8 Chapter 9Table 9.1 Isotope ratios of light stable isotopes.Table 9.2 Coefficients for reduced partition coefficient ratios for oxygen is...Table 9.3 Mineral pair oxygen isotope fractionation factors for temperatures ...Table 9.4 Coefficients for sulfur isotope fractionation: 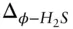 = A × 10Table 9.5 Reference values of nonconventional stable isotope ratios.
= A × 10Table 9.5 Reference values of nonconventional stable isotope ratios.
9 Chapter 10Table 10.1 Characteristics of chondrite groups.Table 10.2 Abundances of the elements.Table 10.3 Van Schmus and Wood petrographic classification of chondrites (aft...Table 10.4 Short-lived radionuclides in the early solar system.Table 10.5 50% Condensation temperatures for solar system gas.Table 10.6 Physical data regarding major solar system objects.Table 10.7 Representative compositions of lunar rocks.Table 10.8 Comparison of the compositions of the Earth and Moon.
10 Chapter 11Table 11.1 Volumes and masses of the Earth's shells.Table 11.2 Comparison of bulk silicate earth major and minor element composit...Table 11.3 Bulk silicate Earth compositions.Table 11.4 Estimated composition of the Earth's core.Table 11.5 Statistical summary of the isotopic composition of oceanic basalts...Table 11.6 Major element composition of the oceanic crust.Table 11.7 Trace element composition of the oceanic crust (all concentrations...Table 11.8 Composition of the upper continental crust.Table 11.9 U, Th, and K concentrations and heat production in various rock ty...Table 11.10 Composition of the middle and lower continental crust.Table 11.11 Composition of the continental crust.
11 Chapter 12Table 12.1 Composition of soil humic and fulvic acids.Table 12.2 Composition of fulvic and humic acids dissolved in natural waters.Table 12.3 Dissolved methane in natural waters.Table 12.4 Stability constants (Log β ) for metal ion–organic ligand compl...Table 12.5 Free energy changes for bacterial reactions.Table 12.6 Kerogen composition
12 Chapter 13Table 13.1 pε of principal aquatic redox couples.Table 13.2 SiO 2/(Al 2O 3+ Fe 2O 3) ratios of soils.Table 13.3 Elemental composition of dried plant matter.Table 13.4 Concentrations in bulk precipitation and throughfall in the Vosage...Table 13.5 Residence times of organic matter and nutrients in forest litter (...Table 13.6 Composition of water from the Coweeta watershed (ppm).Table 13.7 Average composition of dissolved loads of rivers.Table 13.8 Compositions of representative major rivers.Table 13.9 Saline lake brines.
13 Chapter 14Table 14.1 Concentrations of elements dissolved in seawater and river water.Table 14.2 Major ions in seawater.Table 14.3 Residence times of selected elements in seawater.Table 14.4 Composition of representative hydrothermal vent fluids.Table 14.5 Global fluxes to seawater from high-temperature ridge crest hydrot...Table 14.6. Atmospheric flux to the oceans.Table 14.7 Concentrations and fluxes in clays of the Nares Abyssal Plain.Table 14.8 Average composition of Mn nodules and crusts.
14 Chapter 15Table 15.1 US EPA maximum contaminant level in drinking water.
1 Chapter 1 Figure 1.1 The periodic table showing symbols and atomic numbers of naturall... Figure 1.2 The predicted sequence of orbital energies for electrons in atoms... Figure 1.3 The periodic table of naturally occurring elements showing the el... Figure 1.4 First ionization potential of the elements. Figure 1.5 Electronegativities of the elements. Nonmetals are characterized ... Figure 1.6 Ionic radii of the elements. Figure 1.7 Van der Waals interactions arise because of the polar nature of s... Figure 1.8 (a) Geometry of the water molecule. (b) Hydrogen bonds between wa... Figure 1.9 (a) The silica tetrahedron, 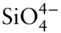 , consists of a silicon atom surroun... Figure 1.10 Geometric relationships between cations and their coordinating a... Figure 1.11 Silicate mineral structures. (a) In orthosilicates such as olivi... Figure 1.12 The Earth in cross-section. The outer rocky part of the planet, ... Figure 1.13 Cross-section of the Earth illustrating relationships between li...
, consists of a silicon atom surroun... Figure 1.10 Geometric relationships between cations and their coordinating a... Figure 1.11 Silicate mineral structures. (a) In orthosilicates such as olivi... Figure 1.12 The Earth in cross-section. The outer rocky part of the planet, ... Figure 1.13 Cross-section of the Earth illustrating relationships between li...
