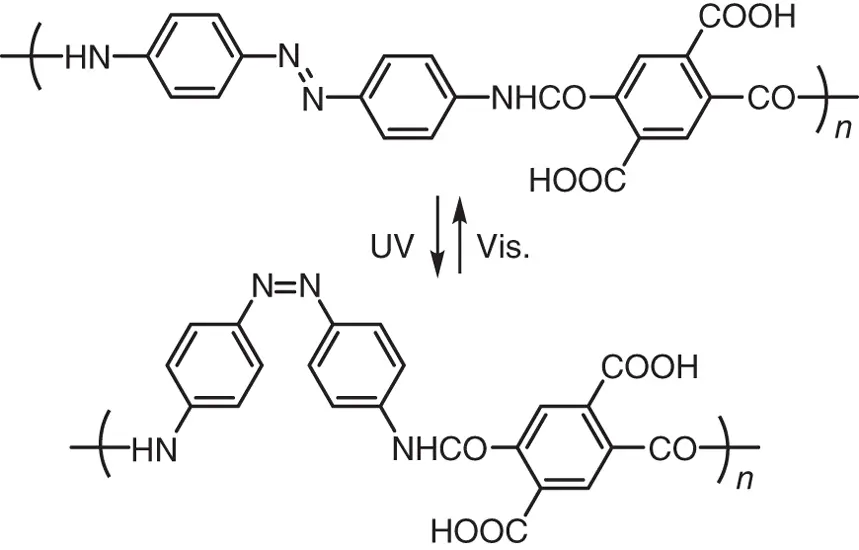
Figure 1.2 Schematic illustration of the photoinduced conformational change of a polymer having azobenzene units in the backbone.
Just like azobenzene, stilbene also undergoes the trans–cis photoisomerization reaction. The photoresponsive polymer research was extended to polymers having stilbene units. A polymer having stilbene units in the backbone can be prepared by 1,4‐addition radical polymerization of 2,3‐diphenylbutadiene, which is prepared from acetophenone, as shown in Figure 1.3[29]. Upon irradiation with 313‐nm light, the poly(2,3‐diphenylbutadiene) efficiently underwent photocyclization reactions to produce a polymer having yellow colored dihydrophenanthrene units in a deaerated dichloromethane solution, instead of the trans – cis photoisomerization. The trans–cis photoisomerization of stilbene units in the backbone was strongly suppressed due to rigidity of the polymer chain. The dihydrophenanthrene units readily returned to the initial 2,3‐diphenyl‐2‐butene units and the yellow color disappeared in less than 10 minutes at room temperature.
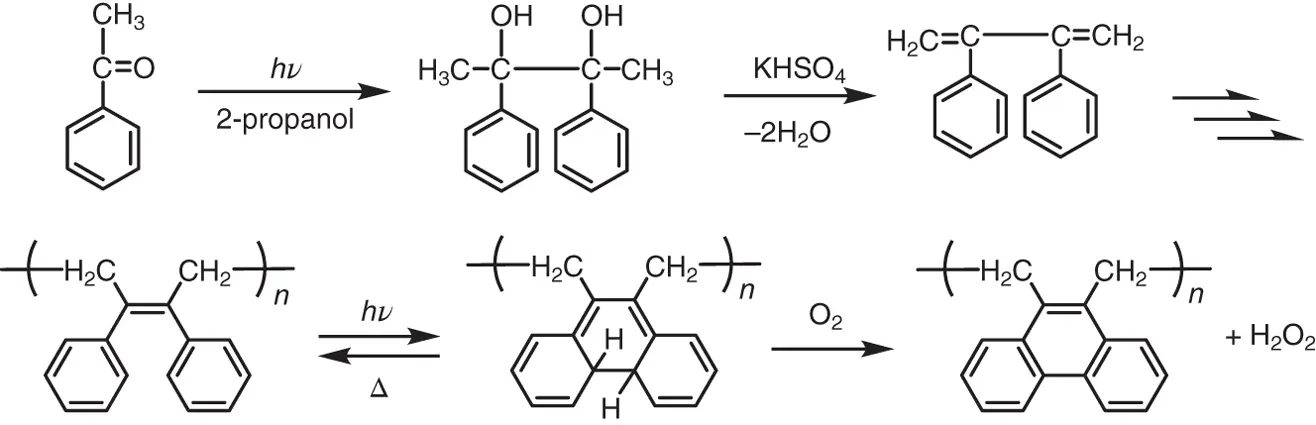
Figure 1.3 A synthesis route of poly(2,3‐diphenylbutadiene) and its photochemical and thermal reactions.
