Fast ion conductor ceramics, also known as active inorganic electrolytes, exhibit a high ionic conductivity of up to 10 −2S/cm at 25 °C. Composite of fast ion conductor ceramics with polymer can strengthen interfacial contact. Since 2009, polymer‐in‐ceramic (PIC) approach was introduced into the field of composite polymeric electrolytes [111]. It consists of introducing polymer salt complexes into porous ceramic structures. The studied systems exhibited excellent mechanical properties, high conductivities, and good stability vs Li metal electrodes under prolonged storage [111]. As was reported recently, composite electrolytes from “ceramic‐in‐polymer” (CIP) to PIC with different sizes of garnet particles are investigated for their effectiveness in dendrite suppression [112]. With a large specific surface area, nanoscale garnet ceramic fillers improve the transition rate of ions [113]. The PIC electrolyte with 80 vol% 5 μm Li 6.4La 3Zr 1.4Ta 0.6O 12(LLZTO) (PIC‐5 μm) shows the highest tensile strength of 12.7 MPa [113]. The morphology, distribution, and three‐dimensional structure of ceramic filler particles may affect the ionic conductivity of polymer composite electrolyte [114]. Cui et al. explored the different morphologies of LLZO to increase ionic transport [115]. These LLZO nanowires show an ionic conductivity of 6.05 × 10 −5S/cm at 30 °C. A polymer–ceramic composite electrolyte of PEO and Li 0.33La 0.557TiO 3nanowires has been studied and exhibited the lithium‐ion conductivity of 2.4 × 10 −4S/cm at 25 °C [116]. Furthermore, glass–ceramic electrolytes are another solid ceramic electrolytes in which ions transfer through the ceramic phase by means of vacancies or interstitials within the lattice. Typical examples of this are glass/glass–ceramic Li 2S‐P 2S 5and ceramic thio‐lithium superionic conductor (LISICON) Li 4 − xGe 1 − xP xS 4(0 < x < 1), which are the most promising electrolytes to increase ion conductivity up to 10 −5S/cm [117].
The effects of ceramic fillers on the ionic conductivity of solid polymer systems have been discussed for quite many years. The published papers show that increasing the conductivity is the key parameter. Polymer‐ceramic composites offer superior mechanical properties and great conductivities. However, exploring ceramics materials is still a challenge.
2.3.5 Ionic Liquid Polymer Electrolytes
ILs are molten salt liquid at room temperature (<100 °C) and consisted of dissociated ions [118]. IL electrolytes have been widely applied in electrochemical application due to non‐volatility, thermal stability, non‐flammability, high ionic conductivity, and a wide electrochemical stability window [119]. Recently, the incorporation of room‐temperature ILs into PEs to prepare solvent‐free PEs has been considered as a promising approach to improve the performance of ECDs. For example, room temperature ionic liquid (RTIL) (1‐butyl‐3‐methyl‐imidazolium hexafluorophosphate [BMIM] PF 6) [120] was reported to increase the optical contrast and coloring efficiency of the ECD. Zhou et al. have studied an RTIL non‐volatile polymer electrolyte based on poly(propylene carbonate) as the host for ECD application [121]. In this work, 1‐butyl‐3‐methylimidazolium tetrafluoroborate (BMIM +BF 4 −) with high ionic conductivity, good electrochemical stability, and low viscosity was added into poly (propylene carbonate) (PPC)/LiClO 4SPE to form novel PEs with improvement in ionic conductivity, thermal and electrochemical properties, and safety [121]. Recently, combining RTIL and ceramic filler into PEs have been explored. Murali and coworker [122] studied zinc ion conducting gel polymer electrolytes (ZCGPEs) made of poly(vinyl chloride) (PVC)/PEMA blend, zinc triflate [Zn(OTf) 2] salt, 1‐ethyl‐3‐methylimidazolium bis(trifluoromethylsulfonyl) imide (EMIMTFSI) IL, and silica (SiO 2), which exhibited the highest ionic conductivity.
PILs are polyelectrolytes consisting of a polymeric backbone and IL components in monomer repeating units. Variability and designability of ILs enrich the performance, which attract great attention in the field of polymer material [123]. Mecerreyes in 2011 reviewed the synthetic aspects of ILs, the synthetic methods of PILs, and their physicochemical properties [124]. Yuan et al. also reported the applications of PILs and ILs [125]. ILs can be widely applied in diverse field through adjusting ionic conductivity, hydrophilicity, hydrophobicity, chemical durability, and thermodynamic stability. Marcilla et al. [126] have reported a new PE based on ILs and PILs in ECDs having configuration PEDOT/electrolyte/PEDOT. The electrolyte was prepared by mixing 1‐butyl‐3‐methylimidazolium‐bis(trifluoromethanesulfonimide) [bmim][Tf 2N], 1‐butyl‐3‐methylimidazolium tetrafluoroborate [bmim][BF 4], and 1‐butyl‐3‐methylimidazolium bromide [bmim][Br], with poly(1‐vinylethyl‐imidazolium) bearing similar counter anions [Tf 2N], [BF 4] and [Br]. By using this electrolyte, the cycle life was significantly increased. Hydrophobic ILs (P(EO) 10lithium bis (trifluoromethanesulfonyl) imide (LiTFSI) + 0.96PYR 14TFSI) electrolyte was reported to improve the performance of the all‐solid‐state ECD [127]. Puguan and Kim reported a switchable single‐molecule ECD derived from a viologen‐tethered triazolium‐based poly(ionic liquid) ( Figure 2.10) [128].
