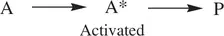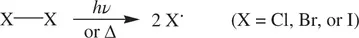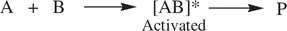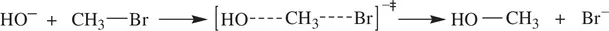Figure 1.1shows reaction profiles for concerted and stepwise reactions using examples of S N2 and S N1 reactions, respectively [1]. In a concerted reaction such as the S N2 reaction of bromomethane (CH 3Br) with hydroxide (OH −) ( Fig. 1.1a), as the reactant molecules start colliding effectively, namely that OH −approaches (attacks) the carbon atom in CH 3Br from the opposite side of the –Br group, formation of a new bond (the O─C bond) and breaking of an old bond (the C─Br bond) occur simultaneously. At the same time, the hydrogen atoms in CH 3Br move gradually from the left side toward the right side. The reaction proceeds via a single transition state (activated complex) in which the old C─Br bond is being partially broken, coincident with the partial formation of a new O─C bond. The hydrogen atoms have moved to the “middle,” forming a roughly trigonal‐planar configuration. The transition state possesses the maximum energy level in the reaction profile. It is short‐lived and highly reactive. As the reaction further progresses, the transition state collapses (dissociates) spontaneously to lead to full breaking of the old C─Br bond in the reactant and concurrent complete formation of the new O─C bond in the product. Simultaneously, the hydrogen atoms move to the right side. The overall process is a one‐step transformation. The extent of the reaction is determined by the difference in free energy (Δ G ) between reactants and products.
In contrast to a concerted reaction, a stepwise reaction proceeds via more than one transition state. It consists of two or more elementary (concerted) steps ( Fig. 1.1b), and distinct reactive intermediate(s) is formed in the course of the reaction [1]. The S N1 reaction of 2‐bromo‐2‐methylpropane (Me 3CBr) in Figure 1.1b demonstrates the general feature of a stepwise reaction. The first step is the dissociation of Me 3CBr to a reactive carbocation Me 3C +intermediate. In the second step, Me 3C +reacts with water to give a tertiary alcohol product (via a hydronium Me 3C–OH 2 +which is very often omitted as its subsequent deprotonation to Me 3C–OH is spontaneous and very rapid, and nearly simultaneous). Each concerted step proceeds via a transition state. The extent of a stepwise reaction is also determined by the difference in free energy (Δ G ) between reactants and products, while the overall reaction rate is dictated by the relative stability of the reactive intermediate(s).
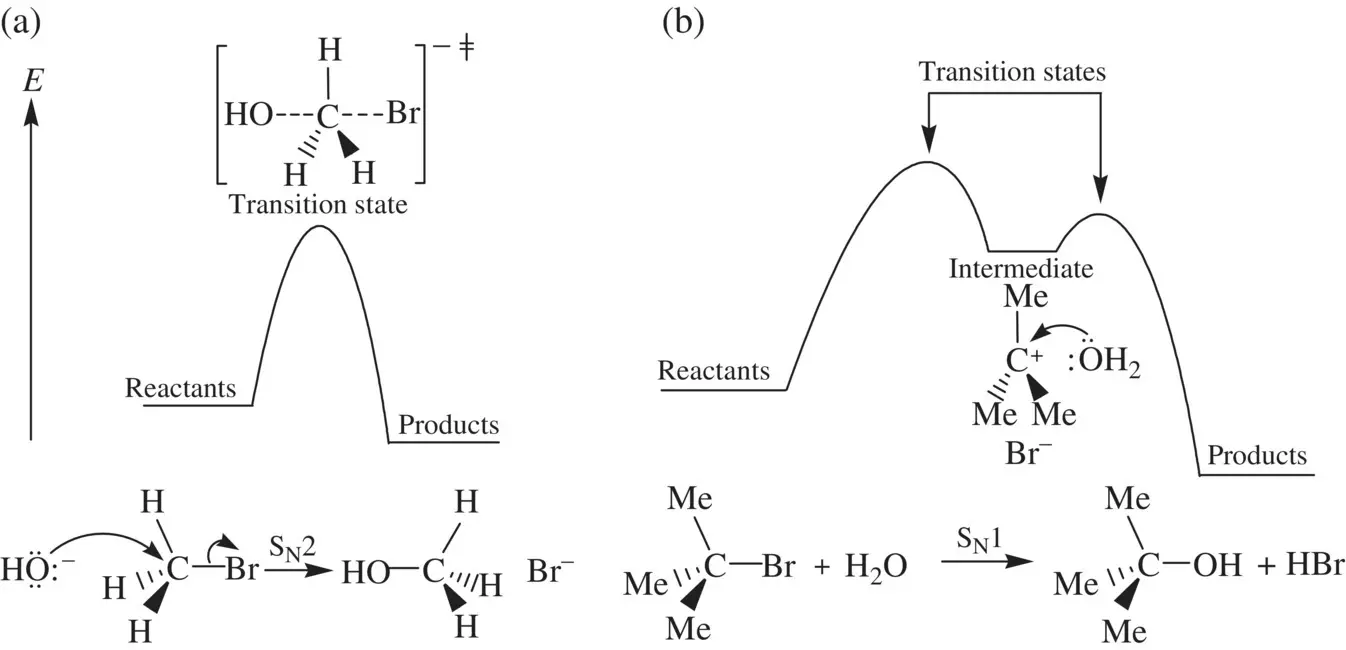
FIGURE 1.1 Reaction profiles for a concerted S N2 reaction (a) and a stepwise S N1 reaction (b).
