Passage of carbohydrate carbon through PDH represents an irreversible ‘gate’ through which the carbon cannot gain re-entry, committing carbohydrate to energy provision, either by immediate oxidation of acetyl-CoA, or by storage of the acetyl-CoA as lipid (fatty acid, triacylglycerol) for reconversion back to acetyl-CoA and oxidation at a later date (e.g. in subsequent starvation); this is the reason why PDH is such a highly regulated enzyme – it represents the major control point between carbohydrate and lipid metabolism ( Figure 1.13).
1.3.1.4 Tricarboxylic acid (TCA) cycle
Oxidation of the 2-carbon acetyl (CH 3·CO–) group of acetyl-CoA is achieved by the TCA cycle within the mitochondrial matrix, with the energy from the oxidation of one acetyl group released in the form of electrons (associated with a hydrogen atom as a hydride (H −ion; see Section 1.2.1.3)) and carried by NAD +(×3 per acetyl group, i.e. per turn of the cycle) and FAD (×1) in their reduced forms (NADH; FADH 2). In addition, one step involves a substrate-level phosphorylation – a chemical step which is directly linked to the phosphorylation of ADP (or GDP) to ATP (or GTP) without the need for mitochondrial oxidative phosphorylation – converting GDP into GTP (or ADP into ATP in some cells).
In the TCA cycle ( Box 1.5), the 2-carbon acetyl group of acetyl-CoA combines with oxaloacetate (4 carbons) to form the 6-carbon compound citrate (a TCA, hence the name of the cycle; it is also referred to as the citrate cycle or Krebs cycle). The citrate undergoes two decarboxylation reactions, yielding both the two carbon dioxides and 2 NADH (‘oxidative decarboxylation’ reactions), to form succinyl-CoA (4 carbons). The remainder of the cycle concerns regenerating oxaloacetate from the succinyl-CoA: this process involves the (substrate-level) phosphorylation of GDP to GTP and two further oxidations, yielding the FADH 2and the third NADH, together with the oxaloacetate.
Box 1.5 Tricarboxylic acid cycle: overall scheme
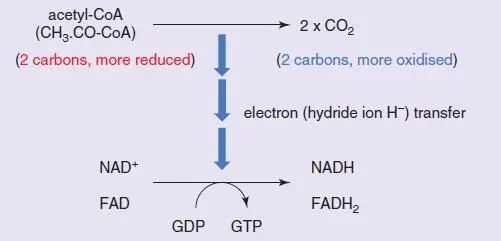
Acetyl-CoA is oxidised by losing electrons (H −ions) and ends up as CO 2. The electrons are captured by NAD +and FAD to become their reduced forms, NADH and FADH 2(and they will in turn pass these electrons on to other electron carriers in the electron transport chain, becoming re-oxidised themselves and ready for further electron carriage). This is achieved in a step-wise fashion by the TCA or Krebs cycle. In addition, as part of the controlled release of energy, one of the steps of the TCA cycle does not have sufficient energy to reduce NAD+ or FAD but does have sufficient energy to phosphorylate GDP to GTP. One acetyl-CoA molecule reduces one FAD and three NAD +molecules.
The stoichiometry is precise, such that both the carbons of the acetyl group are oxidised (though as shown by radiolabelling experiments, not the actual two carbon atoms that entered the cycle), hence all their useful energy is extracted, resulting in two carbon dioxide molecules as ‘waste’ products. The NADH and FADH 2will subsequently be re-oxidised by passing on the electron(s) they carry down the redox proteins of the electron transport chain, regenerating NAD +and FAD in the process for further electron carriage; by coupling this sequential oxidation-reduction to phosphorylation of ADP to ATP (‘oxidative phosphorylation’), a common energy carrier for diverse cellular functions (ATP) is synthesised (see below).
