1.3.1 Oxidation of Amines
The direct oxidation of primary amines into the corresponding nitro derivatives is very useful for fundamental and industrial applications because it provides targets that may be difficult to obtain via nitration methods.
The pioneering oxidation of primary amines has been performed with various reagents such as ozone [11], NaMnO 4[12], KMnO 4[13], peracetic acid [14], meta -chloroperbenzoic acid (MCPBA) [15], and dimethyldioxirane (DMO) [16].
The reactions proceed ( Scheme 1.9) via a few intermediates and an efficient oxidant is preferred for a rapid conversion to nitroalkanes. In fact, incomplete oxidation was sometimes observed, with consequent formation of by-products such as nitroso and nitroso-dimers.

Scheme 1.9 Oxidation of primary amines.
From an environmental point of view, ozonization is also a benign and atom-efficient oxidation process, but an important drawback deals with the safety since it can lead the formation of unstable and potentially explosive ozonides and peroxides. Moreover, the subsequent quenching step is generally exothermic, thus requiring an efficient temperature control. To overcome the aforementioned disadvantages, few years ago Kappe and coworkers [17] proposed a continuous flow procedure as a valuable alternative to batch protocols for the oxidative conversion of important functionalities, including some primary amines to the corresponding nitroalkanes.
Later, the oxidation of amines has been developed using an alternative and improved oxygen transfer reagent, such as “HOF·CH 3CN complex,” simply obtained passing fluorine through acetonitrile. The method ( Scheme 1.10) [18] proceeds by dissolving the amine in CH 2Cl 2, then the mixture was cooled to −15 °C and added to the glass reactor containing NaF and HOF .CH 3CN (3 equiv) in cold (−15°C) aqueous CH 3CN.
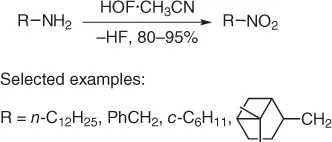
Scheme 1.10 Oxidation of amines using HOF·CH 3CN.
The reaction proceeds for few minutes allowing nitroalkanes in excellent yields. Considering that HOF·CH 3CN cannot be used in a large-scale process due to its instability (half-life = four hours at room temperature), it cannot be packaged and shipped but must be prepared and used immediately. In addition, the oxidation reaction has been performed in batch and all the problems of using concentrated oxidizing media remain. In this context, Sandford and coworkers [19] developed a flow process to solve the above drawbacks. However, for an efficient and selective oxidation, any fluorine gas that enters into the flow channel must be completely consumed before the organic substrate could be added to prevent fluorination and substrate degradation. In addition, the oxidation needs 3 equiv of HOF·CH 3CN to perform the efficient conversion of the ammines to nitroalkanes. Thus, a flow reactor designed specifically for gas–liquid reactions was adapted in order to perform the sequential formation and reaction of HOF·CH 3CN.
Some years before, Krohn and Küpke reported [20] an efficient procedure to convert aliphatic amines into nitro compounds based on a zirconium-catalyzed oxidation of primary amines with tert -butyl hydroperoxide (TBHP), in the presence of Zr(O- t -Bu) 4as catalyst ( Scheme 1.11).
In fact, a solution of amine (10 mmol), in dry CH 2Cl 2, is treated with freshly activated powered molecular sieves (3 Å) and Zr(O- t -Bu) 4(1 mmol). After stirring for 30 minutes, a solution of TBHP (59 mmol) in CH 2Cl 2is added. After complete consumption of the starting material and work-up, good yields of nitroalkanes are obtained. The method works well with a variety of substrates and it is mild enough to preserve other functional groups as acetals and esters.
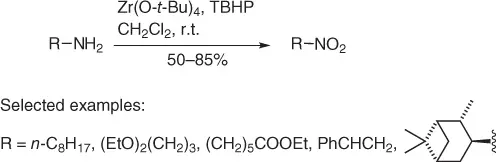
Scheme 1.11 Oxidation of amines using Zr(O- t -Bu) 4.
1.3.2 Oxidation of Oximes
Conversion of carbonyl function to nitro group (retro-Nef reaction) [21] is an important method for the preparation of nitro compounds. Such conversion is generally effected via oximes using strong oxidants ( Scheme 1.12).
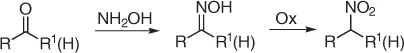
Scheme 1.12 Retro-Nef reaction via oximes.
Following a pioneering procedure performed with CF 3CO 3H [22], there was reported a revised and improved procedure ( Scheme 1.13) [23] using peroxytrifluoroacetic acid obtained through reaction of urea–hydrogen peroxide complex (UHP) with trifluoroacetic anhydride (TFAA) in acetonitrile at (0 °C).
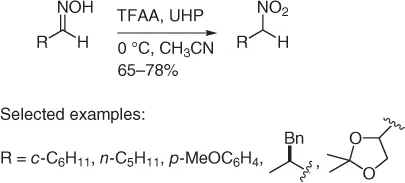
Scheme 1.13 Oxidation of oximes with TFAA/UHP.
The procedure allows good yields with aldoximes, while fails to react with ketoximes. On the other hand, ketoximes can be converted into secondary nitroalkanes, following the Olah method [24], oxidizing ketoximes with sodium perborate in glacial acetic acid. However, this procedure failed with aldoximes.
An interesting conversion of both aldoximes and ketoximes to the corresponding nitroalkanes has been realized by a complementary synthetic route of the UHP method. In fact, the oxidation achieved with “Benz-Mo,” the Mo(VI) oxidiperoxo complex [Benz-MoO(O 2) 2] −( t -Bu) 4N +in acetonitrile, affords good yields of both primary and secondary nitroalkanes ( Scheme 1.14) [25].
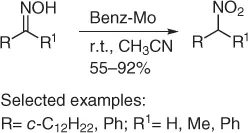
Scheme 1.14 Oxidation of oximes using Benz-Mo.
1.3.3 Oxidation of Azides
Recently azides have been identified as a new precursor of primary nitroalkanes by oxidation. In this context, Rozen et al. [26] found that the carbon-bonded nitrogen of azides would be nucleophilic enough to interact with the oxygen atom of HOF·CH 3CN. Thus, thanks to the efficiency of this oxidant, a variety of azides, easily obtainable from alkyl bromides and sodium azide, have been converted into primary nitroalkanes in quantitative or very good yields and in short reaction times ( Scheme 1.15).
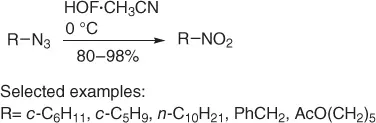
Scheme 1.15 Oxidation of azides using HOF·CH 3CN.
Concerning the use of fluorine, a very corrosive material, it should be used only with the appropriate vacuum line techniques. However, for the occasional user, various premixed mixtures of F 2in inert gases are commercially available.
Moreover, as for the oxidation of primary amines, a flow process has been developed [19] and, following this procedure, quantitative yields (<90%) of primary and secondary nitroalkanes were obtained.
1.4 Reduction of Conjugate Nitroalkenes
Читать дальше



















