Halogen Bonding in Solution
Здесь есть возможность читать онлайн «Halogen Bonding in Solution» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Halogen Bonding in Solution
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 100
- 1
- 2
- 3
- 4
- 5
Halogen Bonding in Solution: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Halogen Bonding in Solution»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Halogen Bonding in Solution
Halogen Bonding in Solution
Halogen Bonding in Solution — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Halogen Bonding in Solution», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
The first example of LCs assembled by halogen bonding was reported by the Bruce lab in 2004 [172]. Here, alkoxystilbazole derivatives were used as halogen bond acceptors and pentafluoroiodobenzene as the donor ( Figure 1.17). X‐ray crystallographic studies suggested the LC formation resulted from a CI⋯N halogen bond. The dimeric complex formed LCs only when cooling, known as monotropic formation. However, alkoxystilbazoles with longer alkyl chains ( n > 6) resulted in enantiotropic LCs (occurring at both heating and cooling cycles). Exchanging the iodine donor for a weaker bromopentafluorobenzene precluded LC formation. The LC formation temperature was lower for the halogen bond derivatives than hydrogen bonding analogues – a property that is generally beneficial for LCs operating near room temperature (e.g. liquid crystal display [LCD] displays). Many other early halogen bond LCs incorporated iodoperfluorobenzene donors, as they form moderately strong halogen bonds and are readily available for purchase [173–176]. However, in 2013, Bruce used molecular iodine as a halogen bond donor to create LCs with stilbazole acceptors [177]. The high temperature stability (>200 °C) of the mesophase was attributed to the intermolecular iodine–iodine contacts. These initial examples demonstrate the ability of the halogen bond to facilitate LC formation with favorable properties.
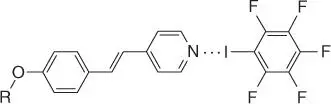
Figure 1.17The first example of a halogen bonding LC developed by Bruce. Alkyl chains R related to LC behavior.
The Bruce lab, along with Metrangolo and Resnati, also contributed to the first example of ionic LCs using halogen bonds [173]. Using tricomponent imidazolium cations and neutral perfluoroiodo halogen bond donors bound to iodide, they showed that the alkyl chains on the organocations did not drive liquid crystallinity and even smaller chain lengths ( n = 2) formed a mesophase. Later studies made these systems light responsive [178].
Photoresponsive LCs, used in displays, nanotechnology, and photo‐driven devices, provide on–off switchable liquid crystallinity. Toward this end, the halogen bond has been integrated into photoresponsive LCs. For example, Priimagi et al. [175] paired the photoactive azo group on a halogen bond donor with an alkoxystilbazole acceptor to produce UV‐active halogen bonding LCs ( Figure 1.18). Separately, neither of these molecules exhibited an LC phase, but together they induced anisotropy when irradiated with polarized UV light. Replacing a CH hydrogen bond donor in tetrafluorobenzene with an iodo halogen bond donor in iodotetrafluorobenzene resulted in a decrease of the phase‐transition temperature that was dependent on the concentration of iodoperfluorobenzene, along with improved chiral light absorption [179].
The Li group doped commercially available achiral LCs with chiral halogen bonding molecular switches to produce helical cholesteric LCs (CLCs) [180]. The CLCs operate reversibly under thermal or light response. Reflection colors for these CLCs were temperature dependent, producing red, green, and blue colors. Additionally, the helical twisting power (HLC), known as the amount of chiral LC formation, could be altered by UV light interacting with the halogen bond CLCs. This concept shows that halogen bonding can be used to optimize doped LC systems to create photonic devices.
1.5.3 Supramolecular Polymers
1.5.3.1 LC Polymers
Supramolecular polymers are arrays of small molecules or linear polymeric chains held together by noncovalent interactions. LC polymers have many of the characteristics of other polymers, including mechanical strength at high temperatures, chemical resistance, and flame resistance, while maintaining LC order. The strength and directionality of the halogen bond make it an interesting noncovalent interaction to be used in polymer science. Yet, there are very limited examples, as detailed by a review in 2012 [181]. Xu et al. created the first LC polymer mediated by halogen bonds using bis(iodotetrafluorophenoxy) alkane donors, which formed halogen bonds with nitrogen acceptors on various stilbazole derivatives ( Figure 1.19) [174]. The formation temperatures of the halogen bond LCs were narrower than hydrogen bond analogues utilizing carboxyl‐pyridine binding. The authors attribute the stabilization of the hydrogen bond derivatives to weak pyridine CH hydrogen bonds to the carbonyl oxygen. A second example comes from Cho et al. who developed an alternating hydrogen bond–halogen bond [182] system, which produced a mesophase at much broader temperatures than halogen bonding analogues. The examples above highlight how the halogen bond can influences LC polymer formation; however more studies are needed to understand the role of halogen bonding in their construction.
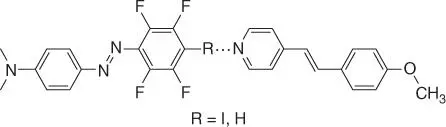
Figure 1.18The first example of a photoactive halogen bonding LC developed by Priimagi et al.
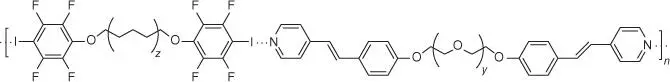
Figure 1.19The first example of a polymeric halogen bonding LC developed by Xu.
(Xu et al. [174].)
1.5.3.2 Light‐sensitive Polymers
A seminal study of light‐sensitive polymers compared hydrogen and halogen bond‐based azobenzene photopolymers [183]. It was found that the halogen‐bonded polymers had a greater light‐induced mass transport efficiency than the hydrogen bond analogues. The use of halogens did not change the photophysical or electronic properties significantly, suggesting that incorporation of halogen bond motifs into other known systems could easily modulate performance. Later studies of azobenzene polymers as light‐induced surface patterning polymers show that halogen bonding species outperform hydrogen bonding ones in terms of patterning efficiency, which the authors attribute to the high directionality of the halogen bond. The efficiency was also shown to be directly proportional with halogen bond strength [184].
1.5.3.3 Block Polymers
Block copolymers consist of two or more covalently linked polymers. The Taylor lab developed a reversible addition‐fragmentation chain transfer (RAFT) polymerization where amine acceptors were combined with iodoperfluorobenzene halogen bond donors, producing supramolecular diblock polymers with higher‐order sphere, vesicle, and rodlike structures [176]. Similar to hydrogen bonding supramolecular diblock polymers, these formations were also highly solvent dependent. Further developments to these systems revealed that well‐defined inverted vesicle morphologies could be facilitated by the hydrophobicity of the halogen bond [185].
Triblock terpolymers are another class of polymer that can form microparticles with more functional domains than diblock polymers. However, strategies to predict how triblock terpolymers assemble are in their infancy. Quintieri et al. first developed ABC 3D styrene‐based triblock terpolymers utilizing the halogen bond [186]. In this study, a variety of hydroxy hydrogen bond and perfluoroiodobenzene halogen bond donors were used to form microparticles under confinement ( Figure 1.20a). Interestingly, the particles formed either lamella–sphere or lamella–lamella morphologies based on the type of donor ( Figure 1.20b). In general, weaker hydrogen or halogen donors lead to increased particle volume. Therefore, they found that both the donor strength and intermolecular packing interactions were important for the overall morphology of the nanoparticle.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Halogen Bonding in Solution»
Представляем Вашему вниманию похожие книги на «Halogen Bonding in Solution» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Halogen Bonding in Solution» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












