Moving to 3D transition metal (TM), such as Cu, Ti, V, Ni, Cr, and Fe adsorbed at the H site in phosphorene. According to [69], TM adatoms induce a magnetic moment ranging from 1.00 to 4.93 μ B . In particular, the Ti adatom states contribute in the midgap and the conduction band (CB), which reduces the band gap to 0.41 eV in the presence of a magnetic order of 1.87 μ B [70]. A magnetic of 2.00 μ B is observed for Fe adatom systems. In the case of Cr and V adatoms, the spin-down is observed in CB. However, the spin-up of V states dominates the Fermi level and splits into two peaks for Cr adatom. The situation is different for the Ni and Cu adatoms which exhibit no spin-polarization.
1.3.2 Half-Oxided Phosphorene
Similar to graphene oxide, oxygen adsorption on phosphorene can be used efficiently to tune the optoelectronic properties as well as the protective layer of phosphorene. The absorption of a single oxygen atom on phosphorene can occupy numerous positions like interstitial, horizontal, diagonal ones [74]. As mentioned previously, phosphorene is stable at low oxygen concentrations.
1.3.2.1 Electronic Structure
An oxidation with a degree of 50% generates nine possible configurations among which only six are stable. As illustrated in Figure 1.9, the unit cell comprises four P-atoms assigned as P 1, P 2, P 3, and P 4and two O-atoms, namely, O A and O B . Oxygen atoms bind to two P-atoms on the same side (more precisely, they are attached either to the up-side 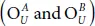 or to the down-side of the surface and
or to the down-side of the surface and 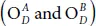 or they are in opposite sides, namely
or they are in opposite sides, namely 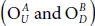 . The index U and D referred to the up and the down side of the P-atoms. For example, to get
. The index U and D referred to the up and the down side of the P-atoms. For example, to get 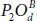 , one should place the OA -atom up on P 1and the OB down on P 2( OB is in the opposite side of OA ) forming a
, one should place the OA -atom up on P 1and the OB down on P 2( OB is in the opposite side of OA ) forming a 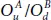 fashion on either side of the plane. The resulting new derivatives can be divided onto three main groups. The first group contains the structures
fashion on either side of the plane. The resulting new derivatives can be divided onto three main groups. The first group contains the structures 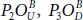 and
and 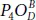 that have only dangling bonds P = O . In three conformers
that have only dangling bonds P = O . In three conformers 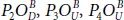 constituting the second group, dangling oxygen motif and bridging bond alternate, respectively. The third class concerns the configurations exhibiting only bridging bonds which are energetically less favorable (see [56] for more details). All the half-oxidized conformers exhibit a high buckling parameter confirming the anisotropic behavior of their properties. It was found, using different methods [24, 56], that half-oxidation of phosphorene (P 4O 2) allows to build a stable material.
constituting the second group, dangling oxygen motif and bridging bond alternate, respectively. The third class concerns the configurations exhibiting only bridging bonds which are energetically less favorable (see [56] for more details). All the half-oxidized conformers exhibit a high buckling parameter confirming the anisotropic behavior of their properties. It was found, using different methods [24, 56], that half-oxidation of phosphorene (P 4O 2) allows to build a stable material.
Half O-functionalization influences significantly the electronic structure of phosphorene mono-layer as depicted in Figure 1.10. The oxidation induces a band gap modulation with the highest value observed in the bridge structures [56]. In all structures the band gap ranges from 0.54 (1.19) to 1.57 (2.88), calculated by GGA (GW) approximation. Moreover, a half O-functionalization tunes the band gap from direct to indirect in all the conformers, except P 2O Dwhich presents a direct band gap.
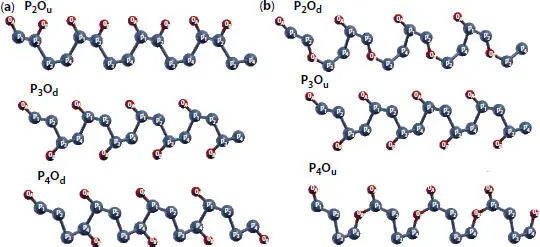
Figure 1.9 Configurations of 50% oxidized phosphorene: (a) dangling structures and (b) bridge structures.
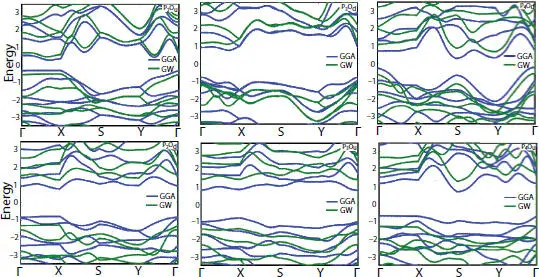
Figure 1.10 GGA and GW band structures of half-oxidized structures.
POs can provide oxygen in its solid-phase to valve regulated Li-O 2batteries. In addition, when the Li atom is absorbed at the surface of the POs, it binds strongly to the O atoms indicating a strong ionic characteristic of the bond between oxygen and lithium [75]. The absolute values of binding energies of the Li atom adsorbed on the PO surface are greater than those of the Li atom on pure phosphorene, MoS 2and graphene [76–78]. POs promise high diffusivity owing to the anisotropy of POs cathode barrier that is reduced by half with respect with the armchair axis for Li diffusion on POs. Besides, Li-PO structures with a number of Li atoms lower than O atoms show stable discharge products for PO cathodes [75].
