19 19 Heller, H.G. and Oliver, S. (1981). Photochromic heterocyclic fulgides. Part 1. Rearrangement reactions of (E)‐α‐3‐furylethylidene(isopropylidene)succinic anhydride. J. Chem. Soc. Perkin Trans. 1: 197–201.
20 20 Irie, M. and Mohri, M. (1988). Thermally irreversible photochromic systems. Reversible photocyclization of diarylethene derivatives. J. Org. Chem. 53 (4): 803–808.
21 21 Irie, M. (2000). Diarylethenes for memories and switches. Chem. Rev. 100 (5): 1685–1716.
22 22 Irie, M., Fukaminato, T., Matsuda, K., and Kobatake, S. (2014). Photochromism of diarylethene molecules and crystals: memories, switches and actuators. Chem. Rev. 114: 12174–12277.
23 23 Irie, M. (1990). Photoresponsive polymers. Adv. Polym. Sci. 94: 27–67.
24 24 Irie, M., Hirano, Y., Hashimoto, S., and Hayashi, K. (1981). Photoresponsive polymers. 2. Reversible solution viscosity change of polyamides having azobenzene residues in the main chain. Macromolecules 14 (2): 262–267.
25 25 Irie, M. and Schnabel, W. (1981). Photoresponsive polymers. On the dynamics of conformational changes of polyamides with backbone azobenzene groups. Macromolecules 14 (5): 1246–1249.
26 26 Irie, M. and Tanaka, H. (1983). Photoresponsive polymers. 5. Reversible solubility change of polystyrene having azobenzene groups. Macromolecules 16 (2): 210–214.
27 27 Irie, M. and Schnabel, W. (1985). On the dynamics of photostimulated conformational change of polystyrene with pendant azobenzene groups in solution. Macromolecules 18 (3): 394–398.
28 28 Irie, M. and Iga, R. (1986). Photoresponsive polymers. 9. Photostimulated reversible sol‐gel transition of polystyrene with pendant azobenzene groups in carbon disulfide. Macromolecules 19 (10): 2480–2484.
29 29 Irie, M. and Schnabel, W. (1982). Photochemical conversion of poly‐2,3‐diphenylbutadiene to poly‐9,10‐dimethylenephenanthrene. Eur. Polym. J. 18: 15–18.
30 30 Hanazawa, M., Sumiya, R., Horikawa, Y., and Irie, M. (1992). Thermally irreversible photochromic systems. Reversible photocyclization of 1,2‐bis(2‐methylbenzo[b]thiophen‐3‐yl)perfluorocycloalkene derivatives. J. Chem. Soc., Chem. Commun. 3: 206–207.
31 31 Uno, K., Niikura, H., Morimoto, M. et al. (2011). In situ preparation of highly fluorescent dyes upon photoirradiation. J. Am. Chem. Soc. 133: 13558–13564.
2 Reaction Mechanism
Thermal irreversibility is an essential and indispensable property for the applications of molecular photoswitches to memory media and switches. Although tremendous efforts were made in the 1970–1980s to provide the thermal irreversibility to molecular photoswitches, all attempts to modify existing photoswitchable molecules failed. We had to wait until the thermally stable molecular photoswitches were serendipitously discovered. In the beginning and the middle of the 1980s, it was found that furylfulgides and diarylethenes undergo thermally irreversible photoswitching. The photogenerated colored isomers practically never revert back to the colorless isomers at room temperature. Although they undergo thermally irreversible photoswitching, the reason why the molecules show the thermal stability was not understood. It was a crucial task to reveal the reason. The basic principle behind the thermally irreversible photoswitching reactivity was elucidated using both theoretical and experimental approaches, as follows.
The 2,3‐diphenyl‐2‐butene unit, shown in Figure 1.3, underwent a thermally reversible photoswitching reaction in a deaerated solution, while the 2,3‐di(2,5‐dimethyl‐3‐thienyl)‐2‐butene unit, shown in Figure 1.4B(b)exhibited a thermally irreversible reactivity. The photogenerated closed‐ring form of 2,3‐di(2,5‐dimethyl‐3‐thienyl)‐2‐butene was found to remain stable and practically never returned to the open‐ring form at room temperature. In addition, the open‐ring isomer was stable even at elevated temperatures and no thermochromic reaction was observed. To reveal the reason why the diarylethene having phenyl rings and that having thiophene rings exhibit such a different reactivity, semiempirical modified neglect of diatomic overlap (MNDO) calculation was carried out for diarylethene derivatives 9–12( Scheme 2.1) [1].
