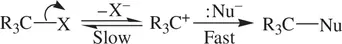Now let us go over briefly some common types of electrophiles and nucleophiles. In Chapters 6and 7on individual types of reactions, we will present more intensive discussions on electrophiles and nucleophiles.
1.9.1 Common Electrophiles
The first type of common electrophiles we will study is carbocations. A carbocation is a species that contains a positively charged trivalent carbon atom possessing a trigonal plannar structure. The simplest carbocation is, in principle, the methyl (CH 3 +) cation ( Fig. 1.16). CH 3 +has not been identified experimentally presumably due to its extremely high instability. It is, however, employed as a prototype from which all the carbocations are derived by replacing one or more hydrogen atoms with different groups. The central carbon atom in CH 3 +is sp 2hybridized, with an empty p orbital holding a positive charge and perpendicular to the trigonal plane defined by the three sp 2orbitals. Each C─H bond is formed by the 1s‐sp 2overlap. When one hydrogen atom in CH 3 +is replaced by an alkyl (R) group, a primary (1°) carbocation RCH 2 +is formed. Replacement of the second and third hydrogen atoms with alkyl groups results in a secondary (2°) carbocation R 2CH +and a tertiary (3°) carbocation R 3C +, respectively ( Fig. 1.16). In general, all types of carbocations are energetic. Thus, they are usually very unstable and highly reactive. Most of them act as strong electrophiles primarily owing to the active empty p orbital in the central carbon atom which has highly strong tendency to accept a pair of electrons from a nucleophile (almost any types of nucleophiles).
The relative stability for different types of carbocations increases in the order of methyl cation CH 3 +, 1° carbocation RCH 2 +, 2° carbocation R 2CH +, and a 3° carbocation R 3C +. This is mainly attributed to the hyperconjugation effectof the C─H bonds in the alkyl groups, which can be well explained using the ethyl CH 3CH 2 +cation (a primary carbocation) ( Fig. 1.17a). In CH 3CH 2 +one of the C─H bonds in the methyl group can overlap in sideway with one lobe of the unhybridized p orbital (hyperconjugation effect) in the primary CH 2carbon. It results in delocalization of the positive charge into the C─H bond domain. In addition, the positively charged CH 2carbon (sp 2hybridized) attracts electrons from the CH 3carbon (sp 3hybridized) through the C─C σ‐bond (inductive effect) also leading to delocalization of the positive charge. The charge delocalization lowers energy of the carbocation. The hyperconjugation effect is further demonstrated in Figure 1.17b using a MO model. The interactions (linear combinations) of a low‐energy C─H bond and a high‐energy p orbital lead to the formation of a bonding MO (with the energy level lower than that of the C─H bonding orbital) and an antibonding MO (with the energy level essentially the same as that of the p orbital). The difference in energy between the C─H bond and the bonding MO represents stabilization of the carbocation (decrease in energy) by a methyl group.
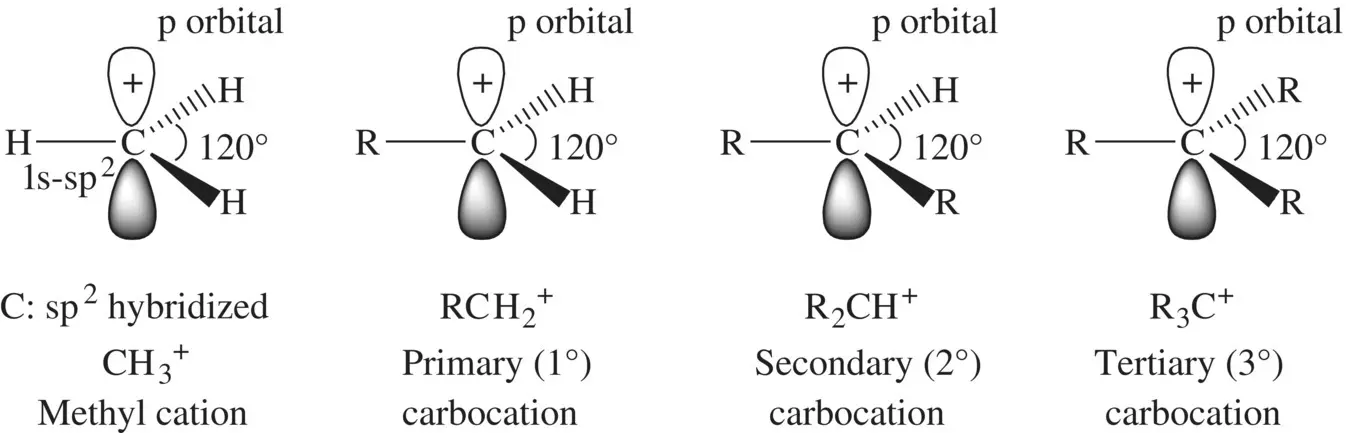
FIGURE 1.16 Structure of different types of carbocations.