2 Chapter 2 Figure 2.1 Systems in relation to their surroundings. The ball represents ma... Figure 2.2 States of a system. Figure 2.3 An ideal gas thermometer. The colored area is the volume occupied... Figure 2.4 A gas-filled box with a removable partition. When the partition i... Figure 2.5 Possible distribution of molecules of a red and a black gas in a ... Figure 2.6 Two copper blocks at different temperatures separated by an insul... Figure 2.7 There are six possible ways to distribute six energy units so tha... Figure 2.8 (a) Probability of one of two copper blocks of equal mass in ther... Figure 2.9 Occupation of vibrational energy levels calculated from the Boltz... Figure 2.10 Vibrational contribution to heat capacity as a function of k T / hν ... Figure 2.11 Transformations on a temperature–pressure diagram. Changes in st...
3 Chapter 3 Figure 3.1 Graphical representation of the system Al 2O 3−H 2O. Figure 3.2 Phase diagram for the system Al 2O 3–H 2O–SiO 2. The lines are calle... Figure 3.3 Comparison of the graphite–diamond phase boundary calculated fro... Figure 3.4 Vapor pressure of water and dioxane in a water–dioxane mixture s... Figure 3.5 Variation of the partial molar volumes of water and ethanol as a... Figure 3.6 Free energy of mixing as a function of temperature in the ideal ... Figure 3.7 Molar free energy in an ideal mixture and a graphical illustrati... Figure 3.8 Schematic plot of the chemical potential of component i in solut... Figure 3.9 (a) Structure of the water molecule. Bond angle in the liquid pha... Figure 3.10 Solvation of a cation in aqueous solution. In the first solvati... Figure 3.11 Relationship of activity and molality, reference state, and sta... Figure 3.12 Apparent molar volume of NaCl in aqueous solution as a function... Figure 3.13 Standard molar volume of NaCl in aqueous solution as a function... Figure 3.14 An ion surrounded by a cloud of oppositely charged ions, as ass... Figure 3.15 Variation of the Ca 2+activity coefficient with ionic stren... Figure 3.16 Ionic strength of natural electrolyte solutions and the applica... Figure 3.17 Log of the solubility constant of barite plotted against the in... Figure 3.18 Electrode reactions in the Daniell cell. Figure 3.19 p ε–pH diagram showing predominance regions for ferric and ferro... Figure 3.20 Stability regions for magnetite and hematite in equilibrium wit... Figure 3.21 pε and pH of various waters on and near the surface of the Eart... Figure 3.22 Oxygen buffer curves in the system Fe−Si−O at 1 bar. QIF, IW, W...
4 Chapter 4 Figure 4.1 Exsolution lamellae of Na-rich feldspar (albite) from a K-rich (o... Figure 4.2 Alkali feldspar solid solution computed at 600°C and 200 MPa (2 k... Figure 4.3 Δ G realof alkali feldspar solution computed for a series of tempe... Figure 4.4 Δ G surface for the alkali feldspar solid solution as a function o... Figure 4.5 Activity and activity cofficient of albite in alkali feldspar sol... Figure 4.6 (a) Schematic isothermal, isobaric G–X plot for a real solution s... Figure 4.7 A small portion of a  – X plot illustrating the origin of th... Figure 4.8 P–T phase diagram for SiO 2. This system has one component b... Figure 4.9 Phase diagram ( T – X ) for the two-component system diopside–anorthi... Figure 4.10 Computed phase diagram for the system anorthite–diopside (CaAl 2S... Figure 4.11
– X plot illustrating the origin of th... Figure 4.8 P–T phase diagram for SiO 2. This system has one component b... Figure 4.9 Phase diagram ( T – X ) for the two-component system diopside–anorthi... Figure 4.10 Computed phase diagram for the system anorthite–diopside (CaAl 2S... Figure 4.11  − X diagram for a two-component system exhibiting complete solut... Figure 4.12 Plot of molar free energy vs. composition (
− X diagram for a two-component system exhibiting complete solut... Figure 4.12 Plot of molar free energy vs. composition ( 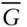 –X 2) for two-phase d... Figure 4.13 Two univariant systems: (a) a liquid plus two solid solutions, a... Figure 4.14
