecules involved in life, the crucial point to understand is that atoms have a tendency to react, losing or gaining electrons, until they have full electron shells. Once electron shells are full, then there are no “extra” electrons to get involved in chemical reactions or “missing” spaces for electrons that tend to be filled in chemical reactions. This explains the inert and stable characteristics of the noble gases, such as argon and neon, which have full electron shells. In a simplified sense, one can say that atoms tend to react until they achieve noble gas configurations of electrons. An understanding of this basic idea goes a long way to explaining how atoms behave and how molecules are formed.
Let's look at an example to illustrate this idea and how it applies to bonding. Consider the sodium atom (Na). It is in group 1 of the Periodic Table (see Appendix). It has 11 electrons (and therefore 11 protons – it has an atomic number of 11). Its electron structure is written as 1s 22s 22p 63s 1. The first number in this sequence is the shell number (in this atom there are three shells: 1, 2, and 3). Each letter (s and p) refers to a different subshell. The superscript shows the number of electrons in each subshell. Starting at the beginning, you can see that it has two electrons in shell 1, subshell s (1s 2). This shell is full. Moving outwards in the layers of electrons, we then see that in the second shell, subshell s, it has two electrons (2s 2). This is also full. In the second shell, we also have a p subshell. You will see that this has six electrons in it (2p 6). The 2p subshell is made of three separate orbitals called x , y , and z of the same shape, each with a pair of electrons in them (they are full), giving the 2p subshell six electrons in total, hence 2p 6. Finally, we come to the outermost shell, number 3, which has one lone electron in its s subshell (3s 1). This electron shell is not full. By losing this lone electron, the sodium atom becomes more stable because the next shell down is full. In other words, in an unscientific turn of phrase, sodium wants to lose this dangling spare electron to achieve a noble gas, stable configuration.
However, there are other consequences of losing this 3s 1electron. In losing it, the sodium atom gains a net positive charge, as it now has 11 protons, but only 10 electrons, making a net positive charge of 1. The product of this electron loss, written as Na +, is called an ion. An ion is an atom that has gained or lost electrons.
To briefly illustrate this concept again on the other side of the Periodic Table, consider the element chlorine. Chlorine, in group 17 of the Periodic Table, has the electronic structure 1s 22s 22p 63s 23p 5. You can see that in its last electron subshell, 3p 5, it has five electrons. It would like to gain one to arrive at six electrons in the 3p subshell and therefore fill the shell. By gaining an electron, it would attain a noble gas electron configuration, making it more stable. If it does this, however, it will now have 17 protons and 18 electrons, resulting in a net negative charge of 1. It will have become the ionCl −.
The tendency of atoms to lose or gain electrons in this way to attain a noble gas electron configuration explains the key features of many bonds that we look at in the next section.
You will also notice that atoms have a tendency to lose or gain electrons in their outer electron shells since the ones below are full. The characteristics of the outer electron shells define the chemical behavior of different atoms. This explains why elements in the same group of the Periodic Table, for example carbon and silicon in group 14, which have the same electron configurations in their outer shells, tend to share similar chemical characteristics. This fact becomes important when we consider elements used in life in Chapter 4.
At this point, it is worth revisiting the question we brought up in Chapter 1that puts an astrobiological perspective on this discussion. How much of what we have just discussed can be said to be a universal characteristic of life? I think you'd agree that we can confidently say that everything we have discussed is universal. If there is an alien intelligence somewhere else in the Universe, they would be drawing diagrams like Figure 3.3. We can say this because we know that the Periodic Table is universal since the Pauli exclusion principle that determines electron structure is universal. This is not conjecture. Using spectroscopy, which is discussed at the end of this chapter, we know that distant galaxies and stars, and therefore the planets they host, are made of the same elements as we have discovered in the Periodic Table. The fundamental chemical structure of atoms that make up all life in the Universe, if it exists elsewhere, is the same as on this planet.
3.5 Types of Bonding in Matter
With this knowledge of atoms and ions, we now consider how they bond together to construct molecules and ultimately large, complex molecules or macromolecules, such as the genetic material DNA, that make up life.
There are five basic types of bonding that hold atoms and molecules together to give rise to the structure of ordinary matter, including life. We consider each of these in this chapter:
ionic bonding,
covalent bonding,
metallic bonding,
van der Waals interactions, and
hydrogen bonding.
The first three of these types of bonding are primarily involved in holding atoms together to make molecules, although ionic and covalent bonds also play prominent roles in holding parts of whole molecules together to generate three-dimensional structures. The last two types of bonding, van der Waals interactions and hydrogen bonding, are primarily involved in mediating interactions between individual molecules. Let's have a look at some of the features of these bonds and, in particular, how they are used in life.
