Halogen Bonding in Solution
Здесь есть возможность читать онлайн «Halogen Bonding in Solution» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Halogen Bonding in Solution
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 100
- 1
- 2
- 3
- 4
- 5
Halogen Bonding in Solution: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Halogen Bonding in Solution»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Halogen Bonding in Solution
Halogen Bonding in Solution
Halogen Bonding in Solution — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Halogen Bonding in Solution», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
Disclaimer aside, ESP maps are still highly informative. They have helped justify the amphoteric behavior of halogens observed in the solid state, where electrophiles approach the halogen “side‐on” orthogonal to the CX bond and nucleophiles “head‐on” in line with the CX bond [4]. In particular, ESP studies by Politzer and Murray [5,141] led to the establishment of the σ‐hole concept, which has proven to be a widely valuable tool for conceptualizing the halogen bond and has contributed to the renaissance of other σ‐hole‐type interactions like chalcogen and pnictogen bonding [142]. Additionally, the ease of constructing ESP maps has led to their use in predicting relative halogen bond strength. For example, Politzer showed that the iodine V S,maxvalues of iodobenzene derivatives largely positively correlate with their interaction energies with acetone [139] ( Figure 1.14; Table 1.2). This relationship has been demonstrated a number of times theoretically [130,143] and has led to the use of V S,maxvalues as predictors of solid‐state structures [72,75,144] and performance in solution [145]. Widespread application of V S,maxand ESP maps has likely contributed to the halogen bond being mistakenly viewed as a purely electrostatic interaction; however other components are frequently important to fully describe the interaction [146]. For example, there are a number of cases where a more positive V S,maxdoes not correlate with a stronger halogen bond [147].
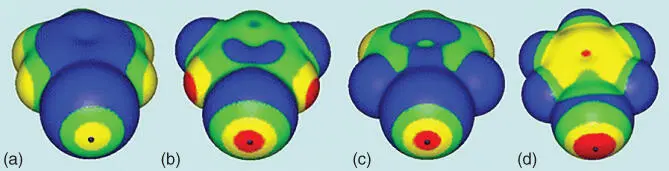
Figure 1.14Computed ESP maps on 0.001 au molecular surfaces of (a) iodobenzene, (b) meta ‐difluoroiodobenzene, (c) ortho ‐difluoroiodobenzene, and (d) pentafluoroiodobenzene. Color ranges, in kcal/mol, are red, greater than 20; yellow, between 20 and 10; green, between 10 and 0; and blue, negative. Black hemispheres denote the positions of the iodine V S,max.
Source: From Riley et al. [139]. © 2011 Springer Nature.
Table 1.2Table of iodine V S,maxvalues and interaction energies (Δ E ) of iodobenzene derivatives with acetone.
Source: Adapted from Riley et al. [139]. Copyright 2011 John Wiley & Sons.
| Interaction angle | |||
| At (X⋯OC) = 180° | At optimum X⋯OC angle | ||
| System | V S,max(kcal/mol) | Δ E (kcal/mol) | Δ E (kcal/mol) |
| Iodobenzene | 17.3 | −2.44 | −3.22 |
| meta‐ Difluoroiodobenzene | 26.1 | −3.38 | −4.13 |
| ortho‐ Difluoroiodobenzene | 25.5 | −3.64 | −4.71 |
| para ‐Fluoroiodobenzene | 35.9 | −4.88 | −5.97 |
1.4.3 Limitations on Electrostatic Potential
While ESP is an effective tool for predicting and conceptualizing interactions, there are limitations. Obviously, contacts that are not primarily electrostatic in nature cannot be accurately predicted, such as those reliant on polarization or charge transfer. Furthermore, ESP maps are only for isolated molecules and therefore do not account for other nuances when two molecules come together. For example, ESP maps do not calculate changes in electron distribution resulting from polarization due to incoming molecules. Therefore, to more accurately predict the strength of a halogen bond, more involved computational techniques that factor additional variables should be considered.
