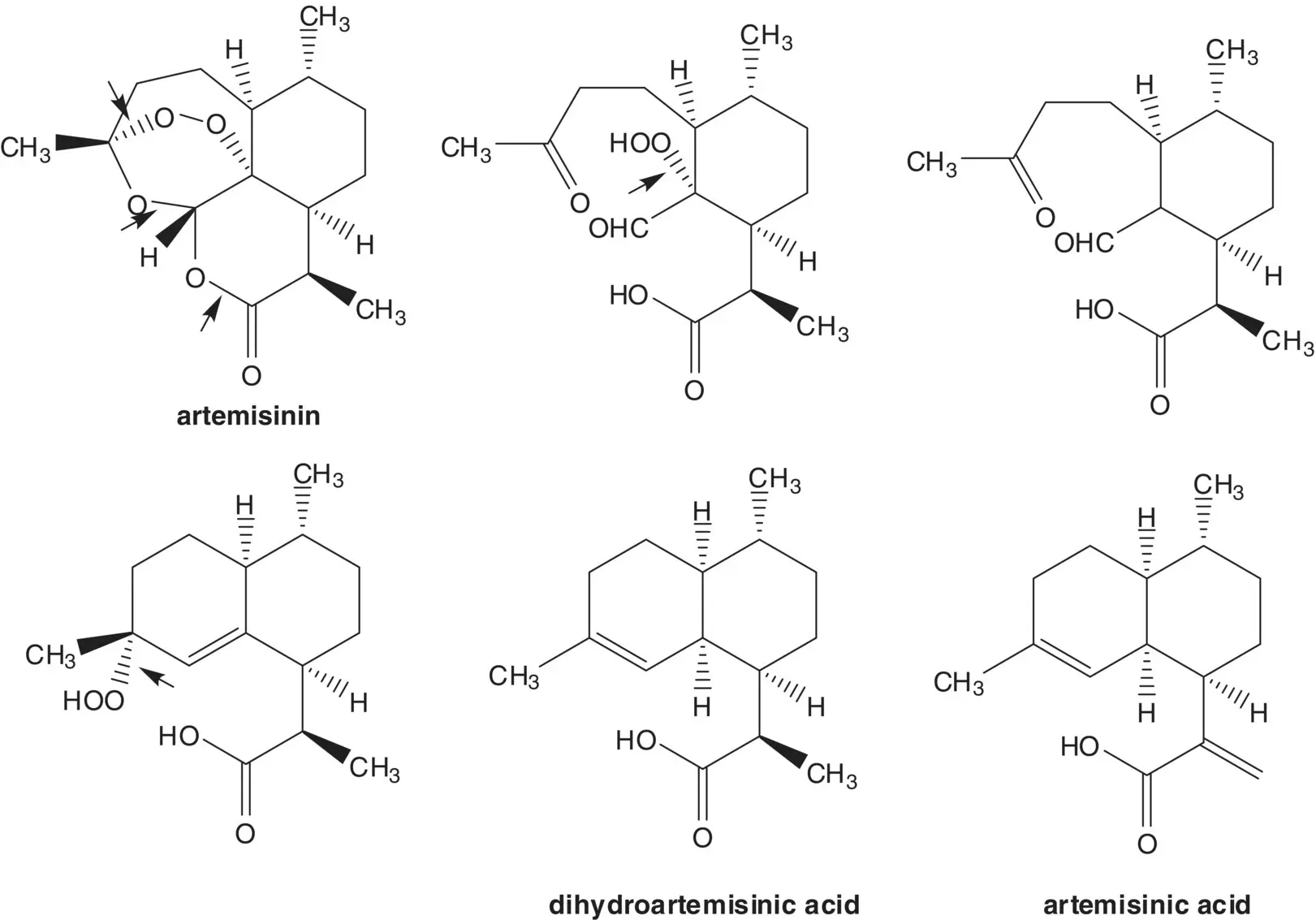
β‐Artemether and α‐artesunate are both formed from dihydroartemisinin. Draw the structures of a retrosynthetic analysis of β‐artesunate.
Vitamins and Minerals
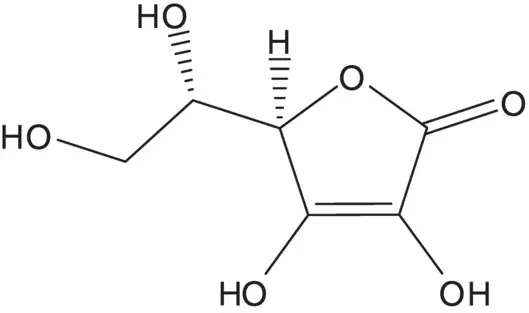
A single‐enantiomer molecule with multiple chiral carbons is often formed by modification of a natural product which has most or all of the chiral carbons already in place.
Discussion.Ascorbic acid (vitamin C) is semisynthetic. Ascorbic acid is formed from 2‐keto‐L‐gulonic acid. 2‐Keto‐L‐gulonic acid is produced by fermentation from L‐sorbose. L‐Sorbose is produced by fermentation from D‐sorbitol.
