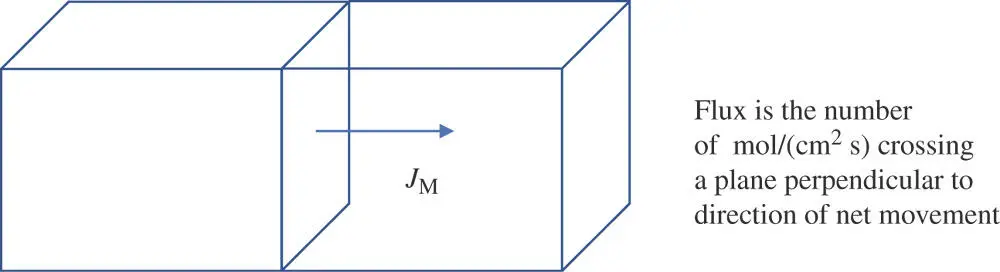
Figure 1.12The definition of flux is the net number of moles of molecules per second crossing a plane of solution with an area of 1 cm 2.
Now, imagine starting the experiment over with a concentration of M at a value of C Mon the left side of the window and a concentration of 0 on the right side of the window. Because there are no molecules on the right side initially, none move through the window from right to left. However, many are going from left to right initially. Consequently, the flux is not zero initially. For ease of discussion, let the direction of left to right represent movement along the x ‐axis in the positive direction. Intuitively, the flux is never going to go from right to left as long as the C Mon the left is greater than it is on the right. It will never be greater on the right. At best, the concentration on the right will reach a value that equals that on the left, but only at equilibrium. Because there are more molecules to consider on the left side, the probability is always greater for a molecule to move from left to right through the window than in the other direction until equilibrium is reached. It also seems intuitive that the bigger the discrepancy in the concentrations on the two sides, the greater the excess in the number of molecules going in one direction. The following equation is a more elegant statement of these ideas. It is known as Fick's first law of diffusion.
(1.30) 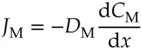
where J Mis the flux of molecule, M, in mol/(cm 2s), C Mis the concentration of M. The proportionality constant, D M, is called the diffusion coefficient in cm 2/s. The gradient in concentration is the driving force for moving molecules across the plane perpendicular to the direction of motion. By convention a decreasing concentration in the x ‐direction is represented by a negative gradient, that is d C /d x < 0 in that case. The negative sign in front of the diffusion coefficient arises in order to make the flux positive for a concentration that decreases in the direction of increasing x . (It is just a convention.) The diffusion coefficient is related to the ion mobility described earlier by the Einstein–Smoluchowski equation [15]:
(1.31) 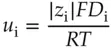
As with ion mobility, the diffusion coefficient decreases with the solvated radius of an ion. The diffusion coefficients for a few ions in water are given in Table 1.2. Note that the diffusion coefficients for OH −and H +are rather large despite the fact that their hydrated radii are also large. That is the case because these ions do not move through water as an individual particle, but rather by exchange of hydrogen ions with molecules of water in their solvation sphere [5].
TABLE 1.2Diffusion coefficients
| Ions in water |
| Ion |
Diffusion coefficienta (cm 2/s) |
Hydrated radiusb (Å) |
| OH − |
52.73 × 10 −6 |
3.5 |
| Na + |
13.34 × 10 −6 |
4.5 |
| K + |
19.57 × 10 −6 |
3 |
| SO 4 2− |
10.65 × 10 −6 |
4 |
| Ca 2+ |
7.92 × 10 −6 |
6 |
| Cl − |
20.32 × 10 −6 |
3 |
| Mg 2+ |
7.06 × 10 −6 |
8 |
| H + |
96.6 × 10 −6 c |
9 |
| NO 3 − |
19.5 × 10 −6 c |
3 |
aFrom Samsonl et al. [18]. Copyright 2003. Used with permission.
bFrom Kielland [19]. Copyright 1937, American Chemical Society. Used with permission.
cCalculated from the electric mobility given by Bakker [15].
In a number of different electrochemical methods discussed in later chapters (see section 5.3), a reaction can change the local concentration of a reactant so that the concentration varies in adjacent regions of solution. This difference in concentration drives a net movement of material in the direction of higher to lower concentration.
