The boron centre is electron deficient because it has only six electrons in its outer shell once it has bonded to the chlorine atoms.
Another molecule that has a trigonal planar arrangement around its central atoms is ethene, C 2H 4. Each carbon atom has four electrons in its outer shell; to attain a complete octet, carbon shares two of these electrons with the single electrons of two hydrogen atoms and shares the remaining two electrons with two electrons from the other bonded carbon atom. Thus, each carbon atom in C 2H 4has two single C—H bonds and one double C=C bond, giving it three distinct areas of electron density. Again, the atoms around each carbon atom spread so that they are approximately 120° apart in a trigonal planar arrangement, as shown in Figure 2.15.
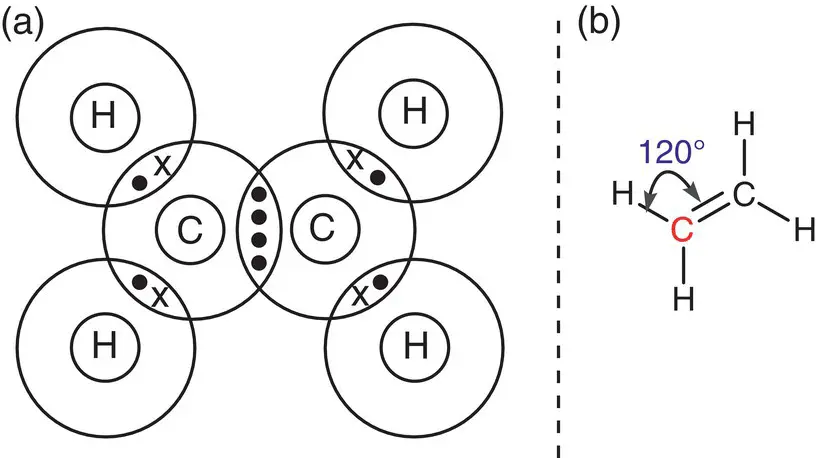
Figure 2.15(a) Bonding in C 2H 4, ethene with trigonal planar centre; (b) showing the bonding angles in ethene.
2.2.4 Four electron centres around the central atom: tetrahedral, pyramidal, and bent molecules
When a central atom has four sets of electron density surrounding it, the areas of electron density are based upon a tetrahedron, but the final shape of the molecule depends on the number of bonded pairs. These shapes are called tetrahedral, pyramidal and, bent (or angular). Figure 2.16shows examples of molecules that adopt each of the three shapes. When there are four single bonds to the central atom, e.g. in methane, the molecule adopts a tetrahedral shape ( Figure 2.16a). When there are three single bonds to the central atom and one lone pair of electrons, e.g. in ammonia, NH 3, the molecule adopts a pyramidal shape ( Figure 2.16b). When there are two bonds and two pairs of electrons around the central atom, e.g. water, H 2O, the molecule adopts a bent or angular shape ( Figure 2.16c). The lone pairs have an impact on the bond angles between the bonded atoms and the central atoms because the lone pairs occupy more space than the bonding pairs. In a symmetrical tetrahedral molecule, the angle between each of the covalent bonds is 109.5°; in a pyramidal compound, the angle is 107°; and in a bent compound, it is 104.5°. This reflects the larger space that the lone pairs occupy, pushing the bonding electrons closer together; see Figure 2.16.
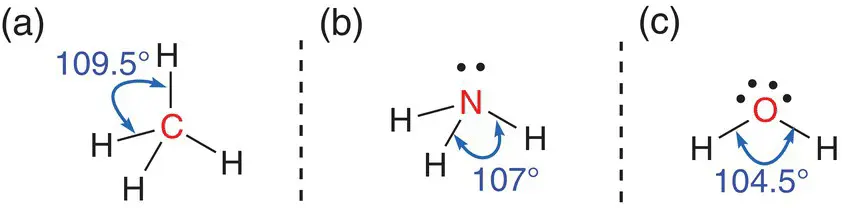
Figure 2.16(a) Bonding angles in a tetrahedral bonding centre; (b) bonding angles in a pyramidal bonding centre; (c) bonding angles in a bent (or angular) bonding centre.