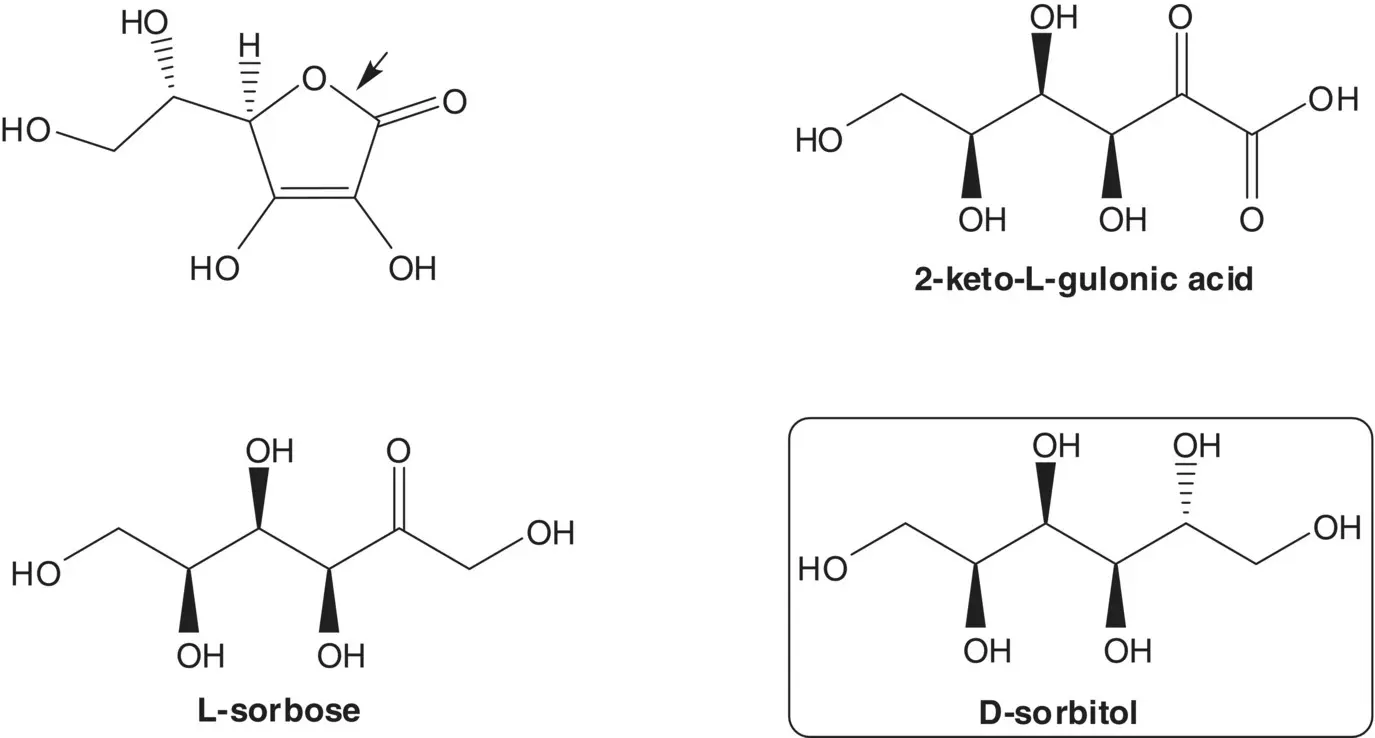
Draw the structures of the retrosynthetic analysis of an alternative non‐fermentation route to 2‐keto‐L‐gulonic acid from L‐sorbose.
Anti‐Infective Medicines/Antiviral Medicines/Antiretrovirals/Protease Inhibitors
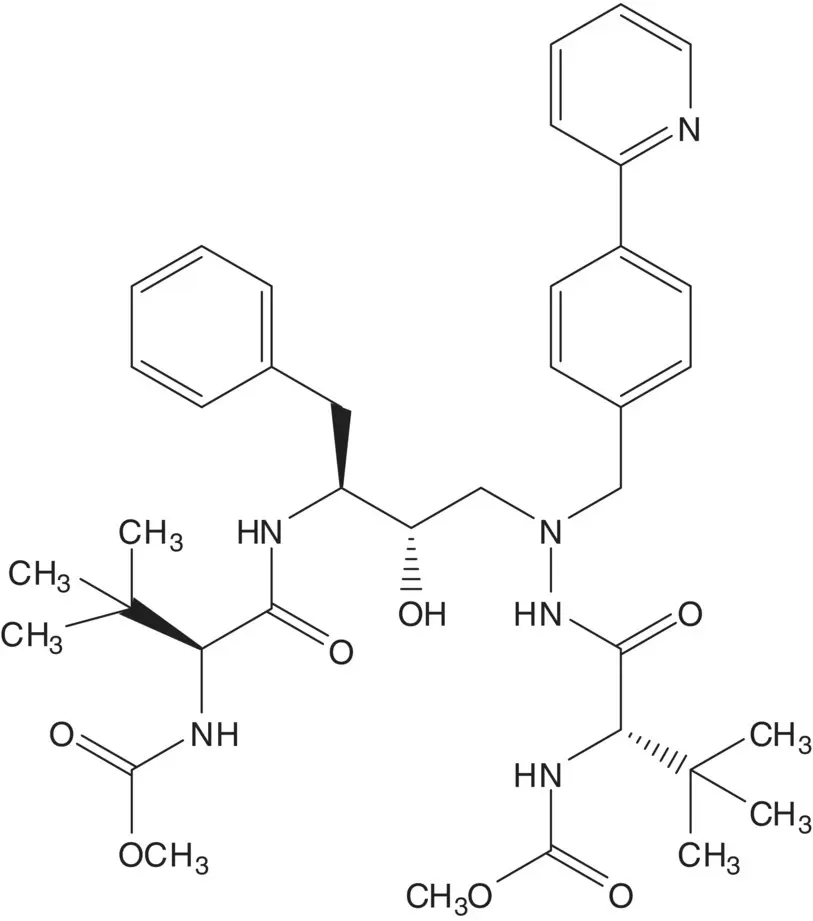
A β‐amino alcohol with a primary β‐C is often formed by ring‐opening of an epoxide by an amine.