The optical absorption spectra of half oxidized phosphorene in Figure 1.11show maximum values for dangling structures observed at 1.46, 1.71, and 2.62 eV in 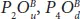 , and
, and  , respectively.
, respectively.
This absorption behavior is required for photodetector with high efficiency. In contrast, pics of spectra describing the bridge structures coincide with the ultraviolet part and the visible light, since the they are located at 1.81, 2.03, and 3.18 eV for 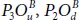 , and
, and 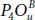 , respectively. One deduces that 50% oxidation is an effective manner to enlarge the absorption range of phosphorene along the light spectrum [20].
, respectively. One deduces that 50% oxidation is an effective manner to enlarge the absorption range of phosphorene along the light spectrum [20].
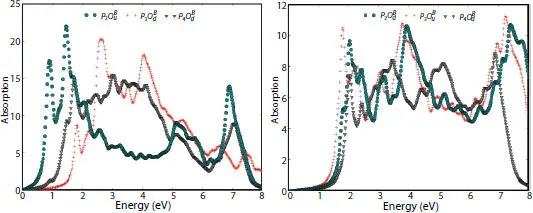
Figure 1.11 Absorption coefficient of dangling structures (on the left) and bridge structures (on the right) of half-oxidized phosphorene sheets obtained using the GW-BSE methods.
Half oxidation is also used to modify the reflectivity of phosphorene as illustrated in Figure 1.11. Indeed, its maximum value in the UV region is around 38%, 50%, and 34% located at 8.21, 8.04, and 7.06 eV in  ,
, 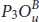 , and
, and 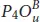 , respectively. In the visible part, these structures show a reflectivity lower than 15% indicating their potential use for transparent electronics. The situation is different for the dangling configurations P 2 OU , P 3 OD , and P 4 OD structures that reflect the visible light with a maximum value of 42%, 38%, and 39% found for the energy 1.48, 2.58, and 1.68 eV, respectively [20].
, respectively. In the visible part, these structures show a reflectivity lower than 15% indicating their potential use for transparent electronics. The situation is different for the dangling configurations P 2 OU , P 3 OD , and P 4 OD structures that reflect the visible light with a maximum value of 42%, 38%, and 39% found for the energy 1.48, 2.58, and 1.68 eV, respectively [20].
Читать дальше

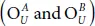 or to the down-side of the surface and
or to the down-side of the surface and 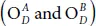 or they are in opposite sides, namely
or they are in opposite sides, namely 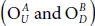 . The index U and D referred to the up and the down side of the P-atoms. For example, to get
. The index U and D referred to the up and the down side of the P-atoms. For example, to get 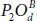 , one should place the OA -atom up on P 1and the OB down on P 2( OB is in the opposite side of OA ) forming a
, one should place the OA -atom up on P 1and the OB down on P 2( OB is in the opposite side of OA ) forming a 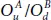 fashion on either side of the plane. The resulting new derivatives can be divided onto three main groups. The first group contains the structures
fashion on either side of the plane. The resulting new derivatives can be divided onto three main groups. The first group contains the structures 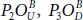 and
and 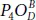 that have only dangling bonds P = O . In three conformers
that have only dangling bonds P = O . In three conformers 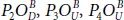 constituting the second group, dangling oxygen motif and bridging bond alternate, respectively. The third class concerns the configurations exhibiting only bridging bonds which are energetically less favorable (see [56] for more details). All the half-oxidized conformers exhibit a high buckling parameter confirming the anisotropic behavior of their properties. It was found, using different methods [24, 56], that half-oxidation of phosphorene (P 4O 2) allows to build a stable material.
constituting the second group, dangling oxygen motif and bridging bond alternate, respectively. The third class concerns the configurations exhibiting only bridging bonds which are energetically less favorable (see [56] for more details). All the half-oxidized conformers exhibit a high buckling parameter confirming the anisotropic behavior of their properties. It was found, using different methods [24, 56], that half-oxidation of phosphorene (P 4O 2) allows to build a stable material.

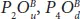 , and
, and  , respectively.
, respectively.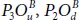 , and
, and 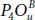 , respectively. One deduces that 50% oxidation is an effective manner to enlarge the absorption range of phosphorene along the light spectrum [20].
, respectively. One deduces that 50% oxidation is an effective manner to enlarge the absorption range of phosphorene along the light spectrum [20].
 ,
, 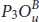 , and
, and 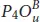 , respectively. In the visible part, these structures show a reflectivity lower than 15% indicating their potential use for transparent electronics. The situation is different for the dangling configurations P 2 OU , P 3 OD , and P 4 OD structures that reflect the visible light with a maximum value of 42%, 38%, and 39% found for the energy 1.48, 2.58, and 1.68 eV, respectively [20].
, respectively. In the visible part, these structures show a reflectivity lower than 15% indicating their potential use for transparent electronics. The situation is different for the dangling configurations P 2 OU , P 3 OD , and P 4 OD structures that reflect the visible light with a maximum value of 42%, 38%, and 39% found for the energy 1.48, 2.58, and 1.68 eV, respectively [20].