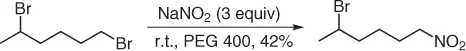An interesting application of the nitration of alkyl halides has been reported [4] for the preparation of fatty nitroalkanes, mainly the polyunsaturated ones, from naturally alcohols ( Scheme 1.2) through their successive conversion into (i) bromo alkanes, (ii) iodo alkanes (more reactive vs bromo derivatives), and (iii) nitroalkanes with silver nitrite, in Et 2O and at room temperature for three days.
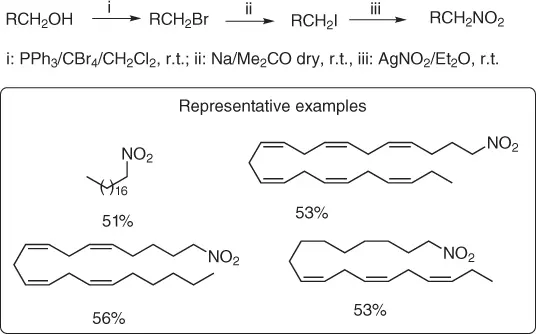
Scheme 1.2 Synthesis of fatty nitroalkanes.
It is important to point out that the need to convert bromo alkanes into the iodo alkanes is due to the fact that, contrary to short-chain bromo alkanes, the longer ones are inert toward the reaction with AgNO 2.
Benzylic nitro compounds (aryl nitromethanes) are an important class of nitroalkanes and their preparation shows some drawbacks due to the lack of selectivity, with consequent formation of an amount of nitrite ester. Guillaumet and coworkers [5] reported an improved approach (based on the Victor Meyer reaction) for the synthesis of aryl nitromethanes, minimizing the complex purification process on the basis of the difference in p K avalues between aryl nitromethane and benzyl nitrite ester (side product). In fact, the nitro compound is quite a strong acid (p K a= 8–12) due to the high stability of the formed nitronate. Indeed, the negative charge is delocalized onto the nitro and the aryl group, while the nitrite ester does not bear any acidic proton because the conjugate base is no longer stabilized in any nitro group. This significant acidic difference has been the ideal property to develop a practical purification method usable on large scales.
In fact ( Scheme 1.3), adding a methanolic solution of sodium methoxide into the filtrate of the reaction, it is possible to observe a precipitate of the formed sodium nitronate that could be simply isolated by filtration. The nitronate, treated with acetic acid and urea, converts into the aci -nitro structure through an O-protonation. The latter quickly precipitates in the acidic medium and isomerizes into the target aryl nitromethane in satisfactory yields (>50%) and with the survival of a variety of other functional groups.
However, the nitration of alkyl halides with AgNO 2(Victor Meyer procedure) shows important drawbacks because usually the need of long reaction times and the use of toxic solvents produce important amounts of by-products and require a tedious work-up. Thus, an improved procedure has been developed, in aqueous medium, treating primary alkyl halides with 4 equiv of silver nitrite (r.t. or 60 °C) [6]. The method allows satisfactory to good yields of a variety of primary nitroalkanes, in short reaction times (0.5–1.25 hours, Scheme 1.4). The need of 4 equiv of silver nitrite is due to the fast of the reaction that can minimize the competitive formation of the corresponding alcohol. In addition, the formation of alkyl nitrite is strongly depressed.
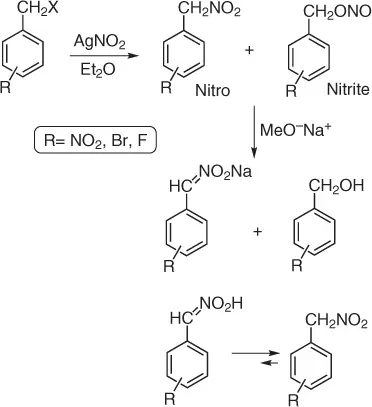
Scheme 1.3 Aryl nitromethanes synthesis under minimized purification step.
