The color changes are ascribed to the electronic structure changes of the derivatives from the open‐ to the closed‐ring isomers. Two typical examples of the electronic structure changes are shown in Figure 1.7. 1,2‐Bis(2,5‐dimethyl‐3‐thienyl)perfluorocyclopentene ( 3) and 1,2‐bis(3,5‐dimethyl‐2‐thienyl)perfluorocyclopentene ( 1) undergo reversible electrocyclic rearrangements. The electrocyclic reactions involve rearrangements of positions of single and double bonds in a molecule. During the reactions, a new single bond is made between the central reactive carbon atoms by the cyclization reaction and the bond is broken as the ring is opened.
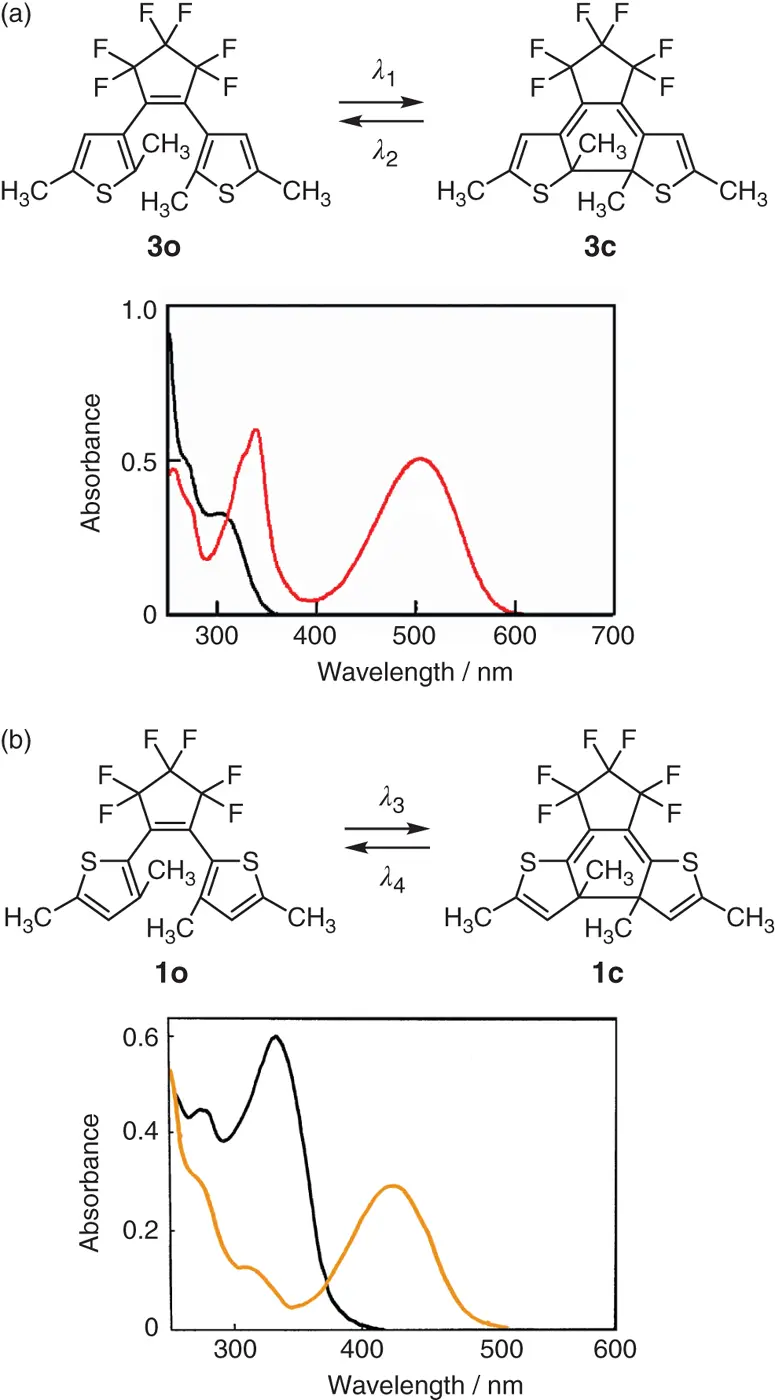
Figure 1.7 Chemical structures and absorption spectra of open‐ and closed‐ring isomers of (a) 3and (b) 1in n‐ hexane.
