Halogen Bonding in Solution
Здесь есть возможность читать онлайн «Halogen Bonding in Solution» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Halogen Bonding in Solution
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 100
- 1
- 2
- 3
- 4
- 5
Halogen Bonding in Solution: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Halogen Bonding in Solution»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Halogen Bonding in Solution
Halogen Bonding in Solution
Halogen Bonding in Solution — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Halogen Bonding in Solution», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
((b) Modified from Makhotkina et al. [105].)
1.3.4 Alternative Motifs and Solid‐state Reactivity
The use of alternative or less common designs in halogen bonding has been reviewed [104]. Some of the less common donors include N ‐iodoimides ( Figure 1.11a), which have been shown to be powerful halogen donors for a diverse range of acceptors [106-109]. In one example, Fourmigué and coworkers demonstrate that altering the donor and acceptor of these N ‐iodoimides can be used to demonstrate the cocrystal to salt continuum, a topic generally reserved for proton transfer between a hydrogen bond donor and acceptor. In the context of halogen bonding, it is iodine transfer that results in a salt ( Figure 1.11b) [105] and has also been the subject of a charge density analysis study [109]. Another alternative halogen bond is the three‐center‐four‐electron halogen bond of the type [NIN] +. These unique motifs are often compared with the low barrier hydrogen bonds of the type [NHN] +and are the subject of an ensuing chapter.
Halogen bonds have also been used to mediate crystalline state reactivity. The first example of a photomediated [2 + 2] olefin cycloaddition was presented by Metrangolo and coworkers [110]. Here, a tetratopic halogen bond donor arranged trans ‐1,2‐bis(4‐pyridyl)ethene for cycloaddition using CI⋯N halogen bonds ( Figure 1.12, top). Similar tactics by Sinnwell and MacGillivray highlighted the use of a ditopic halogen bond acceptor to arrange the olefin‐containing halogen bond donors, diiodooctafluorostilbene ( Figure 1.12, bottom) [112]. Once again, CI⋯N halogen bonds were employed to properly arrange the reactants. Only recently has a halogen bond cocrystal mediated a single‐crystal‐to‐single‐crystal transformation of an olefin cycloaddition [111]. The halogen bond has also been used to arrange polyacetylenes for polymerization. For example, the cocrystallization of 1,4‐diiodo‐1,3‐butadiyne with either dipyridine or dinitrile oxalamide derivatives produced 2D networks driven by both hydