1 ...6 7 8 10 11 12 ...20 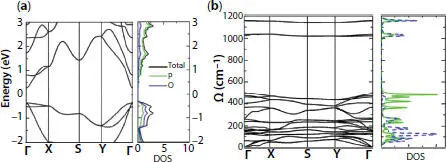
Figure 1.15 Phosphorene oxide (a) band structure and density of states, (b) phonon dispersion curves and density of states.
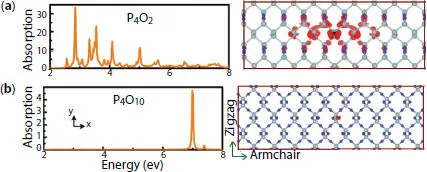
Figure 1.16 Absorption spectrum and exciton wavefunction for the first transition peak for (a) P 4O 2and (b) P 4O 10structures.
When the surface is oxidized, the electrons get transferred forming ions in phosphorene which influences mainly the mechanical response of the material describe by its stiffness against externally applied strains. It results that the oxidation changes the elastic moduli leading to a higher flexible structure [31]. This is also the case for reduced concentrations. Indeed, phosphorene with an oxidation degree of 12.5% can resist to a deformation up to 32% and 35% in AC- and ZZ-axes, respectively, which are higher than that corresponding to pure phosphorene [31]. Moreover, with respect to the pure material, the ideal strength in phosphorene oxide is reduced owing to the enhancement of interatomic distance in the oxide lattice [30, 31] in good agreement with the process of hydrogenating single-layer h-BN [85]. Therefore, the oxidation causes a two times reductions in the value of ideal strength [31].
For a tensile strain varying from −8% to 8%, the band gap in PO increases under compressive strain and decreases with tensile strain, ranging from 0.85 to 0.1 eV for the strain values of [30]. The variation of the gap width results only from the CBM since the VBM is not influenced by the elastic strength. For small tensile strain interval of [−0.006, 0.006], the linear change of polarizations of PO reveals two values of stress piezoelectric responses, namely, e 11= 20.13 10 −10C/m and e 31= 4.06 10 −10C/m that correspond to the piezoelectric coefficients given in [86]. All these results indicate that phosphorene oxide are excellent candidates for potential applications requiring the conversion of energy [86].
1.3.3.3 Thermal Conductivity
Compared with pure phosphorene (P), phosphorene oxide (PO) exhibits a much lower thermal conductivity over the whole temperature range [87]. Indeed, the values of the thermal conductivityfor both P and PO along the armchair axis, namely 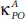 and
and 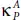 are 2.5 times smaller than
are 2.5 times smaller than 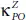 and
and 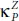 along the zigzag one. At room temperature T = 300 K, the
along the zigzag one. At room temperature T = 300 K, the 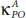 takes the value of 2.42 W/mK, which is very small compared to 65 W/mK reported for pure phosphorene as well as that of other 2D materials such as silicene (26 W/mK) [88] and MoS 2(34.5 W/mK) [89]. Low thermal conductivity renders PO an advantageous novel low dimensional candidate for high-performance thermoelectric materials [87].
takes the value of 2.42 W/mK, which is very small compared to 65 W/mK reported for pure phosphorene as well as that of other 2D materials such as silicene (26 W/mK) [88] and MoS 2(34.5 W/mK) [89]. Low thermal conductivity renders PO an advantageous novel low dimensional candidate for high-performance thermoelectric materials [87].
To highlight the main responsible of such a low lattice thermal conductivity in PO, one should examine the various phonon modes. The puckered structure of PO allows more phonon-phonon scattering of the ZA mode with a contribution of 15% to 17%, while the longitudinal and transverse acoustic modes are the most dominant ones. Furthermore, the lattice thermal conductivity in a material results on the use of different phonon scattering sources. For the case of PO, only the phonon-phonon scattering is considered, since the other sources, such as Umklapp scattering, phonon-electron scattering, impurity effect and boundary effect are so negligible. As shown in Figure 1.17, the anharmonic relaxation times of as a function of frequency indicates that the phonon lifetime corresponding to three acoustic modes (ZA, TA, and LA) and the other higher modes of PO is more lower than that of pure phosphorene. This reduction is mainly owing to dangling bond connecting oxygen atom to its phosphorous neighbor which allow to O not only in the optical P-O vibration, but also vibrate along the in-plane directions together with phosphorous atoms contributing to the acoustic modes. It follows that this contribution is responsible for the acoustic phonon softening which decreases the thermal conductivity of PO [87].
