(1.33) 
The junction potential can introduce errors as big as 50 mV or more [16]. A useful equation for calculating the magnitude of this junction potential was presented by Henderson and is discussed in Appendix C.
Figure 1.14can guide one's thinking about junction potentials in order to determine the effect on the measured cell potential. The reference point is the reference electrode. Going from the potential of the reference electrode through the electrochemical cell to the other side, the path crosses the salt bridge. The argument above indicated that the sample solution will be at a lower potential (more negative) than the reference solution. The path from there, across the indicator electrode interface, starts from this solution potential which appears lower than it would have in the absence of a junction potential. The measured cell potential is indicated as a vector sum for all the transitions in potential between the meter leads. Ideally, the measured voltage is merely a difference between the reference and indicator electrode potentials, but the junction potential must also be included. The diagram indicates that the junction potential would be a negative number in Eq. (1.33). That is, the junction potential leads to an error that makes the measured potential appear more negative (or less positive) in this situation.
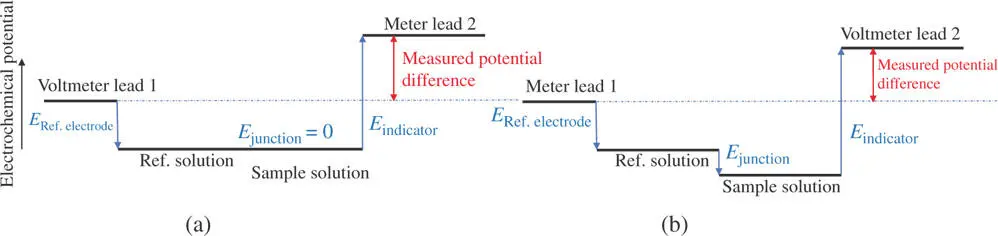
Figure 1.14The measured potential between the indicator and reference electrodes includes all of the transitions in potential in between, represented by vertical arrows in the diagram. If anions are moving across the salt bridge faster than cations in the direction of the reference solution toward the sample solution, then E junctionis negative; the vector for the potential for the junction is downward. (The sample solution potential is lower than that of the reference solution.) In this case, the E indicatoris positive, so it is represented by an upward vector going from the solution potential toward the second meter lead. The cell potential is the value measured between the two voltmeter leads. If the junction potential had the opposite sign, then the vector for the junction would be upward and the measured potential would be larger than shown here. Both situations yield values that are different than the ideal (when E junction= 0). (a) Ideal case and (b) real case; E junction≠ 0.
Of course, one way to avoid this error is to keep the conditions (other than the analyte concentration) of the sample solution and standards as similar as possible. This concern is another reason for using an ionic strength buffer. A further precaution that minimizes the junction potential is to use an electrolyte for both the salt bridge and test solutions in which the cation and anion have very similar diffusion coefficients, such as KCl. Potassium ions have a diffusion coefficient in water of 19.6 × 10 −6cm 2/s which is very similar to that of chloride ions (20.3 × 10 −6cm 2/s).
There is another consideration in setting up a salt bridge. Despite carefully matched diffusion coefficients, another mechanism can give rise to a junction potential. Glass or ceramic material are often used to make porous frits for salt bridges. These materials usually have a net negative charge on their surfaces. For glass with pores on the order of 5–10 nm in diameter, the negative charge can effectively screen anions from crossing the boundary leading to a junction potential on the order of 50 mV even with KCl as a supporting electrolyte (see Figure 1.15). Consequently, salt bridges made from material with larger (micron size) pores are preferable [16]. The trade‐off is a greater leakage of ions from the salt bridge into the sample.