The rule of thumb for deciding the order of interactions between areas of electron density in a molecule is:
Lone pair–lone pair > lone pair–bonded pair > bonded pair–bonded pair
2.2.5 Five electron centres around the central atom: trigonal bipyramidal molecules
If there are five areas of electron density around the central atom, the molecule adopts a trigonal bipyramidal shape, as shown in Figure 2.17for phosphorus pentachloride, PCl 5. The angle in the plane around the phosphorus atom between each of the equatorial bonds is 120°, and the angle between the equatorial bonds and the axial bonds is 90°.
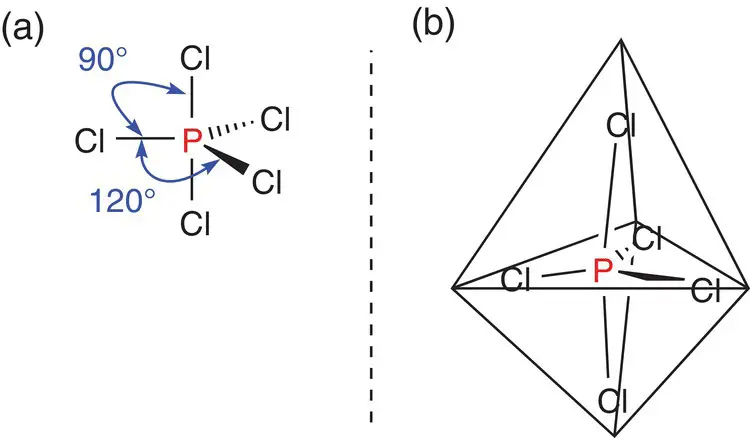
Figure 2.17(a) Bonding angles in a trigonal bipyramidal molecule; (b) phosphorus pentachloride, PCl 5superimposed into a trigonal bipyramid.
You will come across the terms axial and equatorial throughout your studies in chemistry. An axial bond is one that runs vertically up and down along a single axis, whereas an equatorial bond is located horizontally across the page or in the equatorial plane of the molecule.
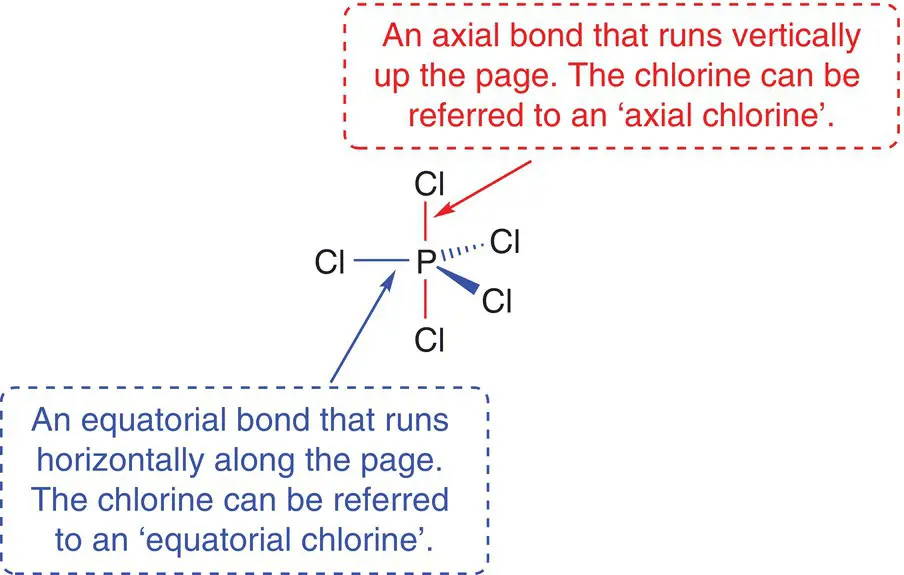
2.2.6 Six electron centres around the central atom: octahedral molecules
When there are six areas of electron density around the central atom or six single covalent bonds, the molecule adopts an octahedral shape. If all six bonded atoms are identical, the angle between each of the bonds is 90°, as this is the furthest apart that all six components can arrange themselves around the central atom. Figure 2.18depicts the shape of the sulfur hexafluoride, SF 6molecule: the six fluorine atoms arrange themselves as far apart as possible around the central sulfur atom. The reason for the name octahedral is that the shape made by the six bonds from a central atom has eight faces, which is an octahedron.