On the other hand, in the presence of air the dihydrophenanthrene units converted to phenanthrene units by hydrogen elimination and the reversibility was lost. To prevent hydrogen elimination and provide reversibility even under aerated conditions, 2,3‐dimesitylbutadiene was designed ( Figure 1.4A(a)). The synthesis of 2,3‐dimesitylbutadiene was attempted by photoreduction of 2,4,6‐trimethylacetophenone, as shown in Figure 1.4B(a). But, the synthesis of pinacol failed because of the bulky size of the mesityl group. To reduce steric hindrance, the mesitylene was replaced with 2,5‐dimethylthiophene ( Figure 1.4A(b)). According to the synthetic route shown in Figure 1.4B(b), 2,3‐bis(2,5‐dimethyl‐3‐thienyl)butadiene was successfully synthesized from 2,5‐dimethyl‐3‐acetylthiophene. The butadiene was polymerized to poly(2,3‐bis(2,5‐dimethyl‐3‐thienyl)butadiene) by 1,4‐addition radical polymerization.
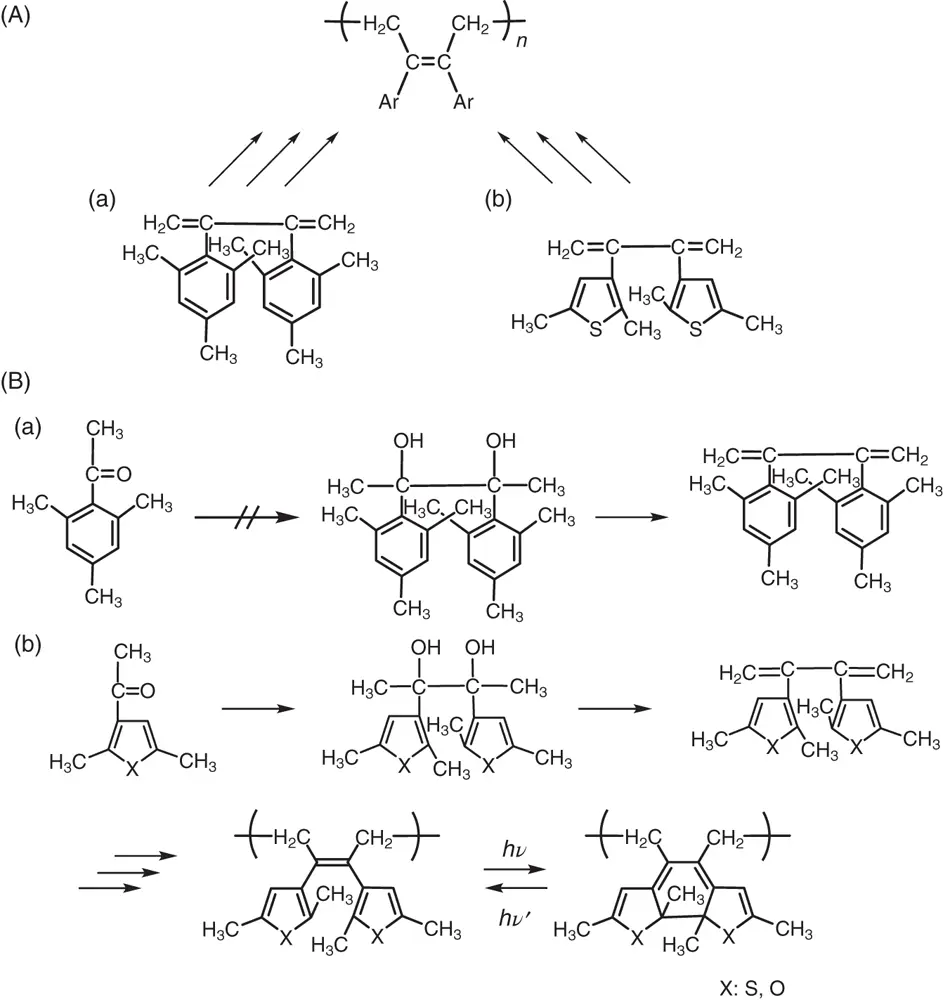
Figure 1.4 (A) Synthesis of polymers having (a) 2,3‐dimesitylbutene units and (b) 2,3‐di(2,5‐dimethyl‐3‐thienyl)butene units in the backbone. (B) (a) A synthetic route to prepare 2,3‐dimesitylbutadiene. (b) Synthetic routes and photochemical reactions of poly(2,3‐di(2,5‐dimethyl‐3‐thienyl)butadiene) and poly(2,3‐di(2,5‐dimethyl‐3‐furyl)butadiene).