Figures 1.7shows the chemical structures of the open‐ and the closed‐ring isomers and their absorption spectra. In both derivatives 3and 1, upon irradiation with appropriate wavelength of light ( λ 1or λ 3) a single bond is formed between the central reactive carbon atoms and the double bonds change the position. Upon irradiation with another wavelength of light ( λ 2or λ 4) the single bond is broken and the molecule returns to the initial structure. The color is controlled by the length of π‐conjugation. In the open‐ring isomers, two thiophene rings have no particular interaction and the spectra are comparable to substituted thiophenes. In the closed‐ring isomers, the π‐conjugation length depends on the attached position of thiophene rings to the ethene bridge. When the thiophene rings are attached to the ethene bridge at 3‐position, such as derivative 3, π‐conjugation is delocalized throughout the molecule in the closed‐ring isomer and the delocalization results in red color. The π‐conjugation is further extended when phenyl groups are substituted at 5‐ and 5′‐positions of the thiophene rings, such as 5. The long π‐conjugation shifts the absorption band to longer wavelengths, resulting in blue color. On the other hand, when the thiophene rings are attached to the ethene bridge at 2‐position, such as derivative 1, π‐conjugation is localized in the central part. The short π‐conjugation results in yellow color in the closed‐ring isomer.
The photoswitching between two discrete states and thermal irreversibility of the two states are indispensable for applications to memory media and switching devices. The bistability is a basic characteristic of diarylethenes. Although the chemical structures of the two isomers suggest photoswitching between two discrete states, in general, the absorption and fluorescence spectra gradually change upon photoirradiation in ensemble systems. Figure 1.8bshows the photoswitching performance of 1,2‐bis(2‐ethyl‐6‐phenyl‐1‐benzothiophene‐1,1‐dioxide‐3‐yl)perfluorocyclopentene ( 8, Figure 1.8a) in the ensemble system in 1,4‐dioxane. The gradual analog increase in the fluorescence intensity upon irradiation with UV light indicates a change in the concentrations of the two isomers. Upon UV irradiation the open‐ring isomers convert to the fluorescent closed‐ring isomers and the concentration ratio of the closed‐ring isomers increases, causing the gradual increase in the fluorescence intensity. Subsequently, upon irradiation with visible light the ratio of the closed‐ring isomers decreases, resulting in disappearance of the fluorescence. The switching between the two discrete states cannot be discerned from the photoswitching performance in the ensemble system.
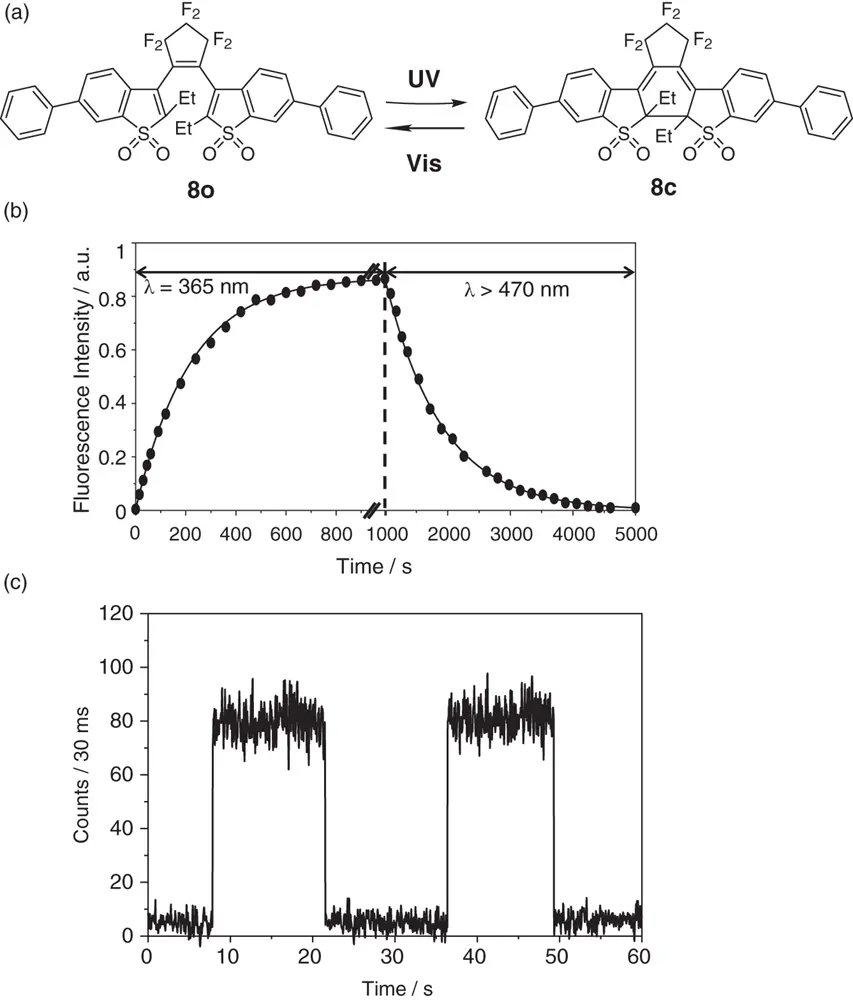
Figure 1.8 (a) Chemical structures of open‐ and closed‐ring isomers of 8. (b) Fluorescence photoswitching of 8upon irradiation with UV and visible light in the ensemble system in 1,4‐dioxane. (c) Fluorescence photoswitching of 8at the single‐molecule level in a Zeonex polyolefin film.