SF 6is used extensively in insulating high‐voltage electrical transmittance cables and switching gear. It is now known to be the most potent greenhouse gas and is banned in all applications apart from in the electrical industry.
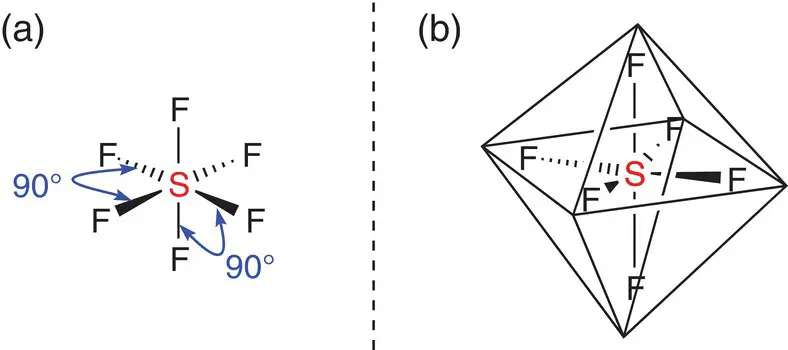
Figure 2.18(a) Bonding angles in an octahedral molecule; (b) sulfur hexafluoride, SF 6superimposed into an octahedron.
We have seen that when considering the arrangement of atoms in a covalently bonded molecule, a molecule can adopt seven basic shapes. These shapes are based on five different distributions of electron density around the central atom and are shown in Figure 2.19. Other combinations of bonded atoms and lone pairs can result in different shapes not included here: for example, the ammonium ion, NH 4 +, has the same shape as a methane, CH 4molecule as the nitrogen atom is surrounded by four N—H bonds which constitute four areas of electron density.
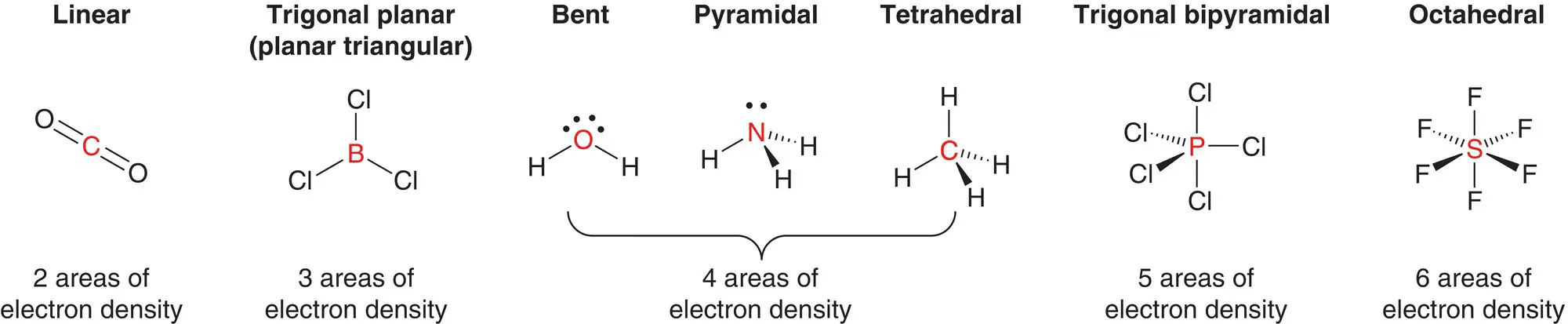
Figure 2.19Common shapes of simple covalent molecules.
