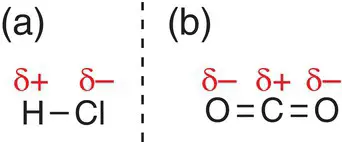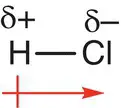However, if one of the atoms (say, atom B) is more electronegative than the other, the electrons will be pulled towards that atom, and the distribution will no longer be evenly spread. Atom B will have a greater share of electrons than atom A, as shown in Figure 2.22. The result of this is that atom B becomes slightly negatively charged compared to atom A. We represent this charge by a δ− sign next to atom B. Because atom B takes up a slight negative charge, atom A must become slightly positively charged to balance the overall charge, and this is represented by the symbol δ+.
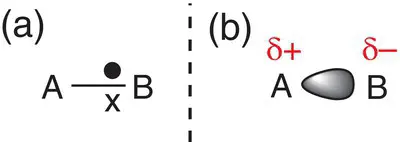
Figure 2.22(a) A pair of electrons shared unevenly between two atoms with different electronegativities; (b) a representation of the cloud of electrons, showing the charge distribution.
The Greek letter δ is pronounced ‘delta’ and in maths and chemistry means ‘a little bit’ or ‘slightly’. So δ− means slightly negative, and δ+ means slightly positively charged.
A bond where there is an uneven distribution of charge, such as that shown in Figure 2.23, is called a polar covalent bond. It occurs between atoms where there is a reasonable difference in electronegativity. Examples of polar covalent bonds are found in hydrogen chloride, HCl, where the chlorine atom is more electronegative than hydrogen; and carbon dioxide, CO 2, where oxygen is more electronegative than carbon.
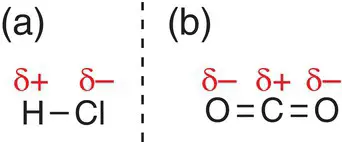
Figure 2.23Polar covalent bonds in (a) hydrogen chloride, HCl and (b) carbon dioxide, CO 2, showing the charge distribution.
If there is a large difference in electronegativity between the atoms in a bond, then the electrons are concentrated on the more electronegative element. In the extreme case, the more electronegative element pulls the electrons completely towards itself and becomes a negatively charged ion (an anion), and the bonding is ionic. This generally occurs when one of the atoms in the bond is a metal and the other a non‐metal. The non‐metal becomes an anion and the metal a cation, and now electrostatic interactions hold the ions together in the lattice.
Pure covalent bonds are formed when the atoms in a bond have the same electronegativity values.
Polar covalent bonds are formed when the atoms in a bond have a small difference in electronegativity values.
Ionic bonds are formed between metals and non‐metals that typically have a large electronegativity difference.
If a molecule has polar bonds, it may be polar overall and have an overall dipole moment, which is important as this can affect how one molecule interacts with another. For example, the molecule H—Cl has a polar bond and an overall dipole moment. This is depicted by an arrow drawn beside the molecule with a line through the flat end. The direction of the arrowhead shows the direction of electron charge in the molecule, and the arrow points from the less electronegative end to the more electronegative end of the molecule, as shown in Figure 2.24.
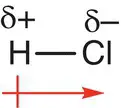
Figure 2.24Hydrogen chloride molecule showing the charge separation and direction of overall dipole moment.
Another example is the molecule chloromethane, CH 3Cl, which has a tetrahedral shape. The C—Cl bond is polar because the chlorine atom is more electronegative than the carbon atom (C has χ = 2.5 and Cl has χ = 3.0). The C—H bonds are usually considered to be non‐polar because the electronegativities of carbon and hydrogen are similar (H has χ = 2.2). The molecule therefore has a dipole moment in the direction of the chlorine atom, as shown in Figure 2.25.
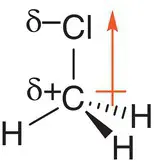
Figure 2.25Chloromethane, CH 3Cl, is a polar molecule.
However, not all molecules with polar bonds have a dipole moment. We have seen that in carbon dioxide, CO 2, both C=O double bonds are polar. However, the molecule is linear, and Figure 2.26shows that the two polar bonds pull charge from the carbon atom in opposite directions to each other. As the size of the dipole is equal in both C=O bonds, there is no net overall dipole moment. You can think of this like two teams in a tug of war pulling against each other. If both teams have exactly the same strength, neither team will win.
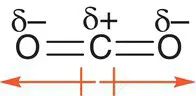
Figure 2.26Carbon dioxide has polar bonds but no overall dipole moment.
