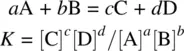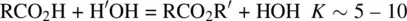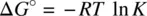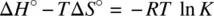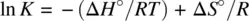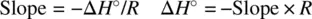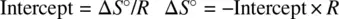Many organic reactions are reversible, namely that the conversion of the reactant to the product is incomplete. When the rate of the forward process is equal to the rate of the backward process for a reversible reaction, the concentrations of all the reactants and products cease to change, and the reaction has reached a dynamic equilibrium.

FIGURE 1.3 The effects of enthalpy and entropy on reversibility of the chemical reactions conducted at constant temperature and pressure.
While the rate constant of a reaction serves as the quantitative measure of how fast the reaction proceeds ( Section 1.4), the equilibrium constant (K)is used as a quantitative measure for the extent of a reversible reaction, which is defined as follows:
(1.50) 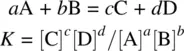
Equation 1.50represents a balanced chemical equation for a reversible reaction (concerted or stepwise). A, B, C, and D represent chemical formulas of different substances (reactants or products). a , b , c , and d represent the corresponding stoichiometric coefficients. [A], [B], [C], and [D] represent the molar concentrations of the species A, B, C, and D, respectively. At a certain given temperature, the K value remains constant, and it is independent of the concentrations of any reactants or products.
The equilibrium constant expression indicates that having one of the reactants (such as B) in excess can increase the percentage of conversion of the other reactant (such as A) to the products. On the other hand, removal of one product (decrease in its concentration) from the reaction system can also increase the percentage of the conversion of the reactants to the products . In the case that one reactant is in very large excess, the conversion of the other reactant (limiting reagent) can be essentially complete (~100%). Therefore, a reversible reaction has been essentially converted to an irreversible reaction.
In organic chemistry, the strategy of using a certain reactant in excess is employed for many reversible reactions to increase the product yields. For example, most of the acid–catalyzed esterification reactions ( Reaction 1.51) have the equilibrium constants K ~ 5–10.
(1.51) 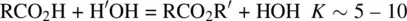
When the carboxylic acid (RCO 2H) and the alcohol (R′OH) are used in 1:1 molar ratio, the conversion of the reactants to the products is 70–75%. If R′OH is used in 10‐folds of excess, the conversion of RCO 2H (limiting reactant) to the ester product will be ~99%. In this case, the reversible reaction has been almost transformed into an irreversible reaction. For some esterification reactions, the essential quantitative conversion of the reactants to the ester product can also be obtained by removal of water from the reaction system once it is formed.
The relationship between the equilibrium constant ( K ) and standard free energy (Δ G °) is formulated as
(1.52) 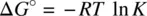
Substituting Equation 1.49for Equation 1.52leads to
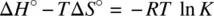
Therefore,
(1.53) 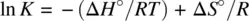
Equation 1.53describes the dependence of the equilibrium constant on temperature. Very often, for an exothermic reaction, the standard enthalpy Δ H° < 0 (−Δ H° / R is positive). The equilibrium constant ( K ) decreases as a function of the temperature ( T ). On the other hand, an endothermic reaction often has Δ H° > 0 (−Δ H° / R is negative). The equilibrium constant ( K ) increases as a function of the temperature ( T ). Therefore, in many cases (exceptions exist) high temperatures facilitate endothermic reactions and low temperatures facilitate exothermic reactions.
Equation 1.53also shows that plot of ln K versus 1/ T defines a straight line, from which both the standard enthalpy (Δ H° ) and standard entropy (Δ S° ) can be obtained from the slope and intercept, respectively, for an unknown reaction.