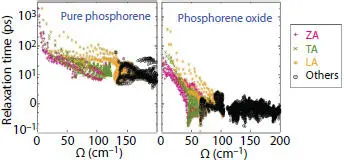
Figure 1.17 Mode-dependent anharmonic phonon relaxation time for acoustic modes.
In this chapter, we have presented an overview of pure phosphorene, its geometric structure, its physical properties, its fabrication methods, and several applications. We have also shown thatowing to its puckered structure and its strong anisotropic electronic, mechanical, magnetic, and optical properties, phosphorene constitutes an ideal candidate for potential applications, including gas sensor, field-effect transistor, and solar cell application. Unstable under atmospheric conditions, we have reported phosphorene oxides and demonstrated how O-functionalization is a promising technique to enhance the features of this novel material.
Lalla Btissam Drissi et al. thank “Académie Hassan II des Sciences et Techniques-Morocco” for financial support.
1. Bridgman, P.W., Two new modifications of phosphorus. Chem. Soc. , 36, 1344–1363, 1914.
2. Aldave, S.H., Yogeesh, M.N., Zhu, W., Kim, J., Sonde, S.S., Nayak, A.P., Akinwande, D., Characterization and sonochemical synthesis of black phosphorus from red phosphorus. 2D Mater. , 3, 1, 014007, 2016.
3. Morita, A., Semiconducting black phosphorus. Appl. Phys. A Mater. Sci. Process , 39, 4, 227, 1986.
4. Liu, Y., Qiu, Z., Carvalho, A., Bao, Y., Xu, H., Tan, S.J., Lu, J., Gate-tunable giant stark effect in few-layer black phosphorus. Nano Lett. , 17, 3, 1970, 2017.
5. Castellanos-Gomez, A., Vicarelli, L., Prada, E., Island, J.O., Narasimha-Acharya, K.L., Blanter, S.I., Zandbergen, H.W., Isolation and characterization of few-layer black phosphorus. 2D Mater. , 1, 2, 025001, 2014.
6. Liu, H., Du, Y., Deng, Y., Peide, D.Y., Semiconducting black phosphorus: Synthesis, transport properties and electronic applications, Chemical Society Reviews. Chem. Soc. Rev. , 44, 9, 2732, 2015.
7. Hu, W. and Yang, J., Defects in phosphorene. J. Phys. Chem. C , 119, 35, 20474–20480, 2015.
8. Elahi, M., Khaliji, K., Tabatabaei, S.M., Pourfath, M., Asgari, R., Modulation of electronic and mechanical properties of phosphorene through strain. Phys. Rev. B , 91, 11, 115412, 2015.
9. Reich, E.S., Phosphorene excites materials scientists. Nat. News , 506, 7486, 19, 2014.
10. Kou, L., Chen, C., Smith, S.C., Phosphorene: Fabrication, properties, and applications. J. Phys. Chem. Lett. , 6, 2794, 2015.
11. Fei, R.X., Faghaninia, A., Soklaski, R., Yan, J.A., Lo, C., Yang, L., Enhanced thermoelectric efficiency via orthogonal electrical and thermal conductances in phosphorene. Nano Lett. , 14, 6393, 2014.
Читать дальше



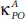 and
and 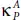 are 2.5 times smaller than
are 2.5 times smaller than 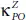 and
and 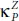 along the zigzag one. At room temperature T = 300 K, the
along the zigzag one. At room temperature T = 300 K, the 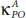 takes the value of 2.42 W/mK, which is very small compared to 65 W/mK reported for pure phosphorene as well as that of other 2D materials such as silicene (26 W/mK) [88] and MoS 2(34.5 W/mK) [89]. Low thermal conductivity renders PO an advantageous novel low dimensional candidate for high-performance thermoelectric materials [87].
takes the value of 2.42 W/mK, which is very small compared to 65 W/mK reported for pure phosphorene as well as that of other 2D materials such as silicene (26 W/mK) [88] and MoS 2(34.5 W/mK) [89]. Low thermal conductivity renders PO an advantageous novel low dimensional candidate for high-performance thermoelectric materials [87].