1.4.4 Atomic Orbital Theory and the σ‐Hole
Formation of the σ‐hole and the halogen bond interaction can also be described using atomic orbital theory. To paraphrase Clark, Murray, and Politzer, the electron‐deficient σ‐hole is caused by depleted occupancy in the outer lobe of a p‐orbital of a covalent bond [8]. The halogen “X” has an s 2p x 2p y 2p z 1electronic configuration where the RX bond is on the z ‐axis. In this electron configuration, two p‐orbitals are filled, and one is half filled, thus highlighting the depleted electron density in the p zorbital. This picture becomes more relevant with larger halogens and is more exaggerated when the halogen is covalently bound to an electron‐withdrawing system. For example, this orbital character does not appear for fluorine. As fluorine is very electronegative, it shares more of the sigma bonding electrons, creating a higher degree of sp hybridization than larger halogens. Moving additional electron density into the p zorbital affectively reduces the σ‐hole. For example, in a CF bond, 71.4% of electrons reside on F, whereas for less electronegative, larger halogens, like I, only ∼50% of the electron density resides on the halogen [8]. Meanwhile, the σ‐hole does not form for neutral, symmetric halogen containing molecules with equal electron distribution (e.g. carbon tetrahalides, hexahalobenzenes). This does not necessarily mean that symmetric or F‐based systems do not form halogen bonds; rather other attractive components become the dominate force.
1.4.5 Charge Transfer
Charge transfer has long been associated with halogen bonding, and Mulliken's investigations of I 2and organics containing O, S, or N heteroatoms are prime examples [42]. More recently, Palusiak utilized the Kohn–Sham molecular orbital (MO) theory to describe the interaction [148]. Halogen bonds and hydrogen bonds can have significant covalent character due to charge transfer from a guest to the antibonding σ* orbital (LUMO) of the R–X or R–H species [149] ( Figure 1.15). The lower‐energy σ* orbital and higher‐energy σ orbital in this halogen bonding example allow for increased orbital mixing (σ orbital mixing shown for R–X donor in Figure 1.15b). These charge‐transfer adducts often result in lengthening of the RX or RH bond, which was highlighted in an early theoretical study of halogen bonding complexes between dihalogens (including interhalogens) and Lewis bases [150]. Here, elongation of the halogen–halogen bond is largest in the strongest complexes, up to 0.065 Å in the FBr⋯NH 3complexes. The study also demonstrated that the most polarizable halogens, and the interhalogens (FBr, FCl, etc.) with the biggest dipole, resulted in the largest interaction energies. Other studies have revealed that charge transfer can be a significant factor in organic halogen bond systems as well. One example evaluated complexes of bromocarbons (e.g. CBr 3F, CBr 3NO 2, CBr 3COCBr 3, CBr 3CONH 2, Br 3CCN) with anions (Br –, N 3 –, NCO –, and NCS –) [151,152]. In these reports, increasing charge transfer was linearly correlated with elongation of the CBr bond length. Therefore, as the interaction strength with the Lewis base increases, the CBr bond lengthens, suggesting that the donation of electrons to the antibonding σ* from the p‐orbital of the Lewis base results in a weakening of the CBr bond. These conclusions are further supported by MO theory where charge‐transfer effects are the leading component for organohalogen halogen bond formation in H 3CX⋯OCH 2and F 3CX⋯OCH 2(X = Cl, Br, I) models [148].
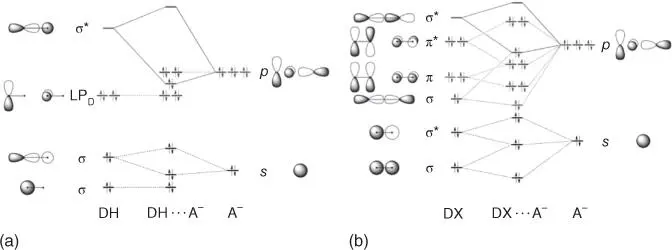
Figure 1.15Simplified orbital‐interaction diagrams for (a) hydrogen‐bonded complexes DH⋯A −and (b) halogen‐bonded complexes DX⋯A −as they emerge from quantitative Kohn–Sham MO analyses.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Halogen Bonding in Solution»
Представляем Вашему вниманию похожие книги на «Halogen Bonding in Solution» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Halogen Bonding in Solution» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












