
Figure 1.2 Graph of electronic features corresponding to 2D BP. (a) the band structure, (b) represents the total and partial density of states.
1.2.2.2 Optical Properties
The puckered structure of phosphorene attributes it interesting optical properties. Phosphorene absorbs transverse radiation along its AC-axis, while it highly transmitted light along ZZ-axis [19, 52]. The photoluminescence excitation spectroscopy (PLE) measures an optical band gap of 1.31 eV owing to the exciton binding energy as discussed in [19, 49] and measured in [53]. Notice that the theoretical values are larger than the measured amounts because of the increased screening from the dielectric substrate that reduces the quasi-particle band gap and consequently the exciton binding energy [54]. Furthermore, phosphorene can absorbs the visible light since its optical absorption peak is located at 1.6 eV. All these features suggest phosphorene as a promising optoelectronic device for future applications.
The absorption peak can be tunable via strain as displayed in Figure 1.3. For deformed phosphorene, the absorption peak ranges from 0.38 to 2.07 eV under compressive and tensile strain revealing that the material absorbs both infrared and visible light. For the electric field vector E ⊥, the graph displaying the imaginary part of the dielectric function E 2(ω) shows a considerable shift of the first peak towards high energies when including quasi-particle corrections. However, the shape of E 2(ω) spectrum changes considerably when taking into account the electron-hole correlations (BSE) [19]. The exciton binding energy is 0.818 eV for the first active one and 0.66 eV for the first dark one. As displayed in Figures 1.4aand b, the dielectric screening enlarges both the gap and binding energy at ω = 0. Furthermore, a large excitonic wave function distribution is observed for the first bright exciton in Figure 1.4cwhose peak emerges in the IR part as shown in Figure 1.4d. The maximum reflectivity R max (ω) of 38% occurs in the IR range while it did not exceed 22% for the visible light (see Figure 1.4e). The electron energy loss spectra in Figure 1.4freveal that the first plasmon peaks in phosphorene sheet has a height of 11.003 dispersed in the IR range of the spectrum.
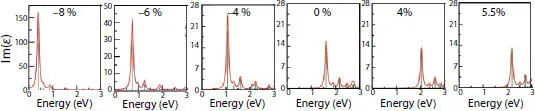
Figure 1.3 Absorption spectra for undeformed monolayer phosphorene with 0% strain and deformed mono-layer under compressive –8%, –6%, and –4% and tensile strain of 4% and 5.5%. The curves are obtained through the GW+BSE method.
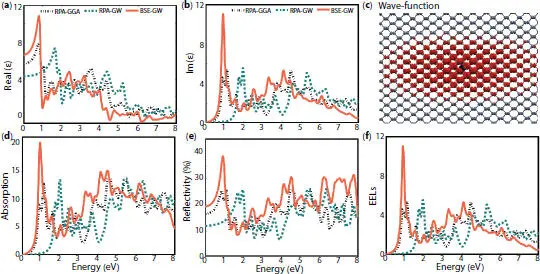
Figure 1.4 (a) and (b) dielectric function, (c) absorption coefficient, (d) reflectivity function, and (e) EELs function, obtained by using three approximations GW-BSE, GW-RPA, and GGA-RPA. (f) Represents the wave function of electron-hole.
In phosphorene multi-layers, the photoluminescence depends significantly on N. The first absorption peak is shifting to lower energies with increasing N. Consequently, the optical absorption coefficient ranges in the interval [0.3–1.2] eV [19], which means that the absorption radiation spectrum include IR and part of visible [52].
1.2.2.3 Elastic Parameters
The elastic properties of phosphorene are also very anisotropic since the Young values in AC-axis (x-direction) is four times lower than the one along the zigzag axis (y-axis), as indicated by the polar diagrams of Υ(θ) illustrated in Figure 1.5a[23, 55]. Notice that the weakest P-P bond strength is the main cause of these small values of the Young parameter [22], compared to 1 TPa and 270 GPa reported for graphene and MoS 2, respectively. Furthermore, the Poisson’s ratio, namely, 0.73 and 0.165 in the AC and ZZ directions, respectively, confirms also the high anisotropy in phosphorene [56]. However, the negative value observed in the small interval [5π, 10/3π] as displayed in Figure 1.5breveals that the material is auxetic. More precisely, for some particular stretch, phosphorene shows a lateral extension instead of longitudinal elongation as it is the case for conventional materials [57, 58]. Besides this, both the shear and compressional acoustic waves propagate more rapidly in the ZZ-axis as it is clearly deduced from the polar plot of the speed of sound in Figure 1.5c. Same anisotropic behavior is found for Debey temperature that is half times lower (see Figure 1.5d).
Besides the strong anisotropy of the elastic parameters, phosphorene exhibits a high elasticity compared with other monolayer materials such silicene, borophene MoS 2, and graphene [59]. Indeed, without breaking phosphorene can withstand large tensile strains along its two possible directions [23]. Whereas, MoS 2, for example, can only withstand deformation up to 13%. At 300 K and under a small magnitude of strain (E), Figure 1.6depicts small ripples on the flat surface of phosphorene. When the compressive E grows, the buckling parameter increases. Interestingly, phosphorene maintains its structural stability in AC-axis at large compressive force up to 80%, but it breaks along the ZZ direction for a 17% deformation, which reveals the super flexible character of this material [60].

Figure 1.5 Polar plot of (a) Young modulus in J/m 2and (b) positive and negative values of Poisson ratio, (c) speed of sound in km/s of pure phosphorene.
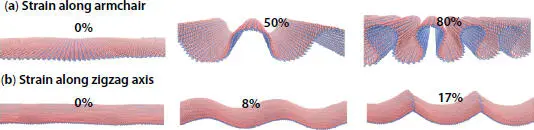
Figure 1.6 Monolayer phosphorene under different values of in-plane compressive strain at 300 K in the two directions.
The remarkable properties and the strong anisotropy observed in phosphorene make it an ideal candidate for photodetectors, modulators, and sensors. Below, we will report some applications.
In 2D materials, the large ratio of surface area to volume renders them promising for gas sensors. In addition, the unique supplementary advantages of phosphorene, like its in-plane anisotropy, structural stability, and high chemical reactivity with molecules, make it highly desirable as a superior gas sensor [61]. Indeed, under gases exposition, phosphorene undergoes multiple modifications [25]. For example, the gas molecules adsorption on phosphorene leads to a reduction or an increase of the resistance that is very required for the markers in sensing applications. Furthermore, phosphorene depends mainly on certain toxic gases because of the high binding strength gas molecules. As a result, the selective behavior of the phosphorene in adsorbing gases influences significantly its transport properties along the two axis directions [61].
1.2.3.2 Battery Applications
According to [16, 17], phosphorene exhibits a specific capacity of 2,596 mAh/g, which is larger than the ones of sulfur and graphene. In addition, the discharge potential in phosphorene, that ranges in the interval [0.4–1.2] V, is smaller than 2.1 V obtained for lithium/sulfur battery [18]. It follows that 2D BP is a potential candidate for new generation of battery [10]. Compared to 2D anode materials, such as graphene and MoS 2, phosphorene exhibits an ultrahigh diffusivity, that is 10 2–10 4times faster. This is owing to its strong anisotropic diffusion barrier that is 0.68 eV, with respect to the small value of 0.08 eV found in the ZZ-axis. Furthermore, phosphorene-based Li battery shows a voltage of 2.9 V which is larger than the one of other 2D layered materials, making this kind of battery of potential use as a rechargeable battery for different electronic and energetic devices.
Читать дальше