Whether a chemical reaction is concerted or stepwise is determined by geometry and electronic structure of reactant and product molecules and reaction conditions. In many cases, the mechanism is predictable. In the individual chapters of this book, we will study the various types of concerted and stepwise reactions and the specific conditions which make them happen.
The number of molecules contained in the transition state of a concerted reaction is called molecularityof the reaction. Clearly, the molecularity is determined by the number of reactant molecules that are involved in the mechanism (microscopic step) of a concerted reaction.
1.3.1 Unimolecular Reactions
The microscopic steps of many concerted chemical reactions only involve a single reactant molecule. Such a concerted reaction whose mechanism only involves one reactant molecule is defined as a unimolecular reaction. It is generalized as follows ( Eq. 1.1):
(1.1) 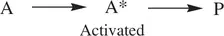
A, A*, and P represent a reactant molecule, an activated reactant molecule (transition state), and a product molecule, respectively. In a unimolecular reaction, a reactant molecule can possibly gain energy and then is activated by several means, including collision of the reactant molecule with a solvent molecule or with the wall of the reactor, thermally induced vibration of the reactant molecule, and photochemical excitation of the reactant molecule. After the molecule is activated, some simultaneous bond‐breaking and bond‐formation processes will take place in A* intramolecularly. As a result, the reactant molecule A will be transformed into one or more product molecules. Common examples of unimolecular reactions are thermal or photochemical dissociation of a halogen molecule ( Reaction 1.2) and intramolecular ring‐opening and ring‐closure reactions ( Reaction 1.3).
(1.2) 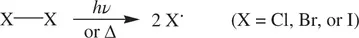
(1.3) 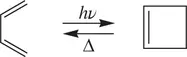
1.3.2 Bimolecular Reactions
For most of concerted chemical reactions, their microscopic steps (mechanisms) involve effective collisions between two reactant molecules. Such a concerted reaction that is effected by collision of two reactant molecules to directly lead to the formation of products is defined as a bimolecular reaction. A bimolecular reaction can be effected by collision of two molecules of a same compound ( Eq. 1.4) or two molecules of different compounds ( Eq. 1.5).
(1.4) 
(1.5) 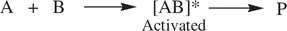
As a result, simultaneous bond‐breaking and bond‐formation take place within the activated complex (transition state) A 2* or [AB]*. This leads to spontaneous collapse of the activated complex (transition state) giving product molecules. Common examples of bimolecular reactions are thermal decomposition of hydrogen iodide (HI) to elemental iodine (I 2) and hydrogen (H 2) ( Reaction 1.6), the S N2 reaction of hydroxide with bromomethane ( Reaction 1.7), and Diels–Alder reaction of 1,3‐butadiene and ethylene ( Reaction 1.8).
(1.6) 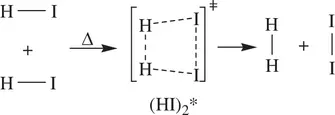
(1.7) 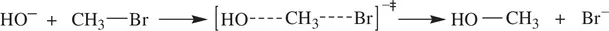
(1.8) 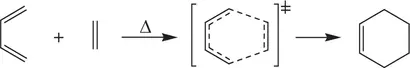
Almost all the concerted processes in organic reactions are either unimolecular or bimolecular steps.
1.4 KINETICS
1.4.1 Rate‐Laws for Elementary (Concerted) Reactions
For elementary reactions, the reaction orders are consistent with the molecularity. A unimolecular reaction is the first‐order in the reactant and a bimolecular reaction has a second‐order rate law.
Читать дальше