Discussion.Two amides are formed with expensive N ‐(methoxycarbonyl)‐L‐tertleucine (Moc‐L‐tertleucine) in the final step. The amine and hydrazine needed to form the amides are released by hydrolysis of tert ‐butoxycarbonyl (Boc) protecting groups. A key C─N bond near the center of the molecule is formed by ring‐opening of an epoxide with a Boc‐protected hydrazine.
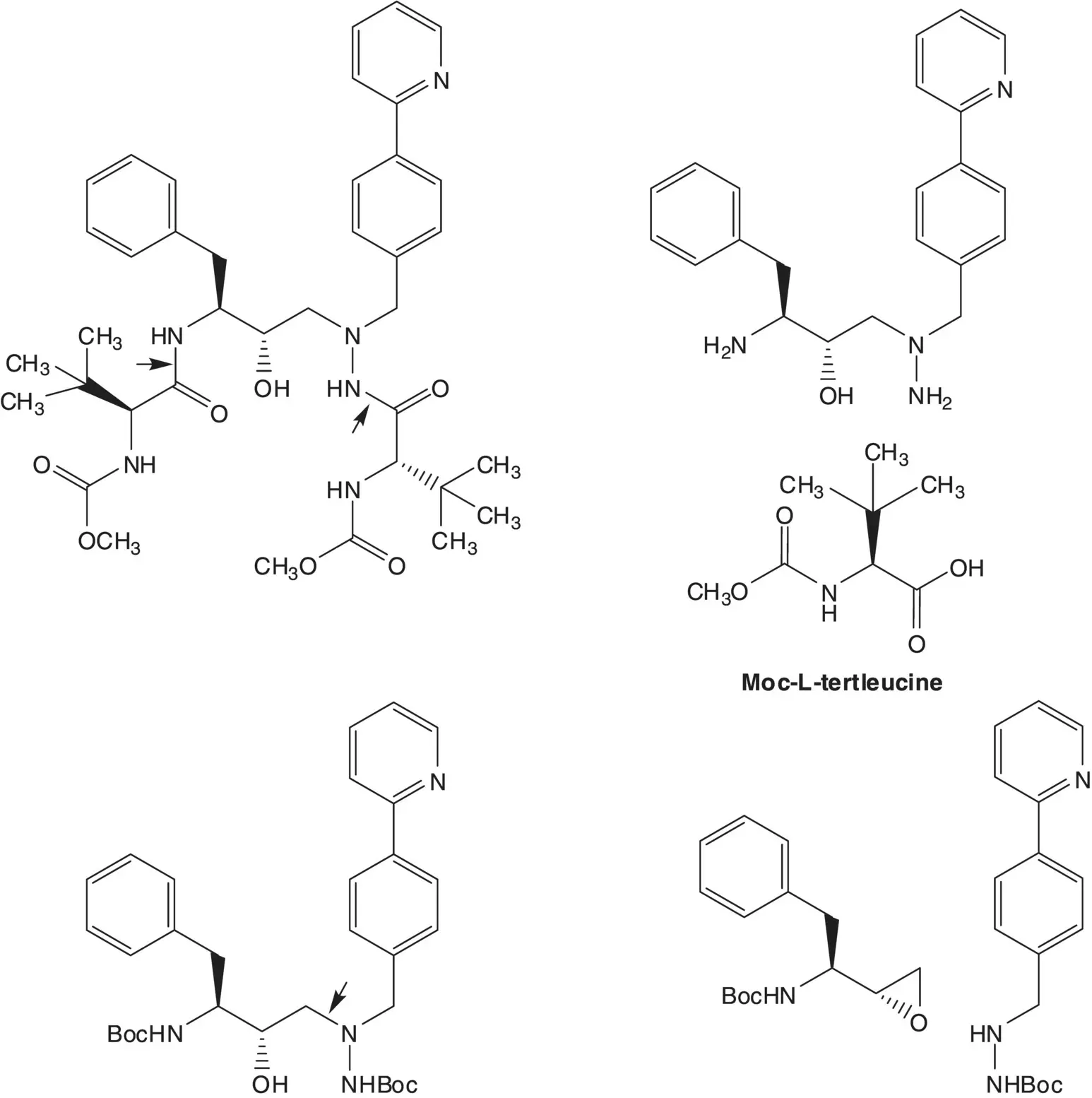
Moc‐L‐Tertleucine is formed from L‐tertleucine and methyl chloroformate. L‐Tertleucine is formed by an enzyme‐mediated reductive amination of trimethylpyruvic acid. The pyruvic acid is formed by oxidation of the α‐hydroxyacid. The α‐hydroxyacid is formed from 1,1‐dichloropinacolone by rearrangement and hydrolysis. 1,1‐Dichloropinacolone is formed by α–chlorination of pinacolone.