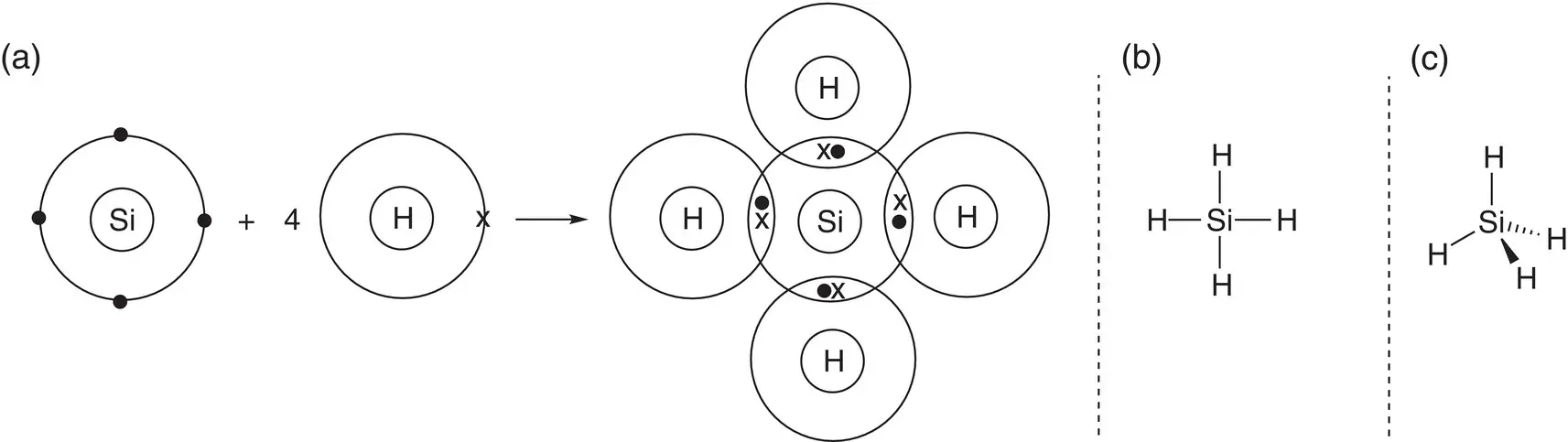
Describe the bonding present in silane, SiH 4, and determine the shape of the molecule.
The first thing to determine is the number of areas of electron density around the central silicon atom. Both silicon and hydrogen are non‐metals; therefore, the bonding is covalent. A dot‐and‐cross diagram can be used to describe the bonding. There are four bonds from the silicon atom to the hydrogen atoms, and no lone pairs; thus there are four areas of electron density, so the shape of silane is tetrahedral. The bonding and shape of the molecule are exactly analogous to methane, CH 4, as both carbon and silicon are in the same group of the periodic table.
Describe the bonding present in hydrogen sulfide, H 2S, and determine the shape of the molecule.
Sulfur, the central atom, is in Group 6 (Group 16), so it has six valence electrons and needs to gain two more to complete the octet. This is achieved by bonding to hydrogen. There are four areas of electron density around sulfur consisting of two single bonds and two lone pairs of electrons. The four areas of electron density arrange themselves as far apart as possible and form a tetrahedral shape. However, because two of these areas of electron density are lone pairs, the actual shape of the molecule appears to be bent (i.e. we can't ‘see’ the lone pairs!). As discussed previously, lone pairs of electrons occupy more space than bonding pairs of electrons, and push the hydrogen atoms more closely together. The H—S—H bond angle is therefore slightly less than 109.5°. So although the arrangement of electron centres is based on a tetrahedron, it is slightly distorted, and hydrogen sulfide is therefore bent (also called v‐shaped or angular ).
Читать дальше



