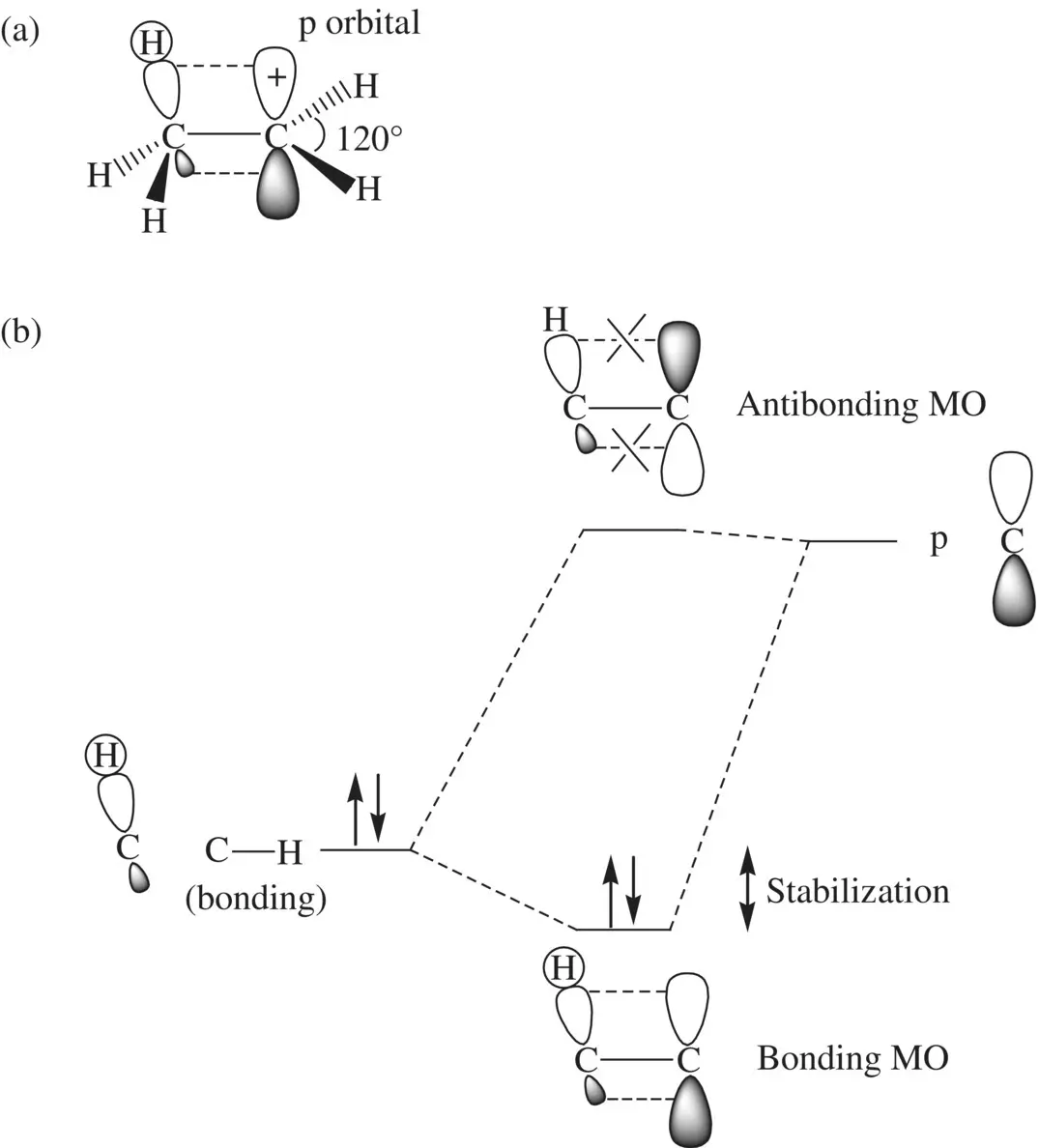
FIGURE 1.17 (a) Overlap of a C─H bond of the methyl group in the ethyl cation (CH 3CH 2 +) with one lobe of the empty p orbital in the carbocation (hyperconjugation) and (b) linear combination of the C─H bonding orbital with the empty p orbital giving rise to formation of bonding and antibonding molecular orbitals.