The polymer having 2,3‐dithienyl‐2‐butene units was dissolved in benzene and the solution was irradiated with 313‐nm light. The colorless solution turned yellow ( λ max∼ 430 nm) along with the formation of cyclized closed‐ring isomers. The yellow color disappeared upon irradiation with visible light. In contrast to poly(2,3‐diphenylbutadiene), the yellow color of the closed‐ring isomer units was found to remain stable overnight in the dark. The yellow closed‐ring units were stable even at 100 °C and returned to the initial colorless open‐ring isomer units with visible light. The dithienylethene unit in the polymer was unprecedentedly found to undergo a thermally irreversible photoswitching reaction. Poly(2,3‐bis(2,5‐dimethyl‐3‐furyl)butadiene) also underwent the thermally irreversible photoswitching reaction. The amazing result led us to study the photochemistry of the monomer unit, 2,3‐di(2,5‐dimethyl‐3‐thienyl)‐2‐butene and its derivatives in detail. This is the course of serendipitous discovery of diarylethene molecular photoswitches.
Since the discovery of thermally irreversible diarylethene molecular photoswitches in the middle of 1980s, various types of diarylethene derivatives have been synthesized to improve their photoswitching performance. Figure 1.5shows a list of main diarylethene derivatives developed in Kyushu University and Rikkyo University until 2017. Upon irradiation with UV light, 2,3‐di(2,5‐dimethyl‐3‐thienyl)‐2‐butene underwent a cis – trans isomerization in addition to the cyclization reaction. To prevent the unfavorable cis – trans photoisomerization, a cyclic bridge, such as maleic anhydride or maleimide, was introduced. Although diarylethene derivatives with the maleic anhydride or maleimide bridge showed photocyclization reactivity in less polar solvents, the reactivity was strongly suppressed in polar solvents, such as methanol or acetonitrile. To provide photoswitching reactivity even in polar solvents, the ethene bridges were replaced with perfluorocycloalkenes with four‐, five‐, and six‐membered rings [30]. The 1,2‐bis(2‐methyl‐1‐benzothiophen‐3‐yl)perfluorocycloalkenes underwent reversible photoinduced cyclization/cycloreversion reactions in polar methanol and acetonitrile. Among the three derivatives having four‐, five‐, and six‐membered rings, five‐membered 1,2‐bis(2‐methyl‐1‐benzothiophen‐3‐yl)perfluorocyclopentene was found to offer the highest resistance to photofatigue. Since then, perfluorocyclopentene derivatives have been mainly studied.