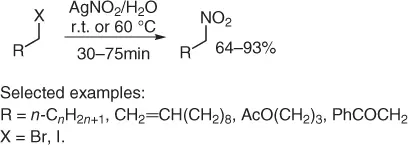
Scheme 1.4 Nitration of halides with silver nitrite.
Although the method works well with both alkyl bromides and alkyl iodides, the latter often shows higher reactivity, while it fails with secondary halo derivatives. Furthermore, due to the mildness of the reaction conditions, other functionalities such as C=C double bond, ester, imide, and ketones are preserved.
An important application of the above method is the synthesis of α,ω-dinitroalkanes from α,ω-diiodo structures ( Scheme 1.5).
Thus, this procedure can be considered as a significant progress in the sustainability of the nitration of halo alkanes. However, a possible drawback consists of the need for a large excess of expensive reagents such as silver nitrite and the formation of the corresponding alcohol due to the competitive action of water vs nitrite.
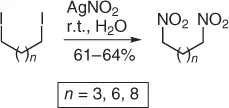
Scheme 1.5 Nitration of halides with silver nitrite (selected examples).
Then, few years later, our group developed an improved eco-friendly nitration of alkyl halides [7], using sodium nitrite in polyethylene glycol (PEG 400) as green reaction medium [8,9]. Thus, treatment of primary alkyl halides with 1.5–3 equiv of sodium nitrite, at room temperature, in PEG 400 as solvent, allows the chemoselective (other functionalities such as esters, hydroxyl group, C=C double bond, ethers, aromatics, and amides are preserved) formation of primary nitroalkanes ( Scheme 1.6). It is important to point out that only traces of alkyl nitrite are observed. In addition, it is relevant that PEG 400, after extraction with cyclohexane, can be recycled at least twice for the same reaction before its disposal as waste.
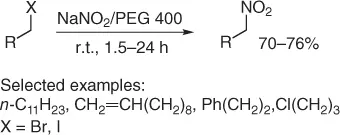
Scheme 1.6 Nitration of halides under PEG solvent.
Although the procedure works well with both alkyl iodides and bromides, the latter needs larger amounts of sodium nitrite (3 equiv vs 1.5 equiv) and longer reaction times ( ratio > 3).
With this procedure, the conversion of secondary halo derivatives fails, and this result can be applied to the chemoselective nitration of primary iodides or bromides in the presence of a secondary one ( Scheme 1.7).
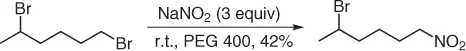
Scheme 1.7 Chemoselective nitration of alkyl halides.
1.2 Nitration of Mesylates and Tosylates
The displacement of alkyl halides with metal nitrite, thanks to the large number of available halides, is the most used procedure for the preparation of nitroalkanes. However, despite the advantages introduced with these methodologies, the search for alternative sources is surely welcomed. In this regard, alcohols are a broad class of molecules easily available from both commercial and nature sources. The conversion of alcohol into nitroalkanes firstly requires the conversion into the corresponding mesylates or tosylates (usually in quantitative yields) that can be easily converted ( Scheme 1.8) into the corresponding nitroalkanes using tetrabutylammonium nitrite (TBAN) in toluene for few hours (2.5 hours for primary compounds and 28–40 hours for the secondary ones) [10].
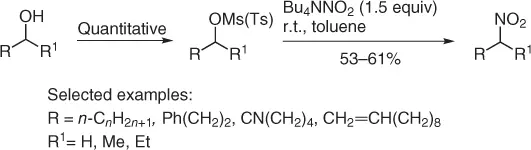
Scheme 1.8 Nitration of mesylates and tosylates.
The method was realized at room temperature and, after a simple work up, a variety of primary and secondary nitroalkanes can be easily obtained is 41–61% overall yields.
1.3 Oxidation of Nitrogen Derivatives
The oxidation of nitrogen derivatives is a useful alternative to furnish nitroalkanes that are difficult to prepare by the direct nitration of hindered halo derivatives.
Читать дальше