Ionic bondingis the electrostatic force of attraction between positively (+ve) and negatively (−ve) charged ions (primarily between non-metals such as chloride or fluoride ions and metals such as sodium or potassium ions). Most ionic compounds are crystalline solids at room temperature.
The crucial feature of an ionic bond is that each atom either gains or loses an electron so that the resulting ion has its lowest energy (noble gas-like) configuration. Table salt, NaCl, is a typical example of ionic bonding, and you can see its structure in Figure 3.4. In this salt, sodium gives up an electron, and chlorine gains this electron so that both ions gain a noble gas configuration, as we saw in Section3.4. In other words, the Na atom has transferred its electron to the Cl atom, and the result is two ions, Na +and Cl −.
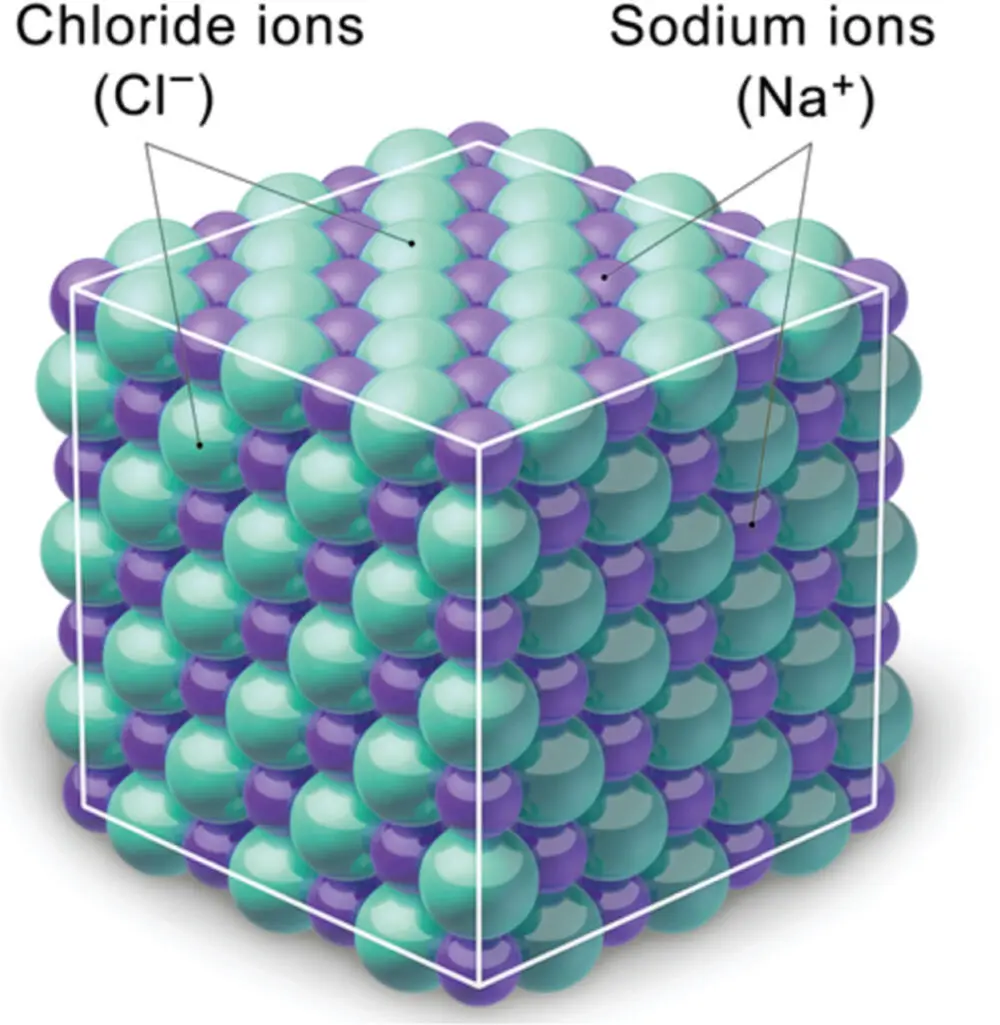
Figure 3.4 The structure of NaCl showing the alternating sodium and chloride ions.
Source: Reproduced with permission of B. Blaus, https://commons.wikimedia.org/wiki/Category:Crystal_structure_of_sodium_chloride#/media/File:Blausen_0660_NaCl.png.
After transferring an electron, we now have two ions, Na +and Cl −, with opposite charges. They are attracted to one another. However, if we have many of these ions, then things get more complicated. Clearly, negatively charged chloride ions will be repelled from each other, and positively charged sodium ions will be repelled from each other. If we place many Na +and Cl −ions together, the natural configuration they take up to maximize attraction and minimize repulsion is an alternating packed cubic structure (Figure 3.4). Other similar examples are cesium chloride (CsCl) and sodium fluoride (NaF).
Читать дальше