–X 2) for two-phase d... Figure 4.13 Two univariant systems: (a) a liquid plus two solid solutions, a... Figure 4.14 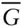 – X diagrams and a T–X phase diagram for the plagioclase–l... Figure 4.15 Phase diagram for a metapelite calculated using THERMOCALC. The ... Figure 4.16 Phase diagram for Al 2SiO 5(kyanite–sillimanite–andalusite) as de... Figure 4.17 Isopleths of Al in orthopyroxene (thin red lines; weight percent... Figure 4.18 a. Lamellae of orthopyroxene (opx; bright lines) exsolved from a... Figure 4.19 Comparison of calculated (solid lines) and experimentally observ... Figure 4.20 Olivine saturation surface constructed by Roeder and Emslie (197... Figure 4.21 The TiO 2–FeO–Fe 2O 3ternary system. Phases are: FeO, wüstite; Fe 2 Figure 4.22 Relationship of composition of coexisting titanomagnetite and il... Figure 4.23 Silicate structures. (a) Short-range silicate structures in melt... Figure 4.24 (a) Structure of pure silica glass and (b) a silica-rich glass w... Figure 4.25 SiO 2concentrations in a melt produced by melting of peridotite ... Figure 4.26 Compositions of pyroxenes found in lavas from the Cameroon Line.... Figure 4.27 (a) Three-dimensional P–T–V phase diagram for liquid... Figure 4.28 (a) Relationship between activity and molality of NaCl in aqueou... Figure 4.29 Observed mean ion activity coefficient, γ ±, of NaCl as a fu... Figure 4.30 Comparison of the electrostatic contribution to the mean ion act... Figure 4.31 In formation of ion pairs, the solvation shells may remain intac... Figure 4.32 Effects of ion association on the activity coefficient. Mean ion... Figure 4.33 Measured mean ionic activity coefficients in MgCl 2solution as a... Figure 4.34 (a). Partial molar heat capacity of aqueous CaCl 2at 17.5 MPa (a... Figure 4.35 Temperature–composition phase diagram for aqueous solutions of N...
– X diagrams and a T–X phase diagram for the plagioclase–l... Figure 4.15 Phase diagram for a metapelite calculated using THERMOCALC. The ... Figure 4.16 Phase diagram for Al 2SiO 5(kyanite–sillimanite–andalusite) as de... Figure 4.17 Isopleths of Al in orthopyroxene (thin red lines; weight percent... Figure 4.18 a. Lamellae of orthopyroxene (opx; bright lines) exsolved from a... Figure 4.19 Comparison of calculated (solid lines) and experimentally observ... Figure 4.20 Olivine saturation surface constructed by Roeder and Emslie (197... Figure 4.21 The TiO 2–FeO–Fe 2O 3ternary system. Phases are: FeO, wüstite; Fe 2 Figure 4.22 Relationship of composition of coexisting titanomagnetite and il... Figure 4.23 Silicate structures. (a) Short-range silicate structures in melt... Figure 4.24 (a) Structure of pure silica glass and (b) a silica-rich glass w... Figure 4.25 SiO 2concentrations in a melt produced by melting of peridotite ... Figure 4.26 Compositions of pyroxenes found in lavas from the Cameroon Line.... Figure 4.27 (a) Three-dimensional P–T–V phase diagram for liquid... Figure 4.28 (a) Relationship between activity and molality of NaCl in aqueou... Figure 4.29 Observed mean ion activity coefficient, γ ±, of NaCl as a fu... Figure 4.30 Comparison of the electrostatic contribution to the mean ion act... Figure 4.31 In formation of ion pairs, the solvation shells may remain intac... Figure 4.32 Effects of ion association on the activity coefficient. Mean ion... Figure 4.33 Measured mean ionic activity coefficients in MgCl 2solution as a... Figure 4.34 (a). Partial molar heat capacity of aqueous CaCl 2at 17.5 MPa (a... Figure 4.35 Temperature–composition phase diagram for aqueous solutions of N...
Читать дальше

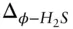 = A × 10Table 9.5 Reference values of nonconventional stable isotope ratios.
= A × 10Table 9.5 Reference values of nonconventional stable isotope ratios.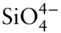 , consists of a silicon atom surroun... Figure 1.10 Geometric relationships between cations and their coordinating a... Figure 1.11 Silicate mineral structures. (a) In orthosilicates such as olivi... Figure 1.12 The Earth in cross-section. The outer rocky part of the planet, ... Figure 1.13 Cross-section of the Earth illustrating relationships between li...