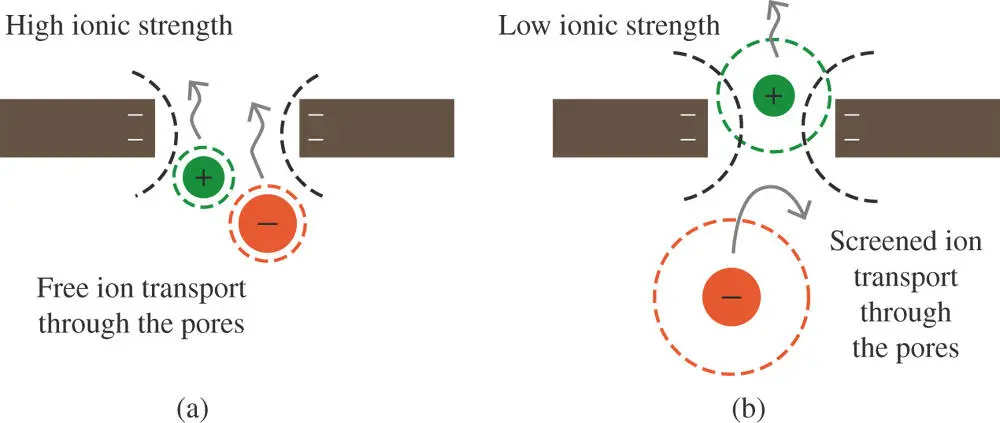
Figure 1.15Microscopic pores in a glass frit used as a salt bridge. The electric field (shown by dashed lines) from negative charges on the glass surface extends a few nanometers into the surrounding solution. This distance is called the Debye length of the electrostatic field. The Debye length is inversely dependent on the ionic strength. (a) At high ionic strength, the anion can pass through the pore without its field interacting with that of the glass. (b) At low ionic strength, the field of the anion and the charge on the glass repel each other decreasing the migration of anions through the pore.
Source: Adapted with permission from Mousavi et al. [16]. Copyright 2016, American Chemical Society.
1 1.1 A bourbon distillery treats its discharge water (that remains behind after the whisky distills off) electrochemically to remove copper by plating it out on an electrode. After 42 days of continuous operation the electrode was replaced. It was weighed before sending it off to a copper recycling service and was found to have recovered 450 g of copper. Calculate the average current during its use assuming that 100% of the current was used in the reduction of Cu2+ to Cu metal.
2 1.2 Consider a carbon electrode with a circular shape and a diameter of 3.0 mm dipping into a 0.1 M NaCl solution with a double layer capacitance of 20 μF/cm2. If the electrode receives a pulse of 1.0 × 1012 electrons, what will the change in voltage be at the electrode solution interface.
3 1.3 The diffusion coefficient for NO3− ion is bigger than the diffusion coefficient for Na+ ion. Consider measuring the electrochemical cell potential in which a salt bridge is used between the reference solution containing 3 M NaNO3 and a sample containing 0.1 M NaNO3 supporting electrolyte solution. Explain whether there will be a junction potential and, if so, whether the cell potential with the junction potential will appear more positive or more negative than without it.
4 1.4 Explain why ohmic loss is more likely to cause a serious error in a voltammetry experiment than it is for a potentiometric experiment.
5 1.5 If 250 μA of current flows when a potentiostat applies −0.351 V to an electrochemical cell with a resistance of 152 Ω, what is the ohmic loss in voltage?
6 1.6 Explain two different mechanisms that could cause the potential of a pH electrode to shift upon the addition of 3 g of KCl to 100 ml of a solution of 0.1 M HCl.
7 1.7 How many moles of electrons would be required to change the voltage on a Pt circular disk electrode with a 2.0 mm diameter from −0.100 to −0.500 V in an electrolyte solution of 0.1 M KCl given the electrode solution capacitance of 24 μF/cm2?
8 1.8 The average thermal energy (or the average kinetic energy) in three dimensions for a molecule is often give as 3/2 kT where k is Boltzmann's constant and T is the absolute temperature. How does the average thermal energy of a molecule at 25 °C compare with 1 eV?
9 1.9 How does the energy of a blue photon at 400 nm compare to 1 eV?
10 1.10 How does the dissociation energy of the carbon–carbon bond in an ethane molecule compare with 1 eV?
1 1 National Research Council (1987). New Horizons in Electrochemical Science and Technology. Washington, DC: The National Academies Press.
2 2 Pilla, A.A. (1974). Bioelectrochem. Bioenerg. 1 (1): 227.
3 3 Xiao, T., Wu, F., Hao, J. et al. (2017). Anal. Chem. 89 (1): 300–313.
4 4 Beni, V., Nilsson, D., Arven, P. et al. (2015). ECS J. Solid State Sci. Technol. 4 (10): S3001–S3005.
Читать дальше















