A dative covalent bond is formed when both electrons in a covalent bond are provided by the same atom.
Simple molecular covalent bonding and giant covalent bonding
Covalent bonding can lead to two different types of structures. These are called simple covalent (or simple molecular) or giant covalent (or giant molecular) structures. The main difference between simple molecular covalent bonding and giant covalent bonding is that a simple molecule is just that: a small molecule that contains covalent bonds. Examples of molecules that have simple molecular bonding are oxygen (O 2), methane (CH 4), chlorine (Cl 2), and ethanol (C 2H 6O). Giant covalent bonding leads to extended repeated units such as in silicon dioxide (SiO 2) and graphite and diamond (both elemental forms of carbon). A substance that has a giant covalent structure is much more complex and contains an extended array of bonds. This type of bonding does not result in discrete molecules because the bonding is repeated throughout the structure. The structures formed are not molecules as the bonding continues repeatedly in three dimensions. The structures of diamond, silicon dioxide, and graphite are shown in Figure 2.11.
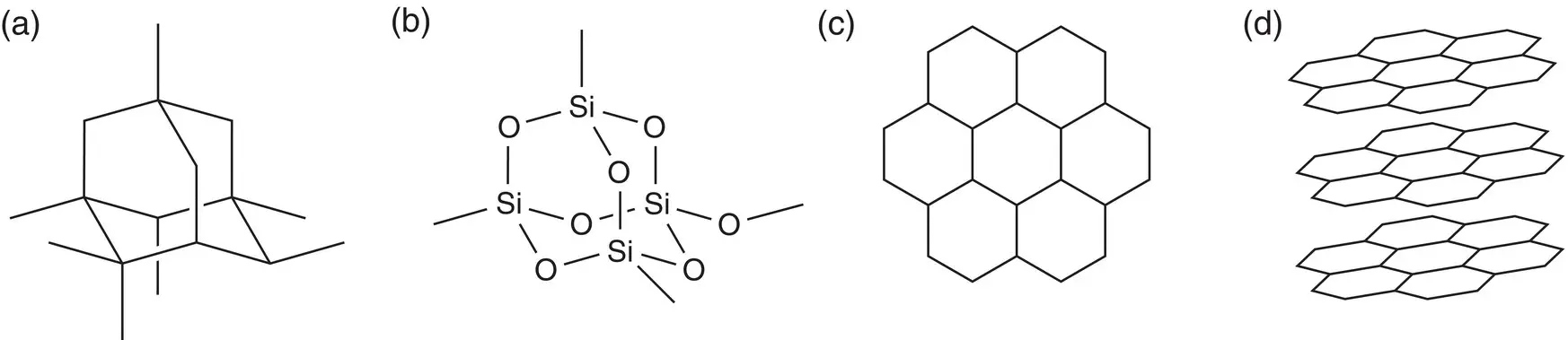
Figure 2.11(a) Bonding in diamond; (b) bonding in silicon dioxide; (c) two‐dimensional bonding in graphite, looking down from above; (d) three‐dimensional bonding in graphite, looking through the layers.
2.2 Valence Shell Electron Pair Repulsion Theory (VSEPR)
The shape of a covalently bonded molecule or ion is determined by the number and arrangement of the pairs of electrons around the central atom. Electron pairs are negatively charged centres and are repelled by neighbouring centres of electron density. The structures formed are most stable when the centres of electron density are as far apart as possible from each other. If you remember this, you will be able to tackle any problems involving the shapes of simple molecules. The process for working out the shape of a molecule is called valence shell electron pair repulsion theory, or VSEPR for short. It is much simpler than the name suggests!
The arrangement of atoms around a bonding centre is entirely based upon the repulsion of areas of negative charge.
In this model, a single area of electron density or charge is defined as:
A lone pair of electrons
A single bond – composed of one pair of electrons
A double bond – composed of two pairs of electrons
A triple bond – composed of three pairs of electrons
2.2.1 How to determine the number of areas of electron density around a central atom
The first stage in working out the shape of a molecule is to determine the number of areas of electron density around the central atom. This can be done by drawing a dot‐and‐cross diagram that shows the number of bonded electrons and the number of lone pairs. The number of areas of electron density is equal to the number of bonded atoms plus the number of lone pairs.
Once we know the number of areas of electron density, there are various fundamental shapes that the molecule can have that minimise the repulsive forces between these negatively charged areas.
In the following section, examples of molecules adopting these fundamental shapes are given so you can see how the overall shape is obtained.
Box 2.1 Depicting the three‐dimensional shape of a molecule
It is difficult on a flat piece of paper or screen to draw a three‐dimensional shape. Most molecules are three‐dimensional with bonds that point in different directions. Chemists use many different ways of showing three‐dimensional structures, but in this book we will use single lines, wedges, and dashed lines. Each type of line means something slightly different and gives important information about the location and direction of the bond in space. The single line means that the bond is in the plane of the page; the wedge means that the bond is coming out of the page towards you; and the dash means that the bond is going into the page, away from you.