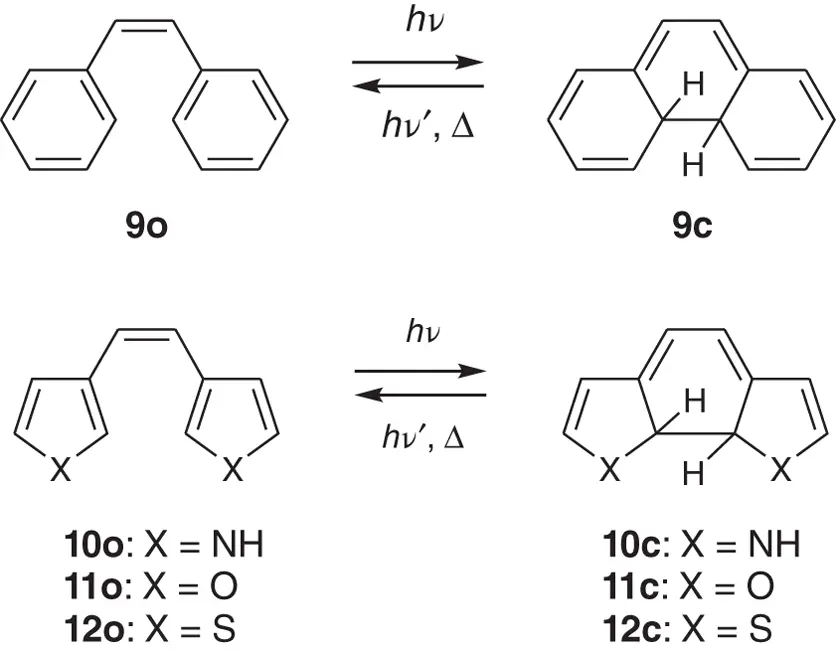
Scheme 2.1 Electrocyclic reactions of diarylethenes 9− 12.
For electrocyclic reactions, two modes of geometrical structure changes, conrotatory and disrotatory, are distinguished, as shown in Scheme 2.2. According to the Woodward‐Hoffmann rule [2] based on π‐orbital symmetries for 1,3,5‐hexatriene (HT), which is the simplest molecular framework of the above molecules, the conrotatory cyclization reaction to cyclohexadiene (CHD) is brought about by light and the disrotatory cyclization by heat.
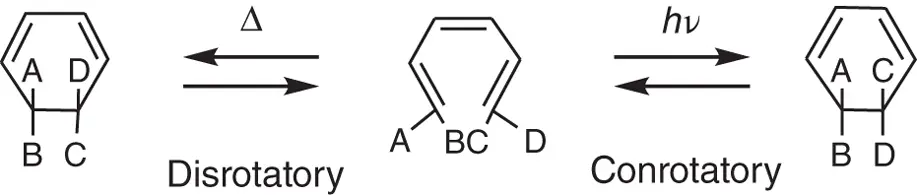
Scheme 2.2 Disrotatory and conrotatory cyclization reactions of hexatriene.
The cycloreversion reaction is allowed photochemically in the conrotatory mode and thermally in the disrotatory mode. From this simple symmetry consideration of the HT molecular framework, the thermal stability of the open‐ring isomer of 2,3‐di(2,5‐dimethyl‐3‐thienyl)‐2‐butene and thermal irreversibility in the cycloreversion reaction cannot be explained. A state energy calculation is necessary to discuss the thermal stability.
Figures 2.1and 2.2show the state correlation diagrams for the electrocyclic reactions of 9and 11in disrotatory and conrotatory modes, respectively. Full lines in the figures show that interconnecting states belong to the same symmetry groups. The relative ground state energy differences between the open‐ and closed‐ring isomers are shown in Table 2.1. The two heterocyclic rings were assumed to be in the parallel orientation for the disrotatory reaction and in the antiparallel orientation for the conrotatory reaction. As can be seen from Figure 2.1, orbital symmetry allows the disrotatory cyclizations in the ground states from 9oto 9cand from 11oto 11c. The relative ground state energies of the closed‐ring isomers of 9and 11are, however, 175 and 113 kJ/mol higher than the respective energies of the open‐ring isomers. This indicates that the open‐ring isomers are stable and the thermal cyclization reaction does not take place practically in both cases.