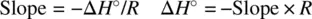
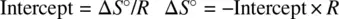
1.6 THE TRANSITION STATE
1.6.1 The Transition State and Activation Energy
The transition state is the structural bridge that links reactants and products for any concerted chemical reactions . In order for the reactant molecules to collide effectively giving the products, they must overcome an energy barrier, which is called activation energy (E a )( Fig. 1.4). This is true for both exergonic (Δ G < 0, Fig. 1.4a) and endergonic (Δ G > 0, Fig. 1.4b) reactions. The activation energy ( E a) is a free energy term, which includes contributions from both enthalpy and entropy. The state in which the reaction system reaches a maximum energy level in the energy profile ( Fig. 1.4a or b) is called transition state. It is also referred to as activated complexin which reorganization of the atoms in the reactant molecules are taking place such that some old bonds are being partially broken, coincident with the partial formation of some new bonds. The transition state (activated complex) is highly energetic and therefore, it is in general very unstable and short‐lived [with the half‐life being in the order of picosecond (10 −12s) for many reactions]. Once formed, it rapidly collapses (dissociates) spontaneously. As a result, the old bonds in the reactants are fully broken, and simultaneously, the new bonds are completely formed, giving the final stable products in the end of the reaction.
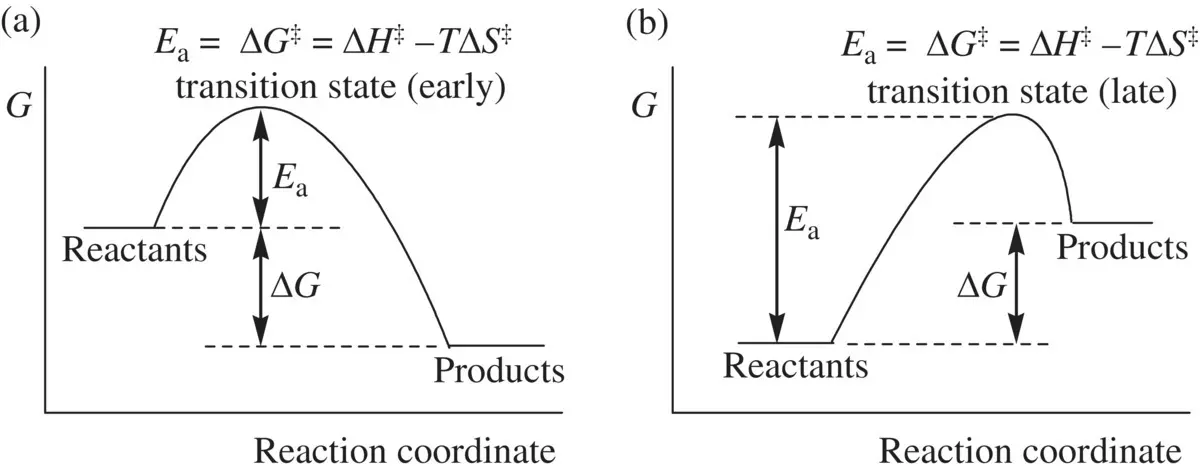
FIGURE 1.4 Early transition state (a) and late transition state (b).
In a concerted reaction ( Fig. 1.4), the activation energy ( E a) is equal to the free energy of the transition state ( E a= Δ G ‡= Δ Η ‡– T Δ S ‡). Δ G ‡, Δ Η ‡, and Δ S ‡are the difference in free energy, enthalpy, and entropy values, respectively, between the transition state and the reactants (Δ G ‡= G TS− G reactants, etc.), and they are referred to as free energy, enthalpy, and entropy of the transition state. In the case that the entropy (Δ S ‡) effect is small and can be neglected, the activation energy is then approximately equal to the activation enthalpy ( E a= Δ Η ‡). In the individual chapters, we will see that this approximation is very often valid and works well for many reactions. The energy level (in the free energy term) that the transition state possesses is the quantitative measure for the stability of the transition state. A fast reaction requires a relatively stable transition state (with a relatively low energy level). A reaction is getting slower as the stability of the transition state decreases (with an increase in its energy level). The quantitative relationship between the rate constant ( k ) of a reaction (concerted) and the activation energy ( E a) is described by the well‐known Arrehnius equation ( Eq. 1.54):
Читать дальше