Digital on/off photoswitching between two discrete states was confirmed by measuring the switching response at a single‐molecule level. Figure 1.8cshows the fluorescence photoswitching of a single molecule of derivative 8upon alternate irradiation with UV and visible light. Upon irradiation with UV light, the fluorescence abruptly switches from the off ‐state to the on ‐state, while upon irradiation with visible light the on ‐state abruptly returns to the off ‐state. The digital photoswitching response definitely indicates that diarylethene photoswitch 8has bistable states. The photoisomerization between two discrete isomer states expressed by the two chemical structures is experimentally evidenced.
1 1 Barltrop, J.A. and Coyle, J.D. (1975). Excited States in Organic Chemistry. London: Wiley.
2 2 Turro, N.J. (1978). Modern Molecular Photochemistry. Menlo Park, CA: Benjamin/Cummings Pub.
3 3 Balzani, V., Ceroni, P., and Juris, A. (2014). Photochemistry and Photophysics: Concept, Research, Applications. Weinheim: Wiley‐VCH.
4 4 Brown, G.H. (1971). Photochromism. New York: Wiley‐Interscience.
5 5 Crano, J.C. and Guglielmetti, R.J. (1998). Organic Photochromism and Thermochromic Compounds. New York: Plenum Press.
6 6 Bouas‐Laurent, H. and Dürr, H. (2001). Organic photochromism. Pure Appl. Chem. 73: 639–665.
7 7 Dürr, H. and Bouas‐Laurent, H. (eds.) (2003). Photochromism: Molecules and Systems. Amsterdam: Elsevier.
8 8 Irie, M., Yokoyama, Y., and Seki, T. (eds.) (2013). New Frontiers in Photochromism. Tokyo: Springer.
9 9 Tian, H. and Zhang, J. (eds.) (2016). Photochromic Materials: Preparation, Properties and Applications. Weinheim: Wiley‐VCH.
10 10 Yokoyama, Y. and Nakatani, K. (eds.) (2017). Photon‐Working Switches. Tokyo: Springer.
11 11 Hartley, G.S. (1937). The cis‐form of azobenzene. Nature 140: 281.
12 12 Fischer, E. and Hirshberg, Y. (1952). Formation of coloured forms of spiropyrans by low‐temperature irradiation. J. Chem. Soc.: 4522–4524.
13 13 Iwahori, F., Hatano, S., and Abe, J. (2007). Rational design of a new class of diffusion‐inhibited HABI with fast back‐reaction. J. Phys. Org. Chem. 20: 857–863.
14 14 Hampson, G.C. and Robertson, J.M. (1941). Bond lengths and resonance in the cis‐azobenzene molecule. J. Chem. Soc.: 409–413.
15 15 Bullock, D.J.W., Cumper, C.W.N., and Vogel, A.I. (1965). Physical properties and chemical constitution. Part XLLIII. The electric dipole moments of azobenzene, azopyridines, and azoquinolines. J. Chem. Soc.: 5316–5323.
16 16 Malic, E., Weber, C., Richter, M. et al. (2011). Microscopic model of the optical absorption of carbon nanotubes functionalized with molecular spiropyran photoswitches. Phys. Rev. Lett. 106: 097401.
17 17 Kryszewski, M., Nadolski, B., North, A.M., and Pethrick, R.A. (1980). Kinetic matrix effects (response and density distribution function): ring closure reaction of indolinobenzospiropyrans in glassy poly(alkyl methacrylates). J. Chem. Soc., Faraday Trans II 76: 351–368.
18 18 Fujita, K., Hatano, S., Kato, D., and Abe, J. (2008). Photochromism of a radical diffusion‐inhibited hexaarylbiimidazole derivative with intense coloration and fast decolouration performance. Org. Lett. 10 (14): 3105–3108.
Читать дальше