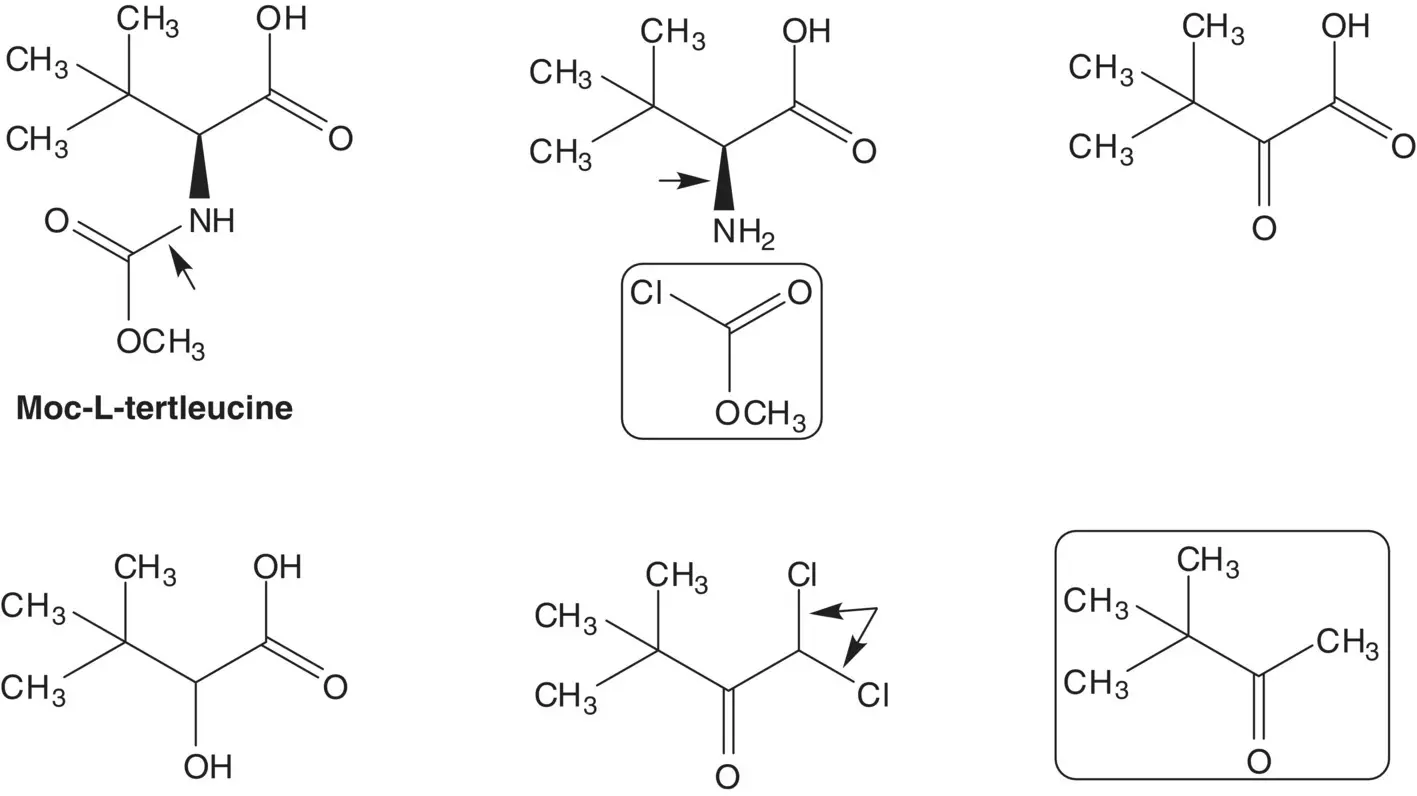
The epoxide is formed from the chlorohydrin by nucleophilic displacement of chloride by oxygen. The chlorohydrin is formed by reduction of the α‐chloroketone, N ‐( tert ‐butoxycarbonyl)‐3( S )‐amino‐1‐chloro‐4‐phenyl‐2‐butanone. The α‐chloroketone is formed by reduction of the α,α‐dichloroketone. The α,α‐dichloroketone is formed from N ‐( tert ‐butoxycarbonyl)‐L‐phenylalanine methyl ester and dichloromethane.