In (CH 3) 2CH +(a secondary carbocation), two C─H bonds (each from one methyl group) can overlap simultaneously with one lobe of the unhybridized p orbital in the secondary CH carbon. In (CH 3) 3C +(a tertiary carbocation), three C─H bonds (each from one methyl group) can overlap simultaneously with one lobe of the unhybridized p orbital in the tertiary carbon. As a result, the increase in number of the C─H bonds overlapping with the unhybridized p orbital (hyperconjugation effects) makes the positive charge delocalize to greater domains and further lowers the energies of the carbocations. In addition, the inductive effects through the methyl–C +σ bonds are getting more appreciable as the number of methyl groups on the positive carbon increases. This also makes the positive charge delocalize to greater domains and further lowers the energies of the carbocations.
When unsaturated groups such as vinyl and phenyl are attached to a positively charged carbon, the carbocations are greatly stabilized. As a result, the stability of allylic cation (CH 3=CHCH 2 +) and benzylic cation (PhCH 2 +) is even higher than a regular tertiary carbocation such as (CH 3) 3C +. The stabilization is due to large conjugation effectsof the unsaturated groups. In each of the allylic and benzylic cations, the positively charged empty p orbital overlaps in sideways with the π bond of the unsaturated group (conjugation effect), which delocalizes the positive charge to the vinyl or phenyl group and lowers energy of the cation.
Usually, a carbocation is produced by dissociation of a tertiary or secondary haloalkane, which in turn is attacked by a nucleophile resulting in an S N1 reaction ( Reaction 1.63) [1, 6].
(1.63) 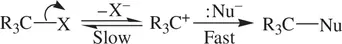
Dissociation of the haloalkane to a carbocation can also be facilitated by a Friedel–Crafts catalyst, such as AlCl 3[1, 6]:

Another common route for generation of a carbocation is the electrophilic addition of a hydrogen halide to an alkene ( Reaction 1.64) [1, 6].
(1.64) 
Once formed, the intermediate carbocation reacts fast with the halide (a nucleophile) to give a haloalkane addition product.
The concentrations of the carbocations in Reactions 1.63and 1.64are extremely low, and their existence cannot be detected by common spectroscopic methods. However, in certain conditions, a secondary or a tertiary carbocation can be stabilized and identified experimentally. For example, 2‐chloromethane dissociates in the medium of antimony pentafluoride (SbF 5, a very strong Lewis acid) to give isopropyl cation (CH 3) 2CH +( Reaction 1.65) [1, 7].
(1.65) 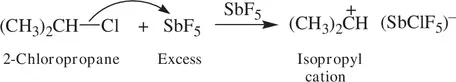
Due to the extremely weak basicity (nucleophilicity) of the (SbClF 5) −anion, the concentration of (CH 3) 2CH +can be enhanced to a sufficient level in SbF 5so that the carbocation has been identified by 1H NMR spectroscopy. The NMR spectrum shows a one‐proton (the CH proton) signal at δ = 13.5 ppm, split into septets by six methyl protons. The –CH 3proton appears at δ = 5.1 ppm. The very low‐field signal of the CH proton (13.5 ppm) is consistent with the strong deshielding effect of the positive charge on the secondary CH carbon [1, 7]. Another NMR‐characterized carbocation is the secondary diphenylmethyl cation Ph 2CH +, which is greatly stabilized by two aromatic groups, and this makes the experimental identification of the carbocation practically possible. In contrast to the secondary and tertiary carbocations, primary carbocations (e.g., the ethyl CH 3CH 2 +ion) are usually not observable experimentally due to their high instability. This is comparable to the above-mentioned situation for the methyl CH 3 +cation.
Читать дальше