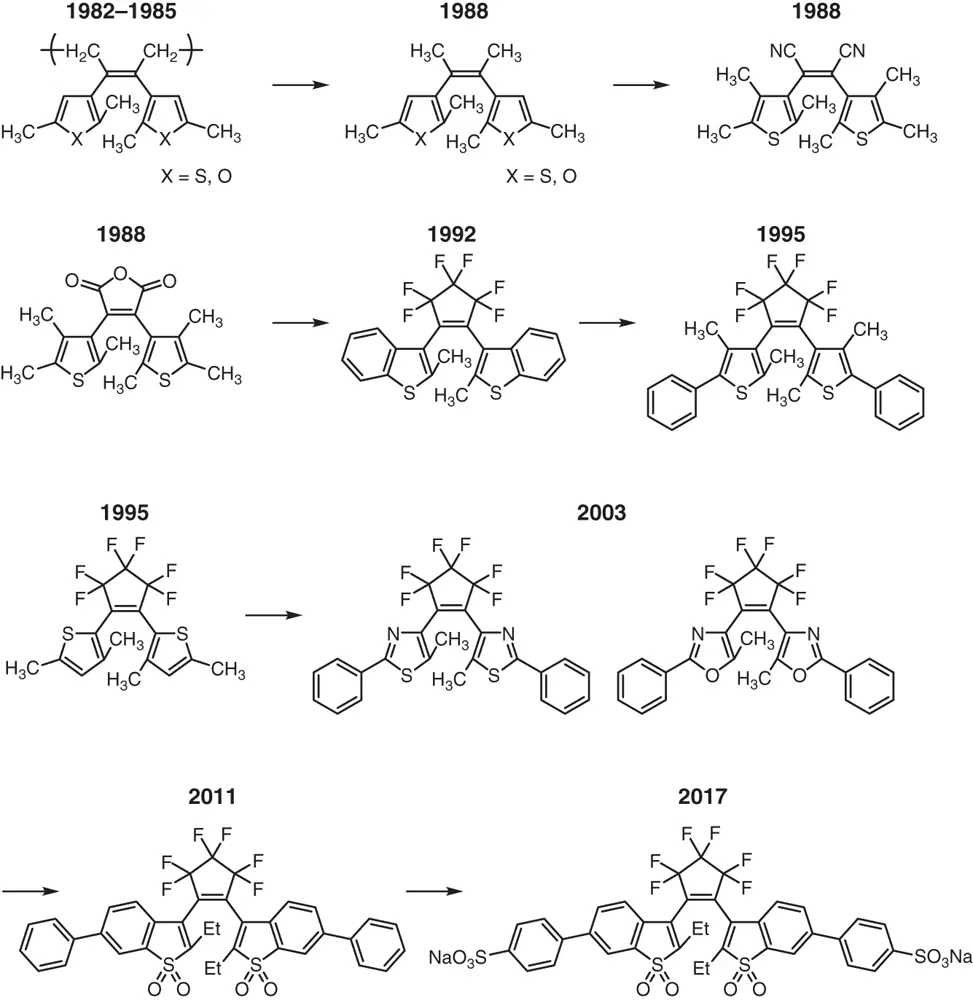
Figure 1.5 Development of diarylethene molecular photoswitches.
Although diarylethene photoswitches exhibit brilliant color changes upon photoirradiation, most of them are nonfluorescent or very weakly fluorescent in both isomer forms. It was a long‐standing ambition to prepare photoswitchable fluorescent diarylethenes without attaching fluorescent chromophores to the diarylethenes. In 2011, sulfone derivatives of 1,2‐bis(2‐ethyl‐6‐aryl‐1‐benzothiophen‐3‐yl)perfluorocyclopentene were found to exhibit very strong fluorescence (fluorescence quantum yield ∼ 0.9) in the closed‐ring isomers [31]. The turn‐on mode fluorescent diarylethenes are now extensively applied to super‐resolution fluorescence microscopy in materials science and biological systems. Diarylethenes are able to switch both absorption (color) and fluorescence emission upon photoirradiation.
At first sight, the most striking phenomenon observed in molecular photoswitches is a photoinduced instantaneous color change. Figure 1.6shows photos of the color changes of diarylethene derivatives in solution. When the toluene solutions of the derivatives are irradiated with UV light, the colorless solutions turn yellow, orange, red, violet, blue, cyan, and green. The chemical structures of the derivatives are shown below in the photos. These colors disappear upon irradiation with visible light. The photoinduced coloration/decoloration cycles upon alternate irradiation with UV and visible light can be repeated many times.

Figure 1.6 Color changes of diarylethene derivatives 1–7in toluene upon irradiation with UV and visible light.
Читать дальше

















