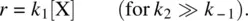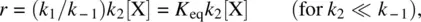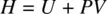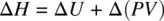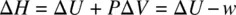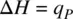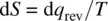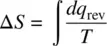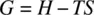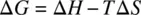1 ...8 9 10 12 13 14 ...23 where k obs= k 1 k 2/( k −1+ k 2) is the observed rate constant.
There are several limiting situations for such a stepwise process [3]. If k 2≫ k −1(the intermediate Y is converted to the product Z much faster than going back to the reactant X), Equation 1.44can be simplified to
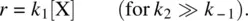
In this case, the first step of Reaction 1.40is the rate‐determining step and actually irreversible.
If k 2≪ k −1(the intermediate Y is converted to the product Z much more slowly than going back to the reactant X), there is a fast preequilibrium between the reactant X and the intermediate Y before the product Z is formed. In this case, the Equation 1.44can be simplified to
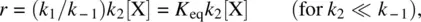
where K eq= ( k 1/ k −1) is the equilibrium constant for the fast preequilibrium between X and Y ( K eq= [Y]/[X]). Therefore, r = k 2[Y], and the second k 2step is the rate‐determining step. Since the fast preequilibrium between X and Y is established prior to the formation of the product, the steady‐state assumption is not necessary if k 2≪ k −1.
If the values of k 2and k −1are comparable, the full steady‐state assumption is needed to establish the rate equation as shown in Equation 1.44.
1.5 THERMODYNAMICS
1.5.1 Enthalpy, Entropy, and Free Energy
Enthalpy ( H ), entropy ( S ), and free energy ( G ) are all thermodynamic state functions. Enthalpy ( H ) is defined as the sum of internal energy ( U ) and the product of pressure ( P ) and volume ( V ), formulated as
(1.45) 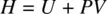
From Equation 1.45, the change in enthalpy (Δ H ) can be calculated as
(1.46) 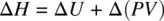
At constant pressure ( P ), Equation 1.46becomes
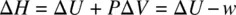
According to the first law of thermodynamics, q P= Δ U − w (heat).
Therefore,
(1.47) 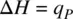
The physical meaning of Equation 1.47is that the enthalpy change in a process (including a chemical reaction) at constant pressure is equal to the heat evolved . Since most of the organic reactions are conducted at constant pressure, the reaction heat can be calculated on the basis of the enthalpy change for the reaction.
Entropy ( S ) is considered as the degree of disorder. In thermodynamics, the infinitesimal change in entropy (d S ) is defined as the reversible heat (d q rev) divided by the absolute temperature ( T ), formulated as
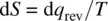
For a finite change in state,
(1.48) 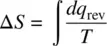
Free energy ( G ) is defined as
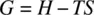
At constant temperature and pressure, the change in free energy (Δ G ) can be calculated as
(1.49) 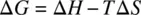
1.5.2 Reversible and Irreversible Reactions
In general, chemical reactions in thermodynamics can be classified as two types, reversible and irreversible reactions. An irreversible reactionis such a reaction that proceeds only in one direction . As a result, the reactant is converted to the product completely (100%) in the end of the reaction.In contrast, a reversible reactionis such a reaction that can proceed to both forward and backward directions. In other words, there is an interconversion between the reactants and the products in a reversible reaction. As a result, all the reactants and the products coexist in the end of the reaction, and the conversion is incomplete.
The reversibility of a chemical reaction can be judged by the second law of thermodynamics. Originally, the second law is stated based on the entropy criterion as follows: A process (including a chemical reaction) is reversible if the universal entropy change (ΔS UNIV ) associated to the process is zero; and a process is irreversible if the universal entropy change (ΔS UNIV ) associated to the process is positive (greater than zero). ΔS UNIV = ΔS + ΔS SURR , the sum of the entropy change in the system (ΔS) and the entropy change in surroundings (ΔS SURR ) .
Since it is difficult to calculate the entropy change in surroundings (Δ S SURR), very often the free energy criterion is used to judge reversibility for any processes that take place at constant temperature and pressure. By employing the free energy change (Δ G ) in a system, the second law can be modified as: At constant temperature and pressure, a process (including a chemical reaction) is irreversible (spontaneous) if the free energy change (ΔG) of the process is negative (ΔG < 0), a process is reversible (at equilibrium) if the free energy change (ΔG) of the process is zero (ΔG = 0), and a process is nonspontaneous if the free energy change (ΔG) of the process is positive (ΔG > 0) . The free energy criterion is widely used in organic chemistry because most of the organic reactions are conducted in open systems at constant temperature and pressure.
According to Equation 1.49, both enthalpy (Δ Η ) and entropy (Δ S ) effects need to be considered when judging reversibility of a reaction using the free energy criterion. The spontaneity of a reaction is favored by a negative enthalpy (Δ Η < 0, exothermic) or a positive entropy (Δ S > 0, increase in disorder), while a positive enthalpy (Δ Η > 0, endothermic) or a negative entropy (Δ S < 0, decrease in disorder) works against a reaction. Variations in temperature ( T ) can change the extent of the entropy effect ( T Δ S ), and therefore they affect the reversibility accordingly. High temperatures favor reactions with a positive entropy change (Δ S > 0), and low temperatures favor reactions with a negative entropy change (Δ S < 0). The effects of enthalpy and entropy on reversibility of the chemical reactions conducted at constant temperature and pressure are summarized in Figure 1.3.
1.5.3 Chemical Equilibrium
Читать дальше