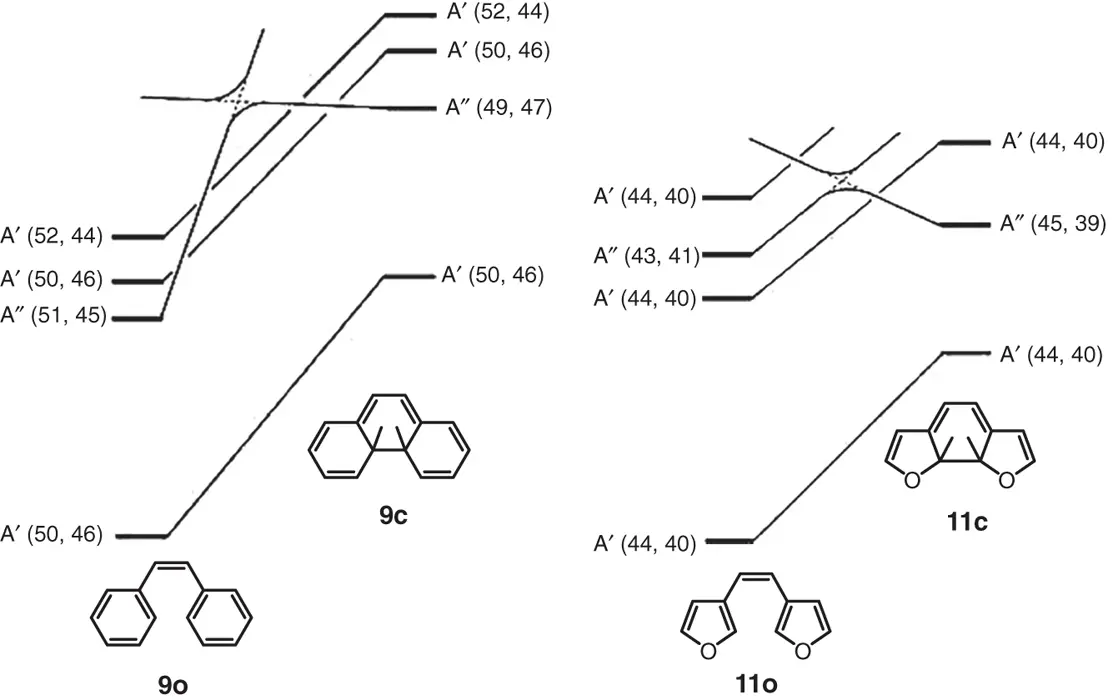
Figure 2.1 State correlation diagrams for the electrocyclic reactions in disrotatory mode. 9cand 11care the closed‐ring isomers, in which two hydrogens attached to the reactive central carbons are in a cis configuration.
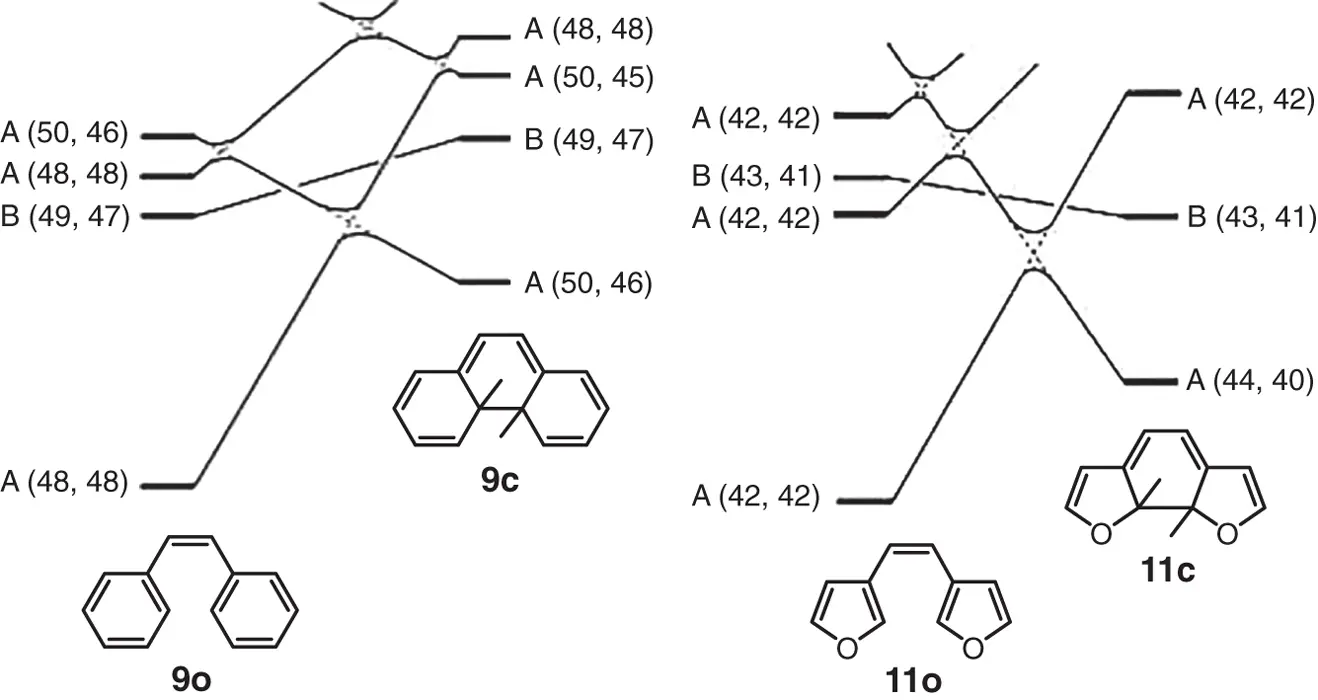
Figure 2.2 State correlation diagrams for the electrocyclic reactions in conrotatory mode. 9cand 11care closed‐ring isomers, in which two hydrogens attached to the reactive central carbons are in a trans configuration.
Читать дальше
















