However, simple covalent molecules such as water don't exist in isolation; they are surrounded by many millions of other similar molecules with much weaker forces between them. The forces that exist between molecules are called intermolecular forces. They are much weaker than the forces holding atoms together but are very important. The intermolecular forces holding water molecules together in liquid water are around 1.3 kJ per gram, which is about one‐twentieth the strength of an O—H bond. If it weren't for these intermolecular forces, we wouldn't have any liquid water to drink – water would exist as a gas on the earth's surface.
Intermolecular forces determine the properties of a covalently bonded compound, such as its melting and boiling point. It is important to understand how they arise in order to have an idea of the strength of these forces.
Figure 2.28shows the strong intramolecular forces within the methane, CH 4, molecule and the weaker intermolecular forces between the methane molecules.
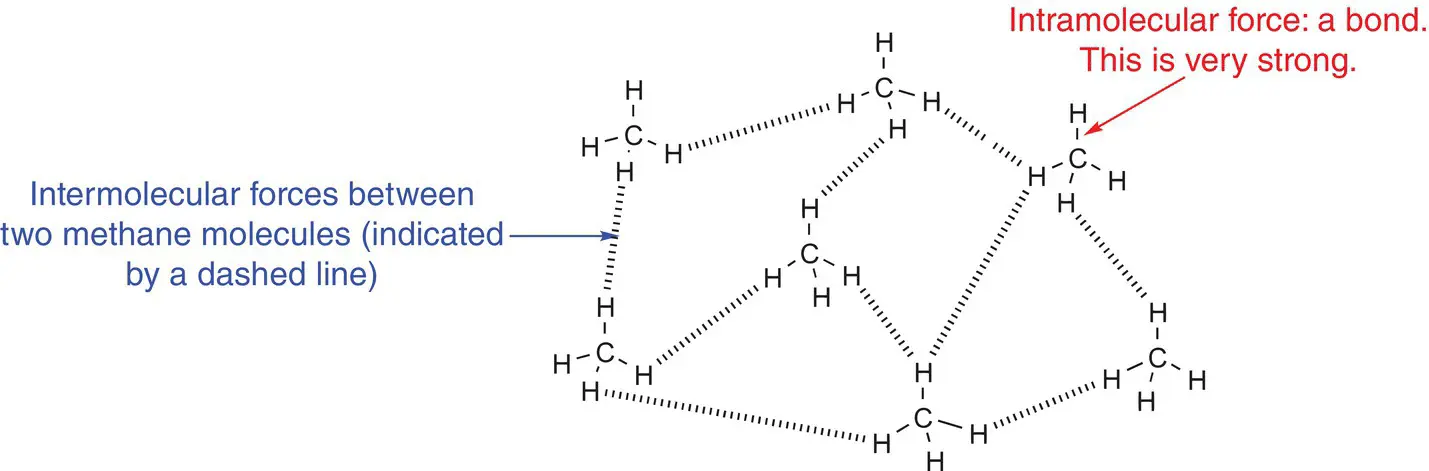
Figure 2.28Comparison of inter‐ and intramolecular forces.
It is important that you understand the difference between inter‐ and intramolecular forces, as it is subtle but very important. Intermolecular forces are between two different molecules (like international flights are between two different countries); intramolecular forces are between the atoms that form a bond and are within a molecule
There are three main types of intermolecular force between molecules:
Instantaneous dipole–induced dipole or London dispersion forces
Permanent dipole–permanent dipole
Hydrogen bonding
The first two types of intermolecular forces that involve dipole‐to‐dipole interactions (both permanent and instantaneous) are called van der Waals forces . Van der Waals forces are attractive forces between slightly positively and slightly negatively charged areas of a molecule. The term van der Waals is reasonably general and does not take into account the type of dipoles that are interacting. The term London dispersion forces is more specific, and this name is used for instantaneous dipole to induced dipole interactions. The third type of intermolecular force, hydrogen bonding, is a special type of dipole–dipole interaction.
The origins of these interactions will be discussed in the following sections.
2.4.1 Permanent dipole–permanent dipole interactions
Permanent dipole–permanent dipole interactions are stronger than London dispersion forces but weaker than hydrogen bonds. These interactions occur through space and are purely electrostatic with a δ+ charge in one molecule interacting with a δ− charge in another molecule. Figure 2.29shows the permanent dipoles set up in HCl molecules, where the chlorine atom is more electronegative than the hydrogen atom. Permanent dipole–permanent dipole interactions are formed between the oppositely charged ends of molecules.
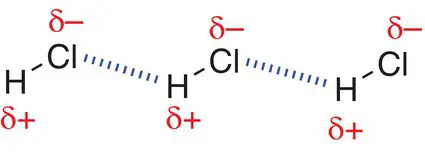
Figure 2.29Permanent dipoles in the hydrogen chloride molecule and resultant permanent dipole–permanent dipole interactions.