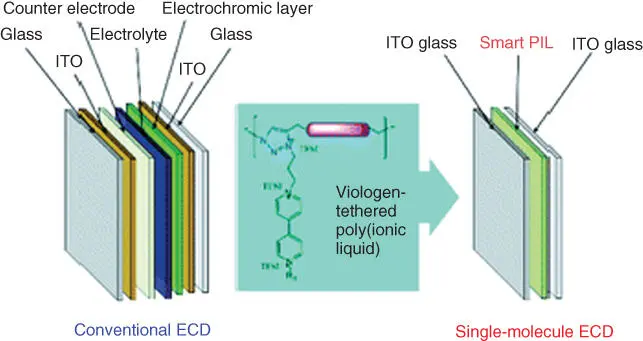
Figure 2.10 A switchable single‐molecule electrochromic device derived from a viologen‐tethered triazolium‐based poly(ionic liquid).
Source: Puguan and Kim [128].
Yuan's group reported a flexible and voltage‐switchable polymer Velcro constructed using host–guest recognition between poly(ionic liquid) strips. [129] The reversible phase transition behavior of PILs can be activated by temperature, electrochemical redox reactions, and chemical redox reactions. Vancso and coworkers prepared a redox‐active organometallic PIL [130]. The prepared polymer Velcro based on PILs exhibited strong adhesion in air and in aqueous solutions (including acidic and basic water and artificial seawater) and could be unfastened and fastened by mechanical, chemical and electrochemical means ( Figure 2.11).
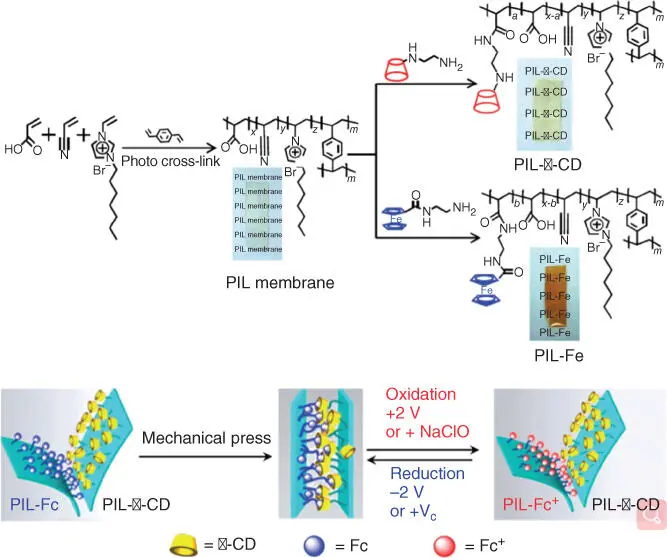
Figure 2.11 Schematic representation of the synthesis of PIL‐b‐CD and PIL‐Fc membranes and the hook‐and‐loop strategy for adhesion based on b‐CD and Fc‐modified PIL membrane surfaces.
Source: Guo et al. [129].
2.3.6 Gelatin‐Based Polymer Electrolytes
Natural polymers can become a promising substitute for synthetic polymers and cause great attention from researchers due to their biodegradability, eco‐friendliness, low production cost, and good physical and chemical properties. Among natural polymers, polysaccharides and proteins are best candidate due to their abundance in environment. Gelatin can be used as PE in EC applications. Gelatin is a polypeptide mainly composed of proline, hydroxyproline, and glycine. Avellaneda et al. [131] have reported a solid gelatin PE prepared by dissolving gelatin, LiClO 4, and glycerol into water for EC applications. The ionic conductivity for this electrolyte reached from 1.53 × 10 −5S/cm at room temperature to 4.95 × 10 −4S/cm at 80 °C. ECDs with the configuration K‐glass/Nb 2O 5:Mo EC‐layer/gelatin‐based electrolyte/(CeO 2) x(TiO 2) 1 − xion‐storage (IS) layer/K‐glass have been assembled and reported to increase the ionic conductivity and good long‐time cyclic stability [132]. Silva et al. explored a solid‐state ECD including gelatin‐based electrolytes [133]. This prototype ECD exhibited good optical density. The ECD display incorporating gelatine I and gelatine II samples presented in the visible region an average transmittance above 68% in the bleached state [133]. A novel gel electrolyte composition combining lithium iodide LiI in 1‐butyl‐3‐methylimidazolium iodide (BMII) IL, triiodide I 3 −/I −redox mediator, and biodegradable gelatin is proposed for ECDs ( Figure 2.12) [134]. Fast switching times and high cycling stability, up to 20 000 cycles, are recorded in this ECD.
Читать дальше