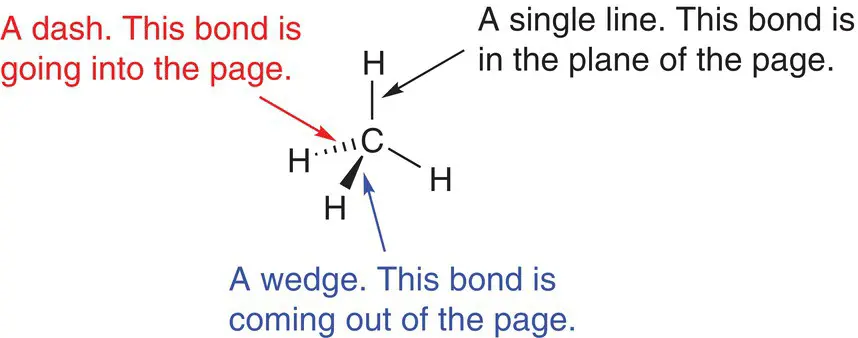
The different representations of bonds in a molecule.
2.2.2 Two electron centres around the central atom: linear molecules
At centres where there are two areas of electron density, the bonded atoms arrange themselves at 180° to each other, generating a linear structure. One of the simplest molecules that adopts this shape is beryllium chloride, BeCl 2. Beryllium has electron configuration 1s 22s 2with just two electrons in its outer shell. Even by sharing electrons with two chlorine atoms, the beryllium atom cannot gain a full outer shell of electrons but shares each of its valence electrons with the unpaired electron of a chlorine atom. In this way, the chlorine atom gains an octet of electrons in its outer shell, and the beryllium atom has just four, as shown in Figure 2.12. Each chlorine atom forms a single bond to the central beryllium atom, and as there are just two centres of electron density, these arrange themselves at 180° to give a linear structure.
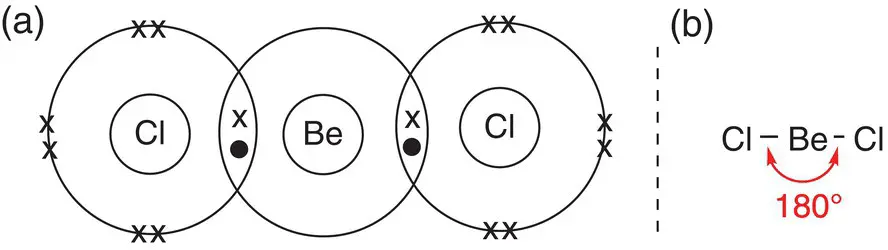
Figure 2.12Dot‐and‐cross diagram for beryllium chloride, BeCl 2showing two centres of electron density and linear shape.
The Be centre is said to be electron deficientbecause it has only four electrons in its outer shell when it has bonded to the chlorine atoms. In the solid state the Be centres accept lone pairs of electrons from chlorine atoms in neighbouring molecules to form long polymer chains. Single molecules of BeCl 2are only present in the gas phase.
Carbon dioxide is another linear molecule. We saw in Section 2.1.4that each central carbon atom has a double bond joining it to an oxygen atom. These two areas of electron density (the two double bonds) spread themselves as far apart as possible in a linear O=C=O arrangement, as shown in Figure 2.13.
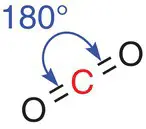
Figure 2.13A linear centre in carbon dioxide, CO 2.
2.2.3 Three electron centres around the central atom: trigonal planar molecules
Centres that have three areas of electron density adopt a trigonal planar structure, with angles of approximately 120° between each of the centres of electron density. A simple molecule with this structure is BCl 3.
Boron has three electrons in its outer shell, and each chlorine atom has seven. Thus boron shares each of its valence electrons with one unpaired electron from chlorine. This gives the boron atom six outer electrons, and the chlorine atoms each have eight outer electrons. The six electrons around boron form three single bonds to the chlorine atoms, thus creating three areas of electron density. These three bonds spread so that they are 120° apart and form a trigonal planar (or triangular planar) shape, as shown in Figure 2.14.
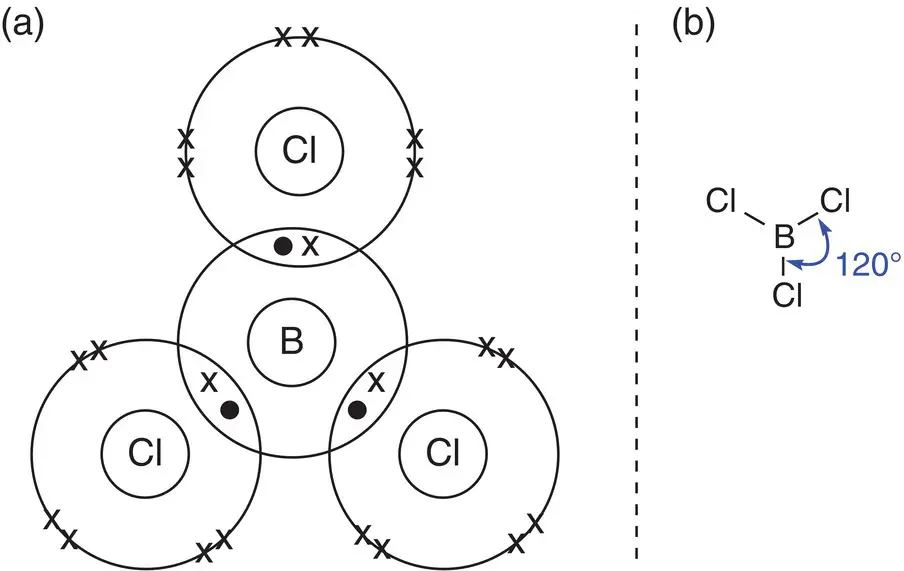
Figure 2.14(a) Bonding in boron trichloride, BCl 3with a trigonal planar centre; (b) showing the bonding angles in boron trichloride, BCl 3.
Читать дальше

