An important point about the TCA cycle is that its intermediates are not specifically dedicated to the cycle – they are also used for other purposes in several other metabolic pathways (e.g. amino acid metabolism; porphyrin synthesis; purine nucleotide metabolism). Processes which utilise TCA cycle intermediates and hence deplete them are termed ‘cataplerotic’ whilst processes which replenish them are termed ‘anaplerotic.’ In order for a substance to be anaplerotic it must have more than two carbons (or equivalent): acetyl groups cannot replenish the cycle because both their carbons (equivalent) are oxidised by the cycle (2 × CO 2) and no net gain of carbon occurs. Since fatty acids represent a chain of 2-carbon groups (they are, effectively, a storage form of acetyl groups – hence their mostly even numbers of carbons), when fatty acids are used for energy (e.g. during starvation) they are broken back down to 2-carbon acetyl groups (by β-oxidation) and cannot therefore be anaplerotic – indeed reliance on fatty acids for oxidation does lead to cataplerosis: if a cell were to derive its energy entirely from fatty acids, eventually cataplerotic depletion of TCA cycle intermediates would occur due to ongoing utilisation of the intermediates by other pathways, combined with the inability of the fatty acids (acetyl groups) to ‘top up’ the same cycle intermediates. The same applies for ketone bodies (acetoacetate, 3-hydroxybutyrate) which are 4-carbon compounds but effectively represent two acetyl groups joined together – a transport form of acetyl-CoA; their utilisation first requires them to be converted back into acetyl-CoA, and they cannot enter the TCA cycle as 4-carbon compounds. Anaplerosis has to occur from carbohydrates or (glucogenic) amino acids, which can bypass the PDH step and insert 4-carbon intermediates (e.g. oxaloacetate; derived from pyruvate) into the cycle. The importance of these anaplerotic pathways, and the enzymes that achieve them, to maintain cellular metabolic efficiency is now well recognised. See later, Box 5.3, for more information.
1.3.1.5 Electron transport chain
The final stage of energy production is the synthesis of ATP by phosphorylation of ADP, coupled to the re-oxidation of NADH and FADH 2to permit further electron carriage to occur (oxidative phosphorylation). The energy is now carried in the phosphoanhydride bonds of the phosphate groups of ATP; these are commonly called ‘ high energy ’ bonds (see Box 1.6).
Box 1.6 High-energy bonds
The term ‘high-energy bond’ is not strictly accurate. They are not ‘special’ bonds, but rather they release their free energy when hydrolysed. They may be denoted ∼, and it can be seen that ADP can act as a source of energy when its terminal phosphate group is hydrolysed to AMP.
AMP: Ad-P
ADP: Ad-P∼P
ATP: Ad-P∼P∼P
ADP has a special role in bioenergetics because beside acting as a (limited) energy source in its own right, its availability is one factor that regulates the rate of ATP synthesis.
A close structural analogue of adenosine triphosphate (ATP) is guanosine triphosphate (GTP) which also carries energy. The presence of these two forms of energy carriage likely represents a form of metabolic compartmentation, separating pathways by their molecular preferences. GTP also has a function in regulation, especially in signal transduction involving G-proteins.
The energy for the phosphorylation is derived from the electrons contained in the reduced forms of the electron carriers. As the electron is passed sequentially down a series of carriers, the released energy is harnessed. The mechanism responsible is the electron transport chain , located in the inner mitochondrial membrane (IMM). The IMM is a highly specialised membrane which is extremely selectively impermeable. It comprises four protein complexes (complex I, II, III, IV) together with a loosely associated cytochrome (cytochrome c) and the (non-protein) quinone CoQ10 (ubiquinone). All these components have variable redox states and act as electron acceptors (oxidising agents) and electron donors (reducing agents); they can function as electron carriers by shifting between their reduced (electron containing) and oxidised (electron deficient) forms. To facilitate this, the proteins contain transition metals (Fe, Cu), complexed in prosthetic groups (e.g. haem) or complexed to sulphur (Fe-S centres) which are readily capable of gaining or losing an electron. They are organised in a sequential arrangement within the IMM such that the electrons are passed down a gradual incremental energy gradient. Complex I oxidises NADH back to NAD +, whilst complex II oxidises FADH 2back to FAD, both passing electrons (as pairs) to CoQ10, thence to complex III, then cytochrome c, and eventually to complex IV. Complex IV, the final redox protein in the chain, combines the electrons it receives with molecular oxygen (O 2, the final electron acceptor), reducing it to water. Complexes I, III, and IV use the energy of electron transfer to pump protons (H +) out of the mitochondrial matrix across the IMM and into the intermembrane space beyond. This creates an electrochemical gradient between the inside and outside of the mitochondrion. The energy of this H +gradient is finally utilised to drive phosphorylation of ADP to ATP: the protons can only re-enter the mitochondrial matrix by passing through ATP synthase (Fo/F 1ATPase; also called complex V although it is not a part of the actual electron transport chain), another IMM-spanning protein. Proton passage through this large protein complex provides the energy for ATP synthesis (the chemiosmotic process). Hence, provided the IMM is otherwise impermeable to protons, the oxidation of NADH/FADH 2by electron transport is tightly coupled to the phosphorylation of ADP to ATP. However, if protons leak through the IMM back into the mitochondrial matrix, the gradient is dissipated (‘uncoupled’) and ATP synthesis cannot occur, leading to mitochondrial inefficiency. The final step in metabolism is the export of ATP out of the mitochondrion and into the cytosol, and this is achieved by an adenine nucleotide translocator also spanning the IMM.
Читать дальше