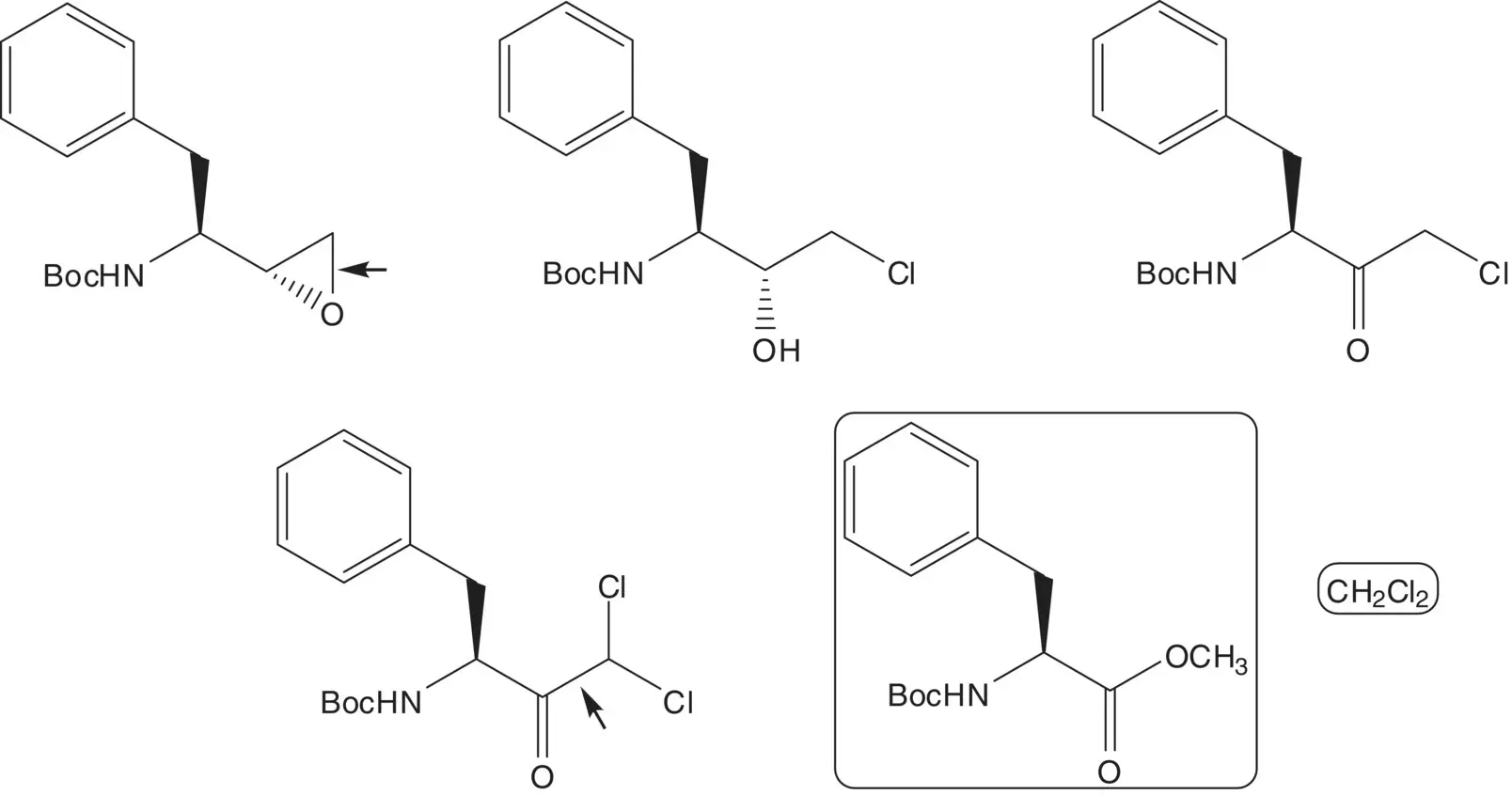
The Boc‐protected hydrazine is formed by reduction of the hydrazone. The hydrazone is formed by reaction of the aldehyde with Boc‐hydrazine. The aldehyde, 4‐(2‐pyridyl)benzaldehyde, is formed from 2‐bromopyridine and 4‐formylbenzeneboronic acid ( Suzuki–Miyaura Coupling). The boronic acid is formed from 4‐chlorobenzaldehyde and trimethylborate via an acetal.
In the route presented, the key C─N bond is formed by ring‐opening of an epoxide. Draw the structures of the retrosynthetic analysis of an alternative route to atazanavir which forms the same key C─N bond but does not involve an epoxide intermediate. List the pros and cons for both routes and select one route as the preferred route.