1.7 Liquid Junction Potentials
Because accuracy in measuring the voltage in potentiometry and in controlling the applied voltage in voltammetry experiments is so important, it is worth noting at this stage a common mechanism that can introduce errors in the cell potential measurement. Most electrochemical measurements involve the use of salt bridges to isolate reference electrodes from the sample solution. The contact between the sample solution and the salt bridge introduces another opportunity for a separation of charge to develop as shown in Figure 1.13. The mechanism for the build‐up of a potential is driven by a difference in the mobility of the ions. For example, consider a salt bridge that contains 1 M NaCl as an electrolyte contacting a sample solution with 0.1 M NaCl.
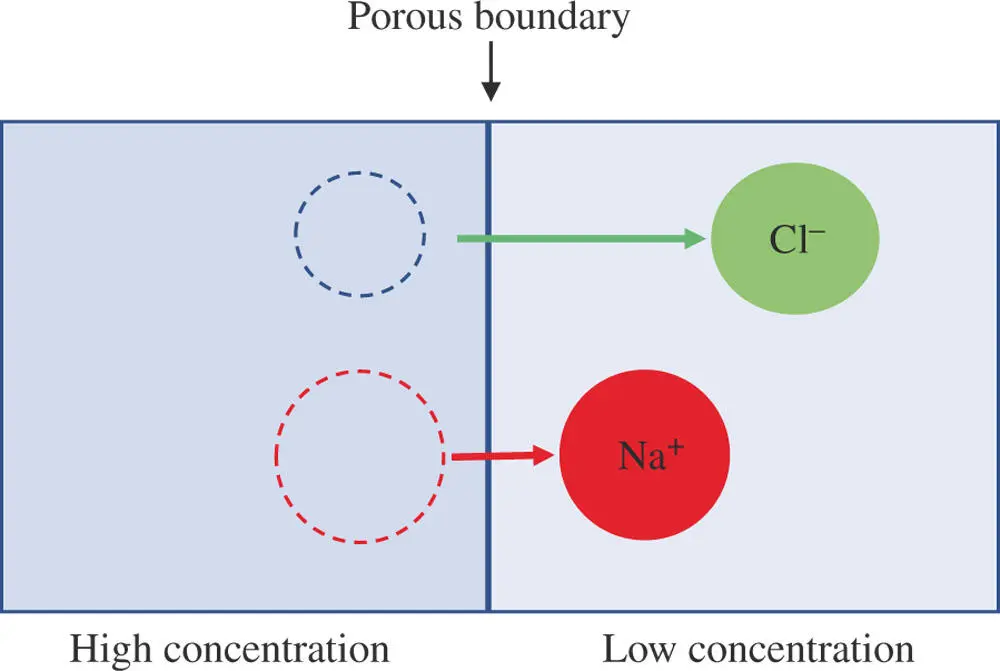
Figure 1.13A salt bridge is a liquid junction between two solutions that allows a small exchange of ions but prevents the two solutions from mixing. A difference in concentration drives ions from the side of high concentration to the low concentration side. Smaller, faster ions move more charge of one sign across the boundary than the other. The result is a net separation of charge at the boundary causing a voltage difference known as a liquid junction potential.
Intuitively, one would expect that a difference in concentration of the ions at the boundary would lead to the movement of ions from the higher concentration toward the medium with the lower concentration. The movement of ions can be modeled mathematically using the Nernst–Planck equation [15]:
(1.32) 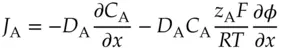
where C Ais the concentration of solute A in mol/cm 3, D Ais the diffusion coefficient of that solute in cm 2/s, and J Ais the flux or moles crossing a plane perpendicular to the direction of movement (here, in the x ‐direction) per square centimeters per second, 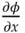 is the potential gradient, F is Faraday's constant, and T is the absolute temperature. (Notice that the flux, in mol/(s cm 2) has the same dimensions as the product of a concentration and a velocity, mol/cm 3× cm/s.) Initially, both the sodium and chloride ions have the same concentration gradient, and there is no electric potential gradient (and the second term on the right is zero). The faster ion is the one with the larger diffusion coefficient. The diffusion coefficients in water of Na +and Cl −are 13.3 × 10 −6and 20.3 × 10 −6cm 2/s, respectively. That suggests that the chloride moves about 50% faster than the sodium ion. Consequently, a negative charge builds up on the lower concentration (sample solution) side and an equal amount of positive charge accumulates on the salt bridge side of the boundary due to the excess of sodium lagging behind. As a result, this process creates a potential energy difference across the boundary. However, the process quickly reaches a steady‐state potential. The potential gradient across the boundary (the second term in Eq. (1.32)) is no longer zero. The potential gradient accelerates the movement of cations and decelerates the movement of anions so that the cation flux and anion flux become equivalent. This mechanism leads to the liquid junction potential. This junction potential is a part of the measured cell potential and it changes with solution conditions.