Consider the series of molecules based on methane, CH 4, where the hydrogen atoms are replaced in turn to give fluoromethane, CH 3F, difluoromethane, CH 2F 2, trifluoromethane, CHF 3, and tetrafluoromethane, CF 4, shown in Figure 2.27. The molecule fluoromethane, Figure 2.27a, has a permanent dipole moment. It has the same shape as chloromethane, shown in Figure 2.25. The dipole moment is along the C—F bond from the carbon atom to the fluorine atom. In difluoromethane, there are two C—F bonds, and each is polar. The molecule is polar overall as the two dipoles along the bonds add together and give an overall dipole moment oriented between the C—F bonds. Trifluoromethane, CHF 3, also has an overall dipole in the molecule that points in the opposite direction to the remaining C—H bond: Figure 2.27c. Finally, tetrafluoromethane, CF 4, does not have an overall dipole because there is no overall charge separation in the molecule. The polar C—F bonds pull charge in opposite directions and cancel each other out: Figure 2.27d.
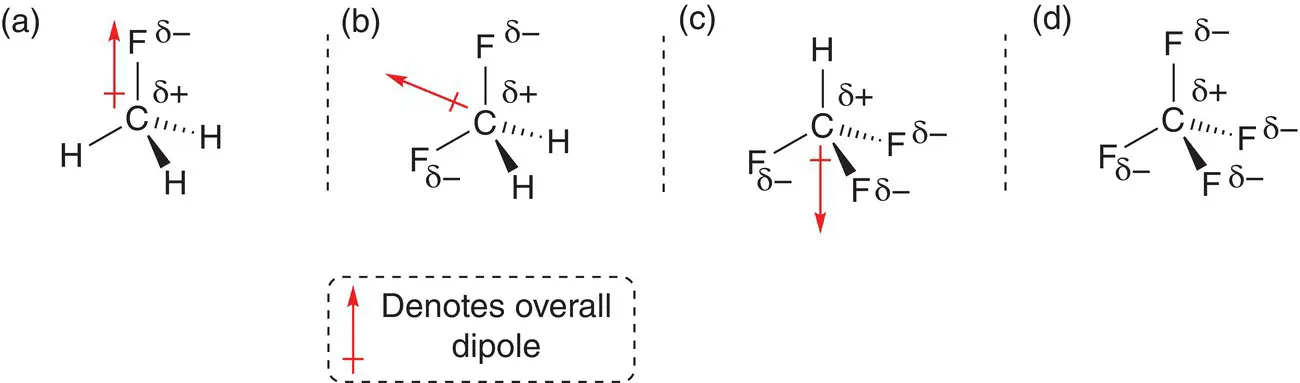
Figure 2.27(a) Overall molecular dipole in fluoromethane; (b) overall molecular dipole in difluoromethane; (c) overall molecular dipole in trifluoromethane; (d) tetrafluoromethane with no overall dipole but polar bonds.
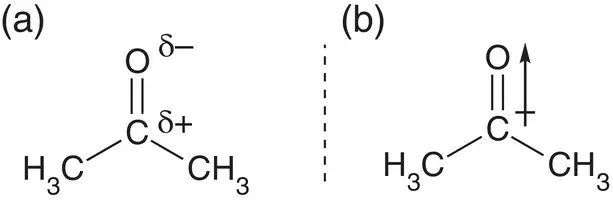
Determine if the molecule propan‐2‐one (acetone), C 3H 6O, is polar and has an overall dipole moment.
Once the structure of acetone has been determined, using a dot‐and‐cross diagram, it is possible to see that it is trigonal planar in shape; there are two single carbon‐to‐carbon bonds, and there is a double bond between oxygen and carbon. The carbon‐carbon and carbon‐hydrogen bonds are not polar, because carbon and hydrogen have similar electronegativities, but the carbon-oxygen bond is polar because oxygen is more electronegative than carbon. This molecule is also polar overall, as charge is pulled towards the oxygen atom, leaving the central carbon atom slightly positively charged.
2.4 Intermolecular forces
The bonds that hold atoms together in a molecule are known generally as intramolecular forces because they are inside a molecule. They are very strong and not easily broken. For example, in the molecule water, H 2O, the covalent O—H bonds are the intramolecular forces. It is very difficult to split water into its component atoms, oxygen and hydrogen. In fact, it takes roughly 51.5 kJ energy to split 1 g of water into its elements, oxygen and hydrogen. If we could easily separate water molecules into gaseous hydrogen and oxygen, we would have a low‐cost way of producing hydrogen as a fuel stock.
Читать дальше