Muscle Relaxants (Peripherally Acting) and Cholinesterase Inhibitors
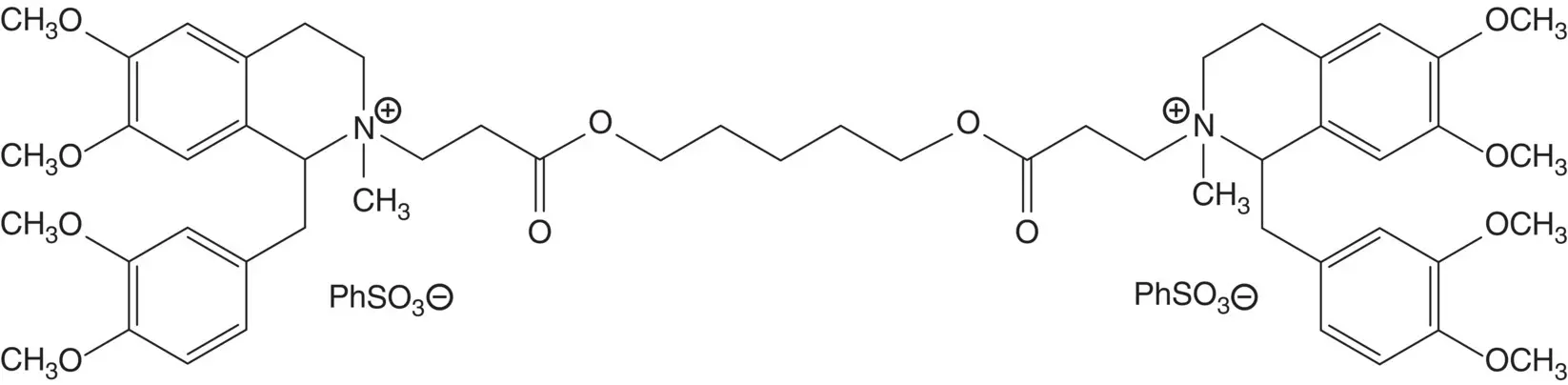
A tertiary amine is often formed by alkylation of a secondary amine.
Discussion.Atracurium besylate is produced as a mixture of 10 stereoisomers (label the four chiral centers in atracurium besylate). The discussion will be limited to routes to one of the 10 stereoisomers, the (1 R ‐ cis , 1′ R ‐ cis )‐isomer known as cisatracurium besylate.
In one preferred route, the quaternary salts are formed in the final step by reaction of the tertiary amines with methyl benzenesulfonate (draw structures for two side products formed in this reaction. How is cisatracurium besylate separated from