 All animal cell plasma membranes are made primarily of molecules called phospholipids. Plant cells also have phospholipid membranes, but plants also have cell walls made of more rigid molecules such as cellulose, which gives them their stiffness. These phospholipid membranes in cells are everywhere, so if you think they’re fundamental to life, you’re almost certainly right! Although we don’t know the details of how single cells evolved from the nonliving soup of organic molecules in ancient waters billions of years ago, the formation of stable phospholipid “bags” of organic molecules probably preceded the evolution of DNA that now controls the activity and reproduction of cells.
All animal cell plasma membranes are made primarily of molecules called phospholipids. Plant cells also have phospholipid membranes, but plants also have cell walls made of more rigid molecules such as cellulose, which gives them their stiffness. These phospholipid membranes in cells are everywhere, so if you think they’re fundamental to life, you’re almost certainly right! Although we don’t know the details of how single cells evolved from the nonliving soup of organic molecules in ancient waters billions of years ago, the formation of stable phospholipid “bags” of organic molecules probably preceded the evolution of DNA that now controls the activity and reproduction of cells.
Why are phospholipid membranes so essential to life? Phospholipids are molecules that spontaneously form membranes in salt solutions such as seawater, which is where life probably evolved. They do this because they’re polarized, with different chemical properties at the two ends of the molecule (the next section explains this in more detail). The important thing about phospholipid membranes is that they’re very stable in salt solutions and almost totally impervious to the movement of water or ions through them. Phospholipid membranes keep the inside of the cell in and the outside of the cell out.
Focusing on phospholipid chemistry
What are phospholipids and how do they form membranes? First, consider the common phospholipid molecule, as shown in Figure 2-1. To understand the membrane function, remember that phospholipid molecules have a hydrophilic (meaning, “water liking”) “head” and two hydrophobic (meaning “water fearing”) “tails.” Molecules with this property are called amphipathic.
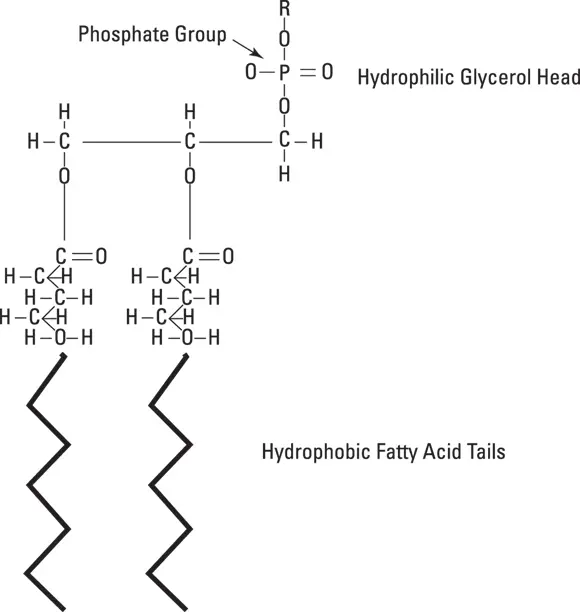
Figure 2-1:Phospho-lipid molecular structure.
 Why is a molecule hydrophilic? Water molecules (H 2O) are polarized because most of their electrons are concentrated around the oxygen atom, leaving the two hydrogen atoms with a deficiency of electric charge. So, the two hydrogen ends of the water molecule are slightly positive, with a slightly negative charge around the oxygen in the middle. Although water molecules move freely in the liquid state, most of the time adjacent water molecules have the hydrogen atoms in one water molecule near the oxygen atoms in adjacent ones.
Why is a molecule hydrophilic? Water molecules (H 2O) are polarized because most of their electrons are concentrated around the oxygen atom, leaving the two hydrogen atoms with a deficiency of electric charge. So, the two hydrogen ends of the water molecule are slightly positive, with a slightly negative charge around the oxygen in the middle. Although water molecules move freely in the liquid state, most of the time adjacent water molecules have the hydrogen atoms in one water molecule near the oxygen atoms in adjacent ones.
 Common table salt (NaCl) dissolves in water because the positively charged sodium atom (Na +) is attracted to the negative oxygen region of the water molecules and “lets go” of the chloride (Cl –) to which it is bonded. The chloride, in turn, tends to stick to the hydrogen ends of the water molecule.