is the potential gradient, F is Faraday's constant, and T is the absolute temperature. (Notice that the flux, in mol/(s cm 2) has the same dimensions as the product of a concentration and a velocity, mol/cm 3× cm/s.) Initially, both the sodium and chloride ions have the same concentration gradient, and there is no electric potential gradient (and the second term on the right is zero). The faster ion is the one with the larger diffusion coefficient. The diffusion coefficients in water of Na +and Cl −are 13.3 × 10 −6and 20.3 × 10 −6cm 2/s, respectively. That suggests that the chloride moves about 50% faster than the sodium ion. Consequently, a negative charge builds up on the lower concentration (sample solution) side and an equal amount of positive charge accumulates on the salt bridge side of the boundary due to the excess of sodium lagging behind. As a result, this process creates a potential energy difference across the boundary. However, the process quickly reaches a steady‐state potential. The potential gradient across the boundary (the second term in Eq. (1.32)) is no longer zero. The potential gradient accelerates the movement of cations and decelerates the movement of anions so that the cation flux and anion flux become equivalent. This mechanism leads to the liquid junction potential. This junction potential is a part of the measured cell potential and it changes with solution conditions.
Читать дальше


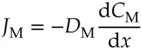
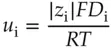

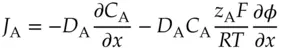
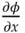 is the potential gradient, F is Faraday's constant, and T is the absolute temperature. (Notice that the flux, in mol/(s cm 2) has the same dimensions as the product of a concentration and a velocity, mol/cm 3× cm/s.) Initially, both the sodium and chloride ions have the same concentration gradient, and there is no electric potential gradient (and the second term on the right is zero). The faster ion is the one with the larger diffusion coefficient. The diffusion coefficients in water of Na +and Cl −are 13.3 × 10 −6and 20.3 × 10 −6cm 2/s, respectively. That suggests that the chloride moves about 50% faster than the sodium ion. Consequently, a negative charge builds up on the lower concentration (sample solution) side and an equal amount of positive charge accumulates on the salt bridge side of the boundary due to the excess of sodium lagging behind. As a result, this process creates a potential energy difference across the boundary. However, the process quickly reaches a steady‐state potential. The potential gradient across the boundary (the second term in Eq. (1.32)) is no longer zero. The potential gradient accelerates the movement of cations and decelerates the movement of anions so that the cation flux and anion flux become equivalent. This mechanism leads to the liquid junction potential. This junction potential is a part of the measured cell potential and it changes with solution conditions.
is the potential gradient, F is Faraday's constant, and T is the absolute temperature. (Notice that the flux, in mol/(s cm 2) has the same dimensions as the product of a concentration and a velocity, mol/cm 3× cm/s.) Initially, both the sodium and chloride ions have the same concentration gradient, and there is no electric potential gradient (and the second term on the right is zero). The faster ion is the one with the larger diffusion coefficient. The diffusion coefficients in water of Na +and Cl −are 13.3 × 10 −6and 20.3 × 10 −6cm 2/s, respectively. That suggests that the chloride moves about 50% faster than the sodium ion. Consequently, a negative charge builds up on the lower concentration (sample solution) side and an equal amount of positive charge accumulates on the salt bridge side of the boundary due to the excess of sodium lagging behind. As a result, this process creates a potential energy difference across the boundary. However, the process quickly reaches a steady‐state potential. The potential gradient across the boundary (the second term in Eq. (1.32)) is no longer zero. The potential gradient accelerates the movement of cations and decelerates the movement of anions so that the cation flux and anion flux become equivalent. This mechanism leads to the liquid junction potential. This junction potential is a part of the measured cell potential and it changes with solution conditions.