the side products?) The tertiary amines are formed by conjugate addition of ( R )‐tetrahydropapaverine to the acrylate.
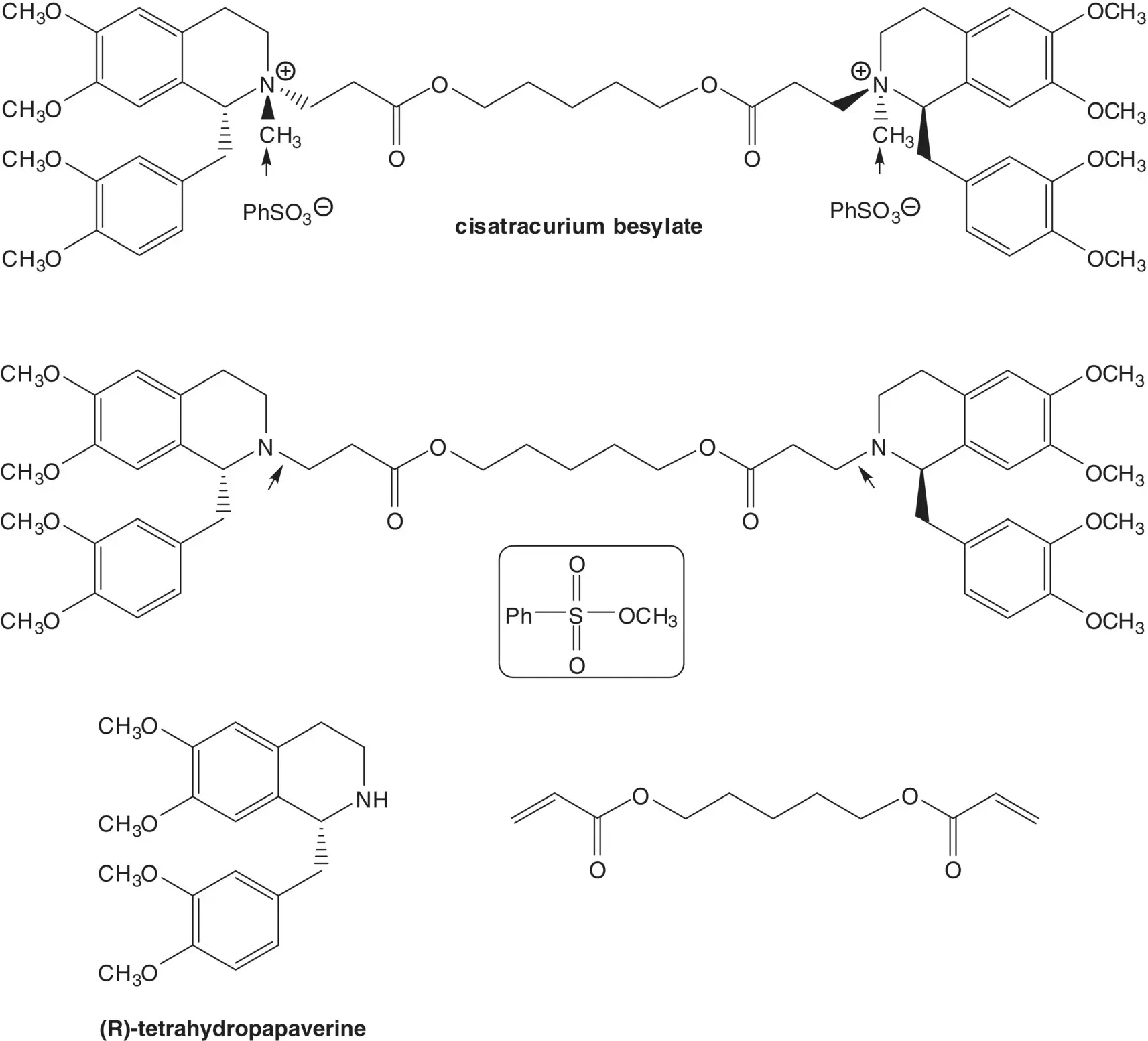
( R )‐Tetrahydropapaverine is formed by resolution of tetrahydropapaverine. Tetrahydropapaverine is formed by reduction of dihydropapaverine. The dihydroisoquinoline ring of dihydropapaverine is formed from the amide ( Bischler–Napieralski Reaction). The amide is formed from 3,4‐dimethoxyphenylacetic acid and 3,4‐dimethoxyphenethylamine.
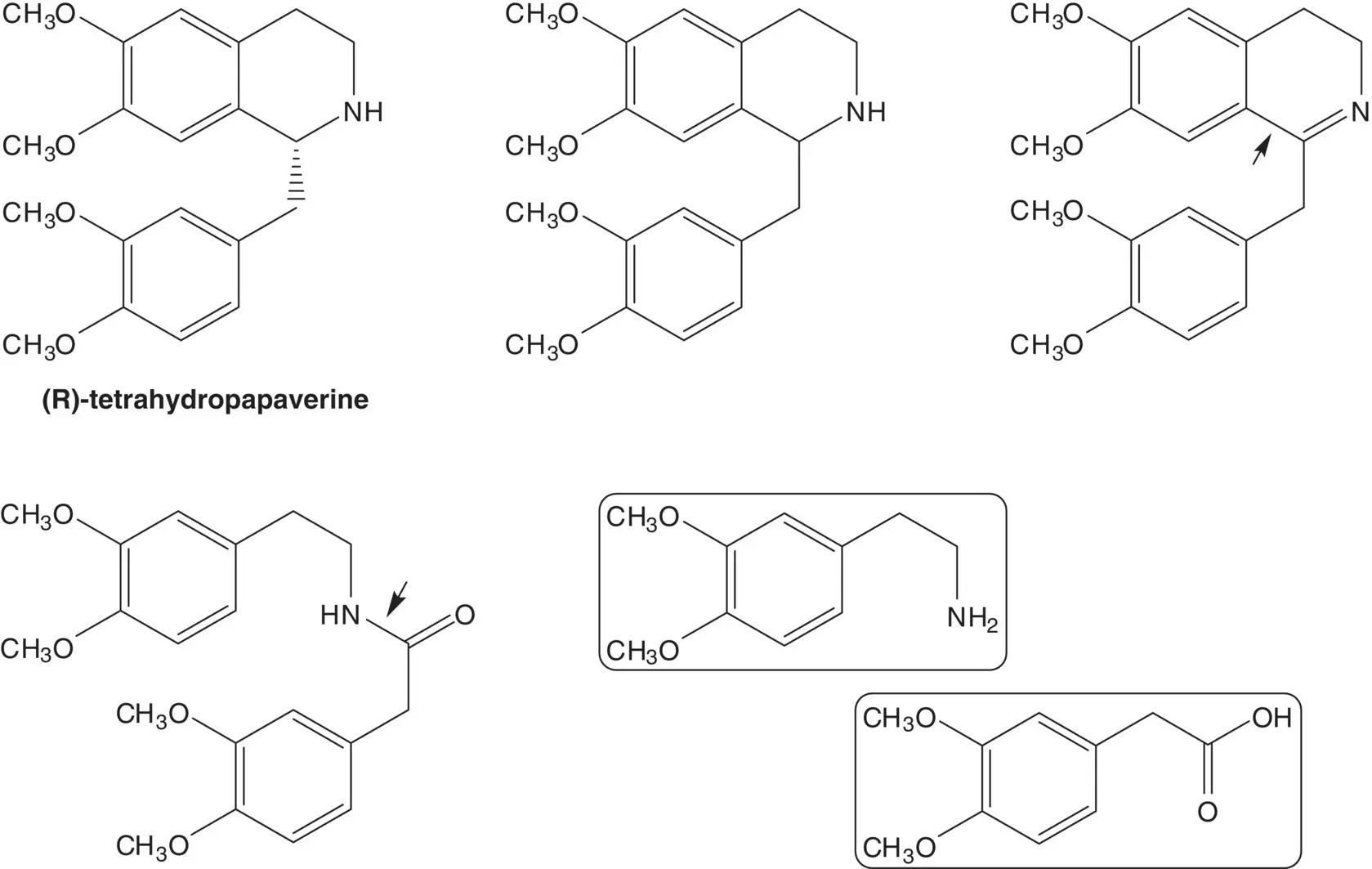
The acrylate is formed by elimination of hydrogen bromide. The diester is formed from 1,5‐pentanediol and 3‐bromopropanoic acid ( Fischer Esterification).
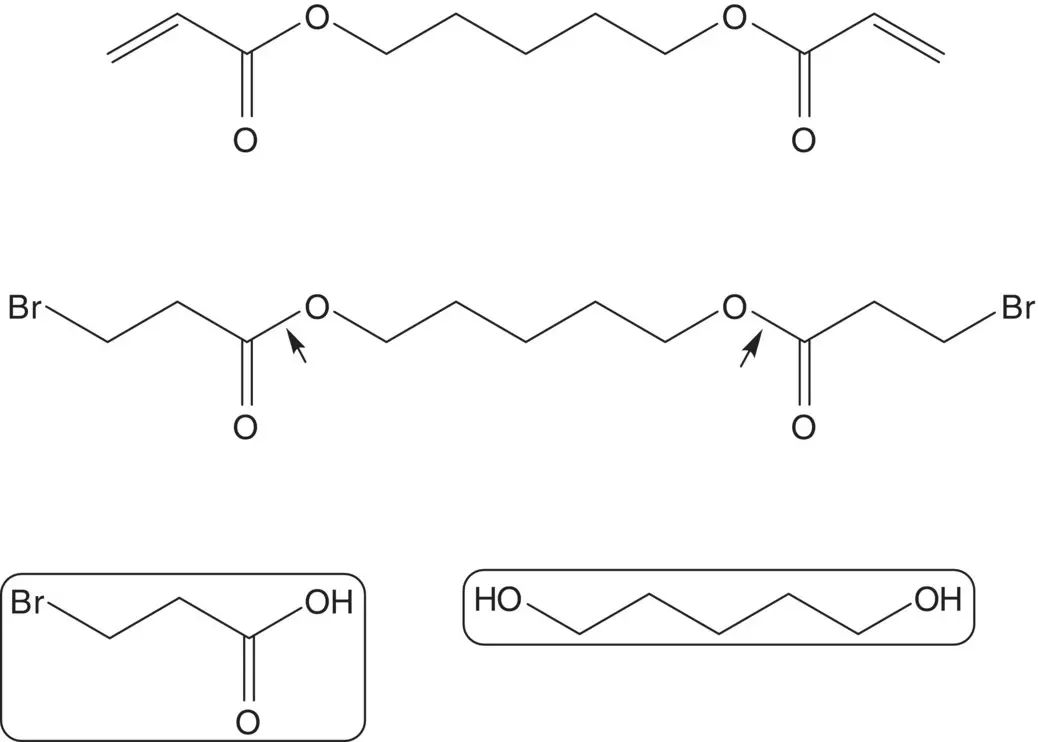
Draw the structures of the retrosynthetic analysis of one alternative route to cisatracurium besylate. List the pros and cons for both routes. Is one route preferred?
Anesthetics, Preoperative Medicines and Medical Gases/Preoperative Medication and Sedation for Short‐Term Procedures
Antidotes and Other Substances Used in Poisonings/Specific
Ophthalmological Preparations/Mydriatics
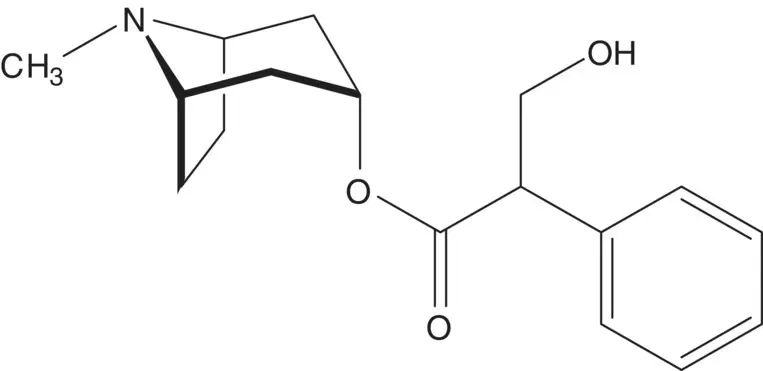
A rigid bicyclic structure is often used to direct the formation of a new chiral carbon.
Читать дальше

























