The hydrogen atom is very small and has only one electron in its 1s orbital. When bonded to an electronegative element such as nitrogen, N, oxygen, O, or fluorine, F, a dipole exists, and the hydrogen atom adopts a partial positive charge (δ+) while the other atom becomes slightly negatively charged (δ−). The partial positive charge is therefore concentrated over a very small volume and makes the hydrogen atom strongly polarising. This allows the hydrogen atom from one molecule to attract electron density from a small electronegative atom (such as nitrogen, oxygen, or fluorine) in a neighbouring molecule, and the hydrogen atom becomes sandwiched between the two more electronegative atoms. This can be seen in Figure 2.32, which depicts the formation of a hydrogen bond between two molecules of water. The highly polarising hydrogen atom in one water molecule (δ+) is able to attract electron density in the form of a lone pair of electrons from a neighbouring oxygen atom (δ−). The lone pair is now ‘shared’ between the hydrogen atom and the oxygen atom of a neighbouring molecule, and a hydrogen bond is formed. Because hydrogen bonding requires both the bonded and the neighbouring atom to be highly electronegative and small, hydrogen bonding generally only takes place between hydrogen and nitrogen, oxygen, and fluorine.

Figure 2.32(a) Formation of a hydrogen bond between two molecules of water where molecules are in constant motion; (b) formation of hydrogen bonds in ice, where molecules are in a fixed position.
In liquid water, hydrogen bonds are constantly forming and breaking, as in Figure 2.32a, whereas in ice ( Figure 2.32b), the molecules are held together in fixed positions by hydrogen bonds. Each oxygen atom can form two hydrogen bonds to neighbouring molecules as it possesses two lone pairs of electrons.
Hydrogen bonds are also formed between different types of molecules. The group of organic molecules known as alcohols contains an —OH bond. Ethanol, C 2H 5OH, is a well‐known alcohol. Hydrogen bonding can occur between molecules of ethanol ( Figure 2.33a). Hydrogen bonding can also occur between molecules of ethanol and molecules of water, as shown in Figure 2.33b.
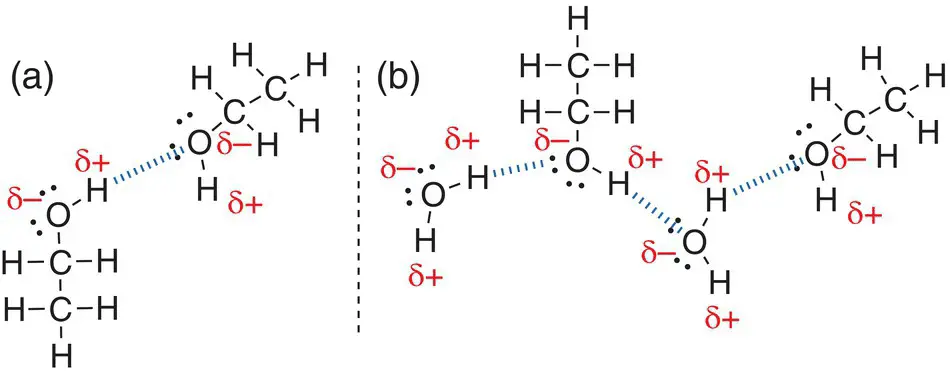
Figure 2.33(a) Hydrogen bonding between two ethanol, C 2H 5OH, molecules; (b) hydrogen bonding between ethanol, C 2H 5OH, and water molecules.
2.4.4 Summary of strengths of intermolecular forces
Intermolecular forces are the weak forces of attraction formed between simple covalently bonded molecules. There are three types of intermolecular forces:
London dispersion forces: instantaneous dipole–induced dipole forces
Permanent dipole–permanent dipole forces
Hydrogen bonding
The order of strengths of these forces is:
instantaneous–induced dipole forces < permanent dipole–permanent dipole forces < hydrogen bonding. Table 2.2gives approximate values for the strengths or energies of different types of intermolecular forces and bond types.
Table 2.2Approximate strengths of different types of bonds and intermolecular forces.
| Bond type/intermolecular force type |
Strength (approx.)/kJ mol −1 |
| Ionic bond (e.g. NaCl) |
771 |
| Single covalent bond (e.g. Cl—Cl) |
242 |
| Instantaneous dipole–induced dipole |
0–10 |
| Permanent dipole–permanent dipole |
5–20 |
| Hydrogen bond |
20–45 |
Determine the types of intermolecular forces present in the following molecules: (a) silane (SiH 4); (b) methanol (H 3COH); and (c) dichloromethane (CH 2Cl 2).
1 Silane is a tetrahedral molecule with four Si—H bonds. The electronegativities of silicon and hydrogen are very similar and the molecule is symmetrical, so there is no overall dipole present. This means that the most likely type of interaction between two silane molecules is instantaneous dipole–induced dipole or London dispersion forces.
2 Methanol contains an O—H bond, so there is a large dipole between the carbon and the oxygen and an even bigger dipole between the hydrogen and the oxygen atoms. This can create permanent dipole to permanent dipole interactions. More specifically, because the hydrogen of the O—H bond is bonded to oxygen, methanol can undergo hydrogen bonding with other molecules of methanol, CH3OH.
3 Dichloromethane contains two carbon‐chlorine bonds, which both have a permanent dipole due to the different electronegativities of carbon and chlorine; carbon is δ+ and chlorine δ−. This is a permanent dipole; therefore, the dominant intermolecular force in dichloromethane is permanent dipole–permanent dipole.
2.4.5 A special case: ion‐dipole intermolecular forces
One final intermolecular force should be considered, although it follows the same rules and conventions as the intermolecular forces already discussed: the ion–dipole force. The ion can be either positively charged (a cation) or negatively charged (an anion). The dipole that it interacts with can be induced, instantaneous, or permanent. It really depends upon the molecules involved.
For example, a sodium ion (Na +) could interact with ethane (a non‐polar molecule) through either of the following:
An instantaneous dipole, where the ethane itself could develop an instantaneous dipole due to random electron movement within the bonds in the ethane molecule
An induced dipole, where the ethane itself could develop an induced dipole due to the close proximity of the sodium ion to it
Another example could be the sodium ion (Na +) ion interacting with methanol (a polar molecule). The methanol itself has a permanent dipole, so the interaction would be between the δ− of the oxygen on methanol and the Na +ion itself.
There are three main types of chemical bonding: metallic, covalent, and ionic. Metallic bonding can be described by a lattice of positive metal ions held together by a sea of negative electrons.
Ions are formed when atoms lose or gains electrons.
Ionic bonding occurs between positive and negative ions held together in a regular lattice by electrostatic forces.
Covalent bonding occurs when atoms share one or more pair of electrons.
Dative covalent bonding occurs when one atom provides both electrons to another atom capable of accepting them.
Covalent bonding results in simple molecular and giant molecular structures.
VSEPR is a theory that allows the prediction of shapes of molecules based upon the arrangement of pairs of electrons around the central atom.
The type of bonding in a compound determines its physical properties.
A polar bond is one that has a permanent dipole moment due to a difference in electronegativities between the atoms in the bond.
A polar molecule possesses an overall dipole moment.
Intramolecular forces are the strong forces within a molecule that hold the atoms together.
Intermolecular forces are weak forces between molecules that hold them together.
There are three main types of intermolecular forces: instantaneous dipole to induced dipole, permanent dipole to permanent dipole, and hydrogen bonding.
1 Draw a dot‐and‐cross diagram to show the bonding present in a molecule of nitrogen, N2.
Читать дальше