2.4.2 London dispersion forces (instantaneous dipole–induced dipole)
An instantaneous dipole can occur in a bond between any two elements, regardless of the electronegativities of the bonded atoms. As the name suggests, they are fleeting and so do not last very long, but they can have an impact upon other molecules that are nearby. The common name for an instantaneous dipole to induced dipole interaction is London dispersion forces .
If the atoms in a bond have similar electronegativities, the electron charge is evenly distributed between them. However, because electrons are constantly moving, there is still a chance that at any one moment, the electrons may suddenly be at one end of the bond, rendering that end of the bond slightly negatively charged (δ−) and the other end of the bond slightly positively charged (δ+) in comparison. This forms an instantaneous dipole. Once an instantaneous dipole has been set up, it induces a dipole in another bond in a nearby molecule: Figure 2.30.
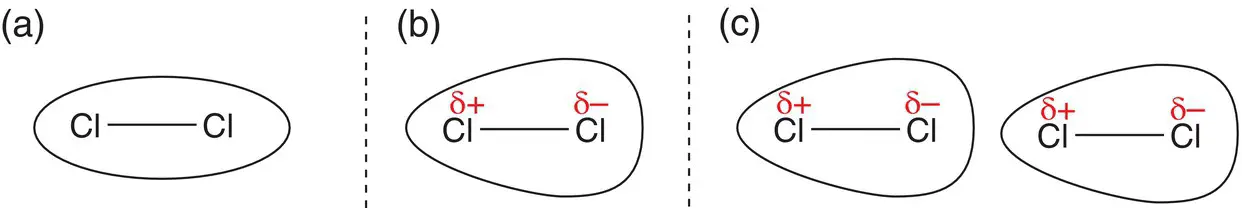
Figure 2.30(a) Chlorine molecule with even distribution of charge; (b) Chlorine molecule with instantaneous dipole showing charge distribution; (c) neighbouring chlorine molecule with an induced dipole, showing charge distribution.
This is because the shift of electrons generating a δ− charge forces the electrons in a nearby bond to be repelled, so a dipole is formed in the nearby molecule, as shown in Figure 2.30c. London dispersion forces are reasonably weak because they are short‐lived, but they are important nevertheless.
Instantaneous dipoles (or dispersion forces) increase with increasing polarisability of the molecule. The more readily polarisable the molecule, the larger the instantaneous dipole and induced dipole. Polarisability is a measure of how easily the charge distribution in an atom or molecule can be distorted by the application of an external electrical field or charge. The greater the number of electrons, the more readily polarisable the molecule is, so instantaneous dipoles increase with increasing molecular mass. In addition, the larger the surface area of a molecule or the larger the area of possible contact between two molecules, the stronger the intermolecular forces.
Instantaneous dipoles are responsible for the very weak intermolecular forces formed between noble gas atoms. The noble gases consist of monatomic molecules: single atoms of neon, argon, etc. Clearly, there are no intramolecular forces here, as there are no bonds, and the intermolecular forces are extremely weak. Instantaneous dipoles are formed by the random movement of electron density from one side of the atom to another, which then induces a dipole in a neighbouring atom, as shown in Figure 2.31. Helium has only two electrons, so the size of the dipole is very small. The heavier noble gases possess more electrons and so have larger dipoles.
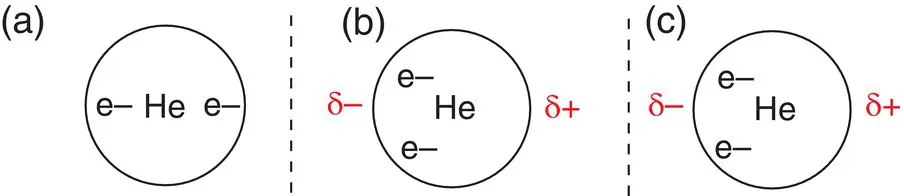
Figure 2.31(a) Helium atom ( Z = 2) showing even distribution of electrons; (b) helium atom with instantaneous dipole due to temporary movement of charge; (c) induced dipole in a neighbouring helium atom.
A monatomic molecule is composed of just one atom. A diatomic molecule such as Cl 2has two atoms. A triatomic molecule such as H 2O has three atoms. A polyatomic atom has several atoms.
The final type of interaction is hydrogen bonding, which is the strongest type of intermolecular force and has about one‐fifth the strength of a typical covalent bond. Hydrogen bonding is actually a special type of dipole–dipole interaction. Hydrogen bonds are formed between molecules that contain a hydrogen atom bonded to a small, strongly electronegative element such as nitrogen, oxygen, or fluorine.
Hydrogen bonding can occur when a hydrogen atom in a molecule is bonded to a strongly electronegative element such as nitrogen, oxygen, or fluorine. A hydrogen bond is formed between the H atom of one molecule and the N, O, or F atom of a neighbouring molecule.
Читать дальше
















