1 ...7 8 9 11 12 13 ...31 The Yu group also demonstrated that rhodium and ruthenium complexes can also catalyze these types of reactions via similar mechanisms ( Scheme 1.25) [36].
Apart from using a directing group in the C–H functionalization process, Catellani‐type reactions can also achieve directed C–H activation without the use of amide‐type directing groups. Dong and coworkers applied this strategy in the development of Pd‐catalyzed electrophilic amination reactions ( Scheme 1.26) [37]. The main difference between this transformation and the standard Buchwald–Hartwig protocol is that the C–H group adjacent to the aryl halide moiety (i.e. the ortho position) gets aminated, while in the Buchwald–Hartwig reactions, the C—X bond itself gets aminated (i.e. the ipso position).
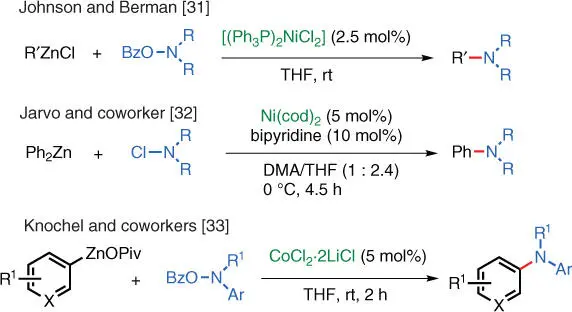
Scheme 1.22 Electrophilic amination catalyzed by other transition metals.
Source: Johnson and Berman [31], Barker and Jarvo [32], and Lutter et al. [33].
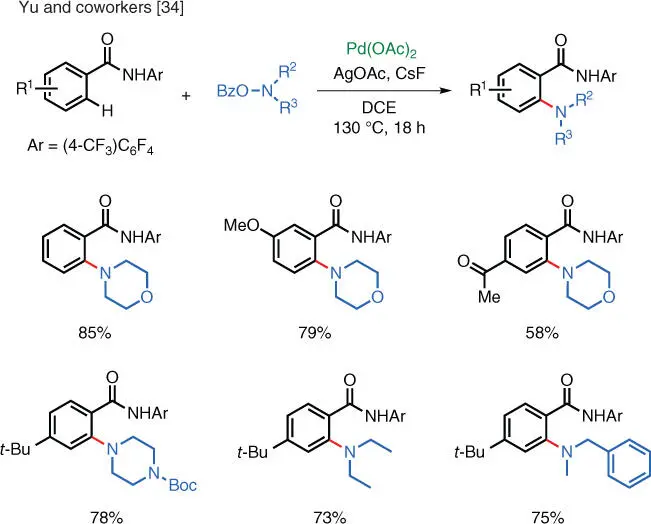
Scheme 1.23 Pd‐catalyzed aromatic C–H amination via electrophilic amination.
Source: Modified from Yoo et al. [34].
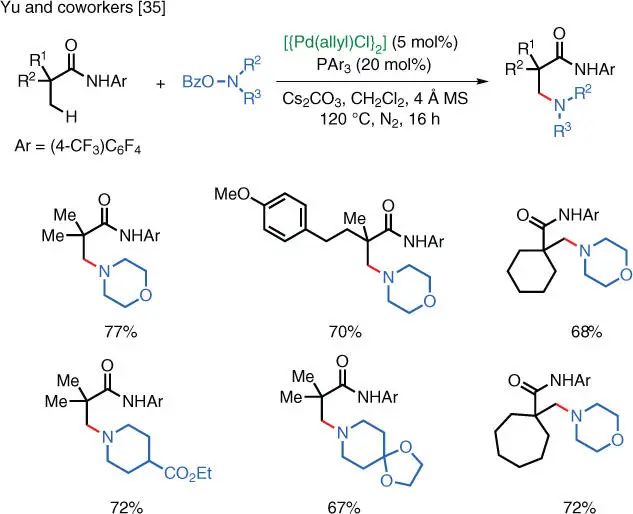
Scheme 1.24 Pd‐catalyzed aliphatic C–H amination via electrophilic amination.
Source: Modified from He et al. [35].
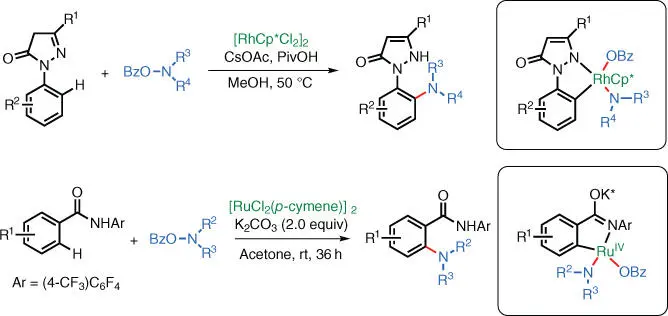
Scheme 1.25 Ru‐catalyzed C–H amination.
Source: Modified from Shang et al. [36].
A unique Fe‐catalyzed reaction was reported by the Yang group in which styrenes undergo formal hydroamination to afford tertiary amines with Markovnikov regioselectivity [38]. In this case, an organoferrates initially formed from a Grignard reagent and the iron(II) catalyst. This organoferrate intermediate engages the styrene substrate to form a hydride complex, which hydrometallates the styrene double bond and subsequently undergoes electrophilic amination ( Scheme 1.27).
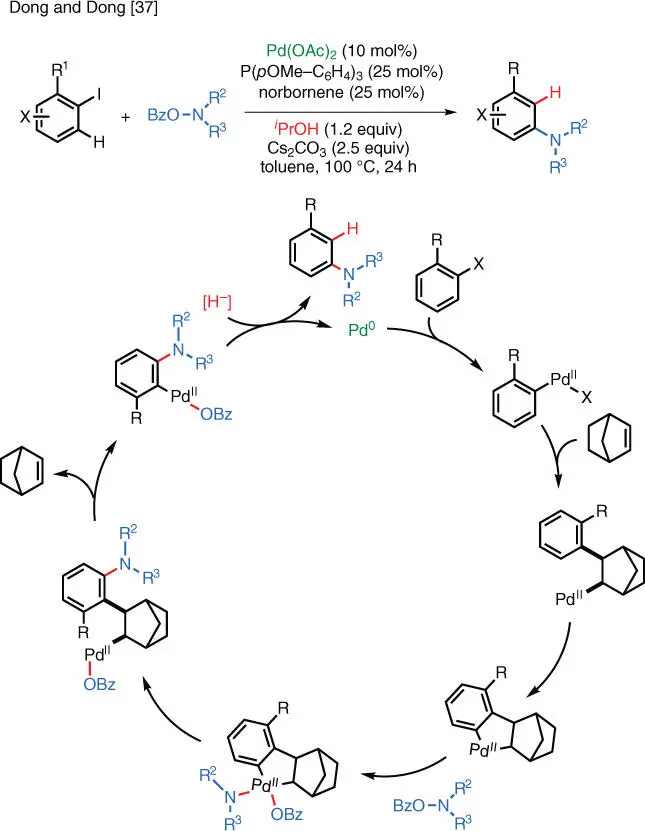
Scheme 1.26 Pd‐catalyzed Catellani‐type C–H electrophilic amination.
Source: Modified from Dong and Dong et al. [37].
1.4 Electrophilic Amination with Hydroxylamine‐derived Metallanitrenes
A nitrene is the nitrogen analog of a carbene, which is a six‐electron electron‐deficient species. Although it is formally uncharged and univalent, it can be considered to be an electrophile because of the unsatisfied octet. These highly reactive intermediates are involved in many chemical reactions, including electrophilic aminations [39–41].
Nitrene intermediates can be generated from various precursors. Pioneering studies by Khan and Kwart in the 1960s showed that elemental copper can catalyze the decomposition of a sulfonyl azide, and the resulting reaction mixture can aminate cyclohexene and give aziridine as one of the products [42]. This result is consistent with the participation of metallanitrene intermediates. However, because of the instability and toxicity of organic azides, this procedure is not widely adapted by the organic chemistry community. In later years, ( N ‐(sulfonyl)imino)phenyliodinane was found to be a suitable alternative nitrogen source. In addition to Cu, Mn and Fe are also capable of catalyzing the formation of metallanitrenes. These “tamed” nitrenes are more stable and have longer lifetimes, making them uniquely suitable for synthetic studies. However, the N ‐sulfonyl groups are hard to remove, limiting the usefulness of the resulting products.
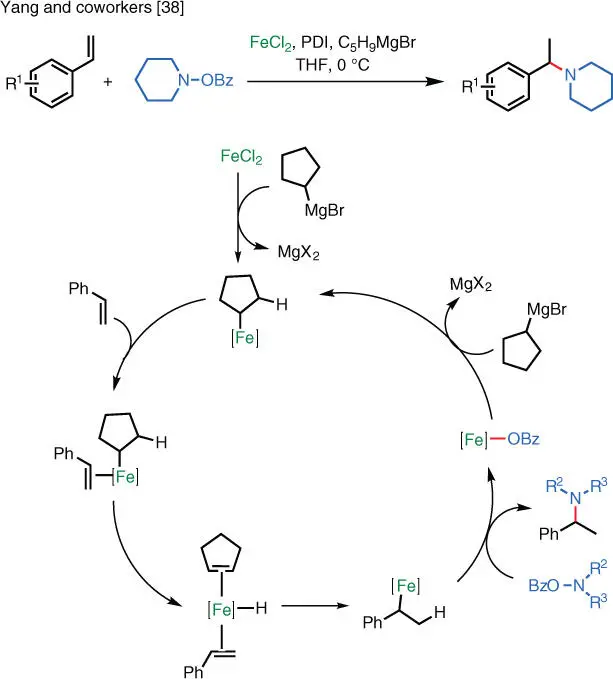
Scheme 1.27 Fe‐catalyzed electrophilic amination of styrenes.
Source: Modified from Huehls et al. [38].
Recently, studies have shown that hydroxylamines can also serve as the nitrogen sources for metallanitrenes. Using hydroxylamines, it is now possible to generate N–H and N–alkyl amine products without sulfonyl groups. This section focuses on reactions that involve hydroxylamine‐derived metallanitrenes.
Metallanitrenes are generated from O‐activated hydroxylamines, in which the nitrogen atom bears a strong leaving group and a transition metal catalyst. In 2014, Kürti, Falck, and coworkers developed an N–H aziridination procedure for unactivated olefins using O ‐(2,4‐dinitrophenyl)hydroxylamine (DPH) as the nitrogen source and a dirhodium carboxylate catalyst. Computational studies conducted by the Ess group support the intermediacy of a Rh–nitrene pathway ( Scheme 1.28) [43].
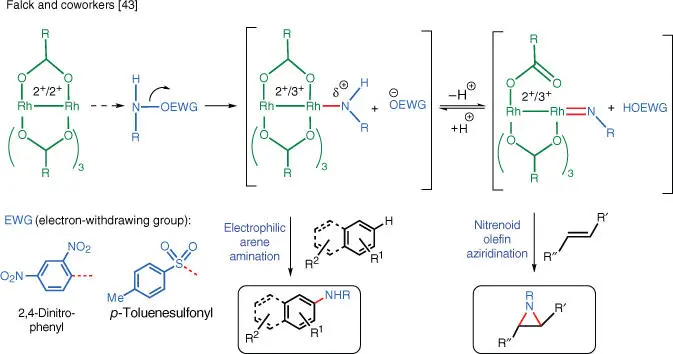
Scheme 1.28 Rh‐catalyzed formation of metallanitrenes.
As the smallest nitrogen‐containing heterocycles, aziridines are important synthetic building blocks for the synthesis of amines. Recently, it has been shown that natural product analogs containing aziridine functionalities can possess improved biological activities than their natural counterparts [44]. Despite their importance, the majority of literature has focused on the synthesis of aziridines bearing electron‐withdrawing sulfonyl groups on the nitrogen atom (i.e. N ‐tosylaziridines). The stability of the sulfonyl group renders the deprotection and functionalization of these aziridines difficult. In comparison, unprotected N–H aziridines can be further functionalized with relative ease.
The transformation is operationally simple as neither the aminating reagent nor the catalyst is air‐ or moisture‐sensitive. The functional group tolerance is excellent as hydroxyl groups, epoxides, and esters in the substrates are unaffected. The NH‐aziridination is stereospecific and no scrambling of the olefin stereochemistry is observed even in sensitive styrene‐type substrates. When more than one C—C double bond is present in the substrate, the more electron‐rich one undergoes aziridination preferentially. The aziridination of terminal double bonds requires a slightly higher catalyst loading. The triple bond of alkynes and electron‐deficient double bonds (i.e. α,β‐unsaturated carbonyl compounds) remain unchanged under the reaction conditions. The choice of solvent is important because the presence of trifluoroethanol is required for aziridination.
The first iteration of this reaction uses the nitro group‐containing DPH as the aminating reagent ( Scheme 1.29). Because of its thermal instability, there were some concerns about its safety in industrial‐scale settings. To address this issue, the Kürti group developed an improved version of this reaction in 2017 [45]. Instead of the nitro‐containing DPH, the new version can proceed with the stable and inexpensive hydroxylamine‐ O ‐sulfonic acid (HOSA) ( Scheme 1.30). Because HOSA is a zwitterionic compound, its solubility in common organic solvents is very low. However, through the addition of an equivalent of a mild base (i.e. pyridine), HOSA can be solubilized. Hexafluoroisopropanol (HFIP) was identified as the optimal solvent, and under these modified conditions, the olefin aziridination reaction can proceed at room temperature and with generally higher rates compared to the original DPH process. The scope of substrates is further expanded and now includes nitrogen heterocycles with basic nitrogen atoms. A further advantage is that the inorganic sulfate by‐product is water‐soluble and nontoxic, which greatly simplifies the purification.
Читать дальше
