, consists of a silicon atom surroun... Figure 1.10 Geometric relationships between cations and their coordinating a... Figure 1.11 Silicate mineral structures. (a) In orthosilicates such as olivi... Figure 1.12 The Earth in cross-section. The outer rocky part of the planet, ... Figure 1.13 Cross-section of the Earth illustrating relationships between li...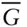 – X plot illustrating the origin of th... Figure 4.8 P–T phase diagram for SiO 2. This system has one component b... Figure 4.9 Phase diagram ( T – X ) for the two-component system diopside–anorthi... Figure 4.10 Computed phase diagram for the system anorthite–diopside (CaAl 2S... Figure 4.11
– X plot illustrating the origin of th... Figure 4.8 P–T phase diagram for SiO 2. This system has one component b... Figure 4.9 Phase diagram ( T – X ) for the two-component system diopside–anorthi... Figure 4.10 Computed phase diagram for the system anorthite–diopside (CaAl 2S... Figure 4.11 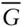 − X diagram for a two-component system exhibiting complete solut... Figure 4.12 Plot of molar free energy vs. composition (
− X diagram for a two-component system exhibiting complete solut... Figure 4.12 Plot of molar free energy vs. composition ( 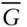 –X 2) for two-phase d... Figure 4.13 Two univariant systems: (a) a liquid plus two solid solutions, a... Figure 4.14
–X 2) for two-phase d... Figure 4.13 Two univariant systems: (a) a liquid plus two solid solutions, a... Figure 4.14 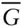 – X diagrams and a T–X phase diagram for the plagioclase–l... Figure 4.15 Phase diagram for a metapelite calculated using THERMOCALC. The ... Figure 4.16 Phase diagram for Al 2SiO 5(kyanite–sillimanite–andalusite) as de... Figure 4.17 Isopleths of Al in orthopyroxene (thin red lines; weight percent... Figure 4.18 a. Lamellae of orthopyroxene (opx; bright lines) exsolved from a... Figure 4.19 Comparison of calculated (solid lines) and experimentally observ... Figure 4.20 Olivine saturation surface constructed by Roeder and Emslie (197... Figure 4.21 The TiO 2–FeO–Fe 2O 3ternary system. Phases are: FeO, wüstite; Fe 2 Figure 4.22 Relationship of composition of coexisting titanomagnetite and il... Figure 4.23 Silicate structures. (a) Short-range silicate structures in melt... Figure 4.24 (a) Structure of pure silica glass and (b) a silica-rich glass w... Figure 4.25 SiO 2concentrations in a melt produced by melting of peridotite ... Figure 4.26 Compositions of pyroxenes found in lavas from the Cameroon Line.... Figure 4.27 (a) Three-dimensional P–T–V phase diagram for liquid... Figure 4.28 (a) Relationship between activity and molality of NaCl in aqueou... Figure 4.29 Observed mean ion activity coefficient, γ ±, of NaCl as a fu... Figure 4.30 Comparison of the electrostatic contribution to the mean ion act... Figure 4.31 In formation of ion pairs, the solvation shells may remain intac... Figure 4.32 Effects of ion association on the activity coefficient. Mean ion... Figure 4.33 Measured mean ionic activity coefficients in MgCl 2solution as a... Figure 4.34 (a). Partial molar heat capacity of aqueous CaCl 2at 17.5 MPa (a... Figure 4.35 Temperature–composition phase diagram for aqueous solutions of N...
– X diagrams and a T–X phase diagram for the plagioclase–l... Figure 4.15 Phase diagram for a metapelite calculated using THERMOCALC. The ... Figure 4.16 Phase diagram for Al 2SiO 5(kyanite–sillimanite–andalusite) as de... Figure 4.17 Isopleths of Al in orthopyroxene (thin red lines; weight percent... Figure 4.18 a. Lamellae of orthopyroxene (opx; bright lines) exsolved from a... Figure 4.19 Comparison of calculated (solid lines) and experimentally observ... Figure 4.20 Olivine saturation surface constructed by Roeder and Emslie (197... Figure 4.21 The TiO 2–FeO–Fe 2O 3ternary system. Phases are: FeO, wüstite; Fe 2 Figure 4.22 Relationship of composition of coexisting titanomagnetite and il... Figure 4.23 Silicate structures. (a) Short-range silicate structures in melt... Figure 4.24 (a) Structure of pure silica glass and (b) a silica-rich glass w... Figure 4.25 SiO 2concentrations in a melt produced by melting of peridotite ... Figure 4.26 Compositions of pyroxenes found in lavas from the Cameroon Line.... Figure 4.27 (a) Three-dimensional P–T–V phase diagram for liquid... Figure 4.28 (a) Relationship between activity and molality of NaCl in aqueou... Figure 4.29 Observed mean ion activity coefficient, γ ±, of NaCl as a fu... Figure 4.30 Comparison of the electrostatic contribution to the mean ion act... Figure 4.31 In formation of ion pairs, the solvation shells may remain intac... Figure 4.32 Effects of ion association on the activity coefficient. Mean ion... Figure 4.33 Measured mean ionic activity coefficients in MgCl 2solution as a... Figure 4.34 (a). Partial molar heat capacity of aqueous CaCl 2at 17.5 MPa (a... Figure 4.35 Temperature–composition phase diagram for aqueous solutions of N...