Common table salt (NaCl) dissolves in water because the positively charged sodium atom (Na +) is attracted to the negative oxygen region of the water molecules and “lets go” of the chloride (Cl –) to which it is bonded. The chloride, in turn, tends to stick to the hydrogen ends of the water molecule.
So, how does the hydrophilic water molecule relate to phospholipid membranes? When phospholipid molecules are floating around in water, the polar heads are attracted to and bind water molecules. But the hydrophobic tails don’t bind water; instead, they’re more likely to interact with each other. The most stable way for these molecules to arrange themselves, then, is for the hydrophobic tails to bind together, with the heads protruding outward from a bilayer and contacting water. The schematic structure of the molecule viewed this way is shown in Figure 2-2, and the stable bilayer (molecular layer two molecules thick) is shown in Figure 2-3.
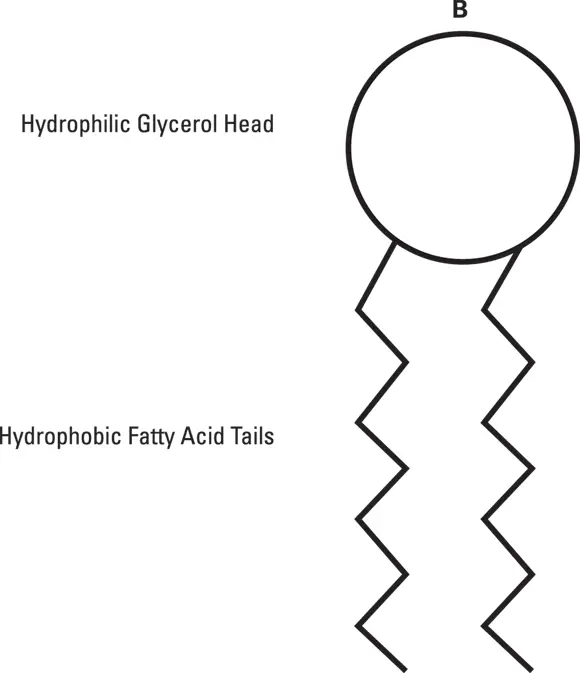
Figure 2-2:A simplified ball-and-stick model of a single phospholipid molecule.
The last piece of this puzzle is that, unlike what might be implied in Figure 2-3, phospholipid bilayers won’t form infinitely large sheets in aqueous environments. Instead, the only stable configuration is for the bilayer to close on itself and form a sphere. The size of this sphere is a function of the water forces and the chemistry of the phospholipid.
 It’s interesting that the extracellular fluid around cells in animals resembles the seawater in which cells originally evolved. If you think this wasn’t an accident, you’re right!
It’s interesting that the extracellular fluid around cells in animals resembles the seawater in which cells originally evolved. If you think this wasn’t an accident, you’re right!
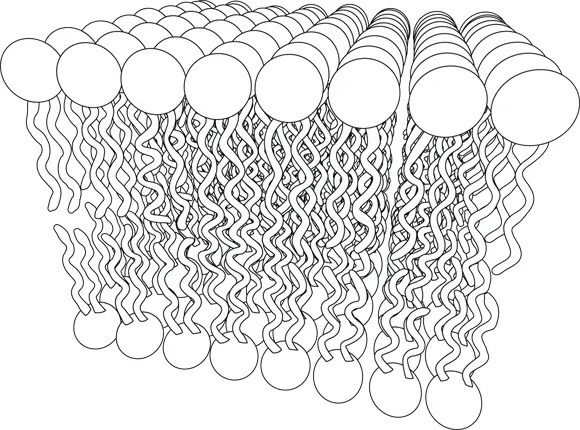
Figure 2-3:Phospho-lipid molecules assemble to form the plasma membrane.
Seeing cells’ differences
All of the cells in your body have the same lipid bilayer structure for their plasma membrane, but all cells are not the same. What makes a kidney cell or neuron or skin cell all different from each other? Two major differences apply to all the hundreds of distinct cell types in your body:
Different biochemical reaction sequences: These sequences are particular to specific cell types controlled by selective expression of a subset of the total genes in each cell (although each cell has identical DNA).
Different protein structures in the plasma membranes: These structures control the flow of ions and other substances through the cell’s membrane.
These two differences are not entirely separate from each other. One of the major functions of the different biochemical reactions going on inside cells is generating and maintaining protein complexes for the cell membrane. All cells, of course, generate the phospholipids to maintain their plasma membranes.
Neurons differ from other cells in your body because they have a large number of specific types of ion channels made of protein complexes specified by a subset of the genes from the DNA in the cell nucleus. Not only do neurons have many more types of these channels than other cells, but many differences exist among neurons because of the hundreds of channel types they express, and the relative numbers of each.
Regulating Water and Cell Volume
The plasma membranes of neurons, as well as other cells in the body, are stable, but not rigid like the cell walls of plants. This means that neurons are flexible and can be deformed by mechanical pressure. Neurons exist within a matrix of glial cells and an extracellular matrix that holds the tissue together.
 Although the extracellular matrix around cells is very effective in maintaining a supporting environment that prevents neurons from being damaged (to a limit, of course) by mechanical forces, neurons must actively resist another type of force — osmotic pressure.
Although the extracellular matrix around cells is very effective in maintaining a supporting environment that prevents neurons from being damaged (to a limit, of course) by mechanical forces, neurons must actively resist another type of force — osmotic pressure.
Observing osmotic pressure
What is osmotic pressure, and why does it matter to cells? Well, you’ve probably heard the classic lines, “Water, water, everywhere; nor any drop to drink.” Consider how people stranded in the middle of the ocean can’t survive by drinking the seawater all around them. High concentrations of salts (primarily sodium chloride) are dissolved in seawater. If seawater is added to the already highly concentrated extracellular fluid around our cells, the result is that water will tend to move from the higher concentration inside our cells to the lower concentration (relative to the concentration of ions) outside.
Читать дальше

 All animal cell plasma membranes are made primarily of molecules called phospholipids. Plant cells also have phospholipid membranes, but plants also have cell walls made of more rigid molecules such as cellulose, which gives them their stiffness. These phospholipid membranes in cells are everywhere, so if you think they’re fundamental to life, you’re almost certainly right! Although we don’t know the details of how single cells evolved from the nonliving soup of organic molecules in ancient waters billions of years ago, the formation of stable phospholipid “bags” of organic molecules probably preceded the evolution of DNA that now controls the activity and reproduction of cells.
All animal cell plasma membranes are made primarily of molecules called phospholipids. Plant cells also have phospholipid membranes, but plants also have cell walls made of more rigid molecules such as cellulose, which gives them their stiffness. These phospholipid membranes in cells are everywhere, so if you think they’re fundamental to life, you’re almost certainly right! Although we don’t know the details of how single cells evolved from the nonliving soup of organic molecules in ancient waters billions of years ago, the formation of stable phospholipid “bags” of organic molecules probably preceded the evolution of DNA that now controls the activity and reproduction of cells.
 Why is a molecule hydrophilic? Water molecules (H 2O) are polarized because most of their electrons are concentrated around the oxygen atom, leaving the two hydrogen atoms with a deficiency of electric charge. So, the two hydrogen ends of the water molecule are slightly positive, with a slightly negative charge around the oxygen in the middle. Although water molecules move freely in the liquid state, most of the time adjacent water molecules have the hydrogen atoms in one water molecule near the oxygen atoms in adjacent ones.
Why is a molecule hydrophilic? Water molecules (H 2O) are polarized because most of their electrons are concentrated around the oxygen atom, leaving the two hydrogen atoms with a deficiency of electric charge. So, the two hydrogen ends of the water molecule are slightly positive, with a slightly negative charge around the oxygen in the middle. Although water molecules move freely in the liquid state, most of the time adjacent water molecules have the hydrogen atoms in one water molecule near the oxygen atoms in adjacent ones. Common table salt (NaCl) dissolves in water because the positively charged sodium atom (Na +) is attracted to the negative oxygen region of the water molecules and “lets go” of the chloride (Cl –) to which it is bonded. The chloride, in turn, tends to stick to the hydrogen ends of the water molecule.
Common table salt (NaCl) dissolves in water because the positively charged sodium atom (Na +) is attracted to the negative oxygen region of the water molecules and “lets go” of the chloride (Cl –) to which it is bonded. The chloride, in turn, tends to stick to the hydrogen ends of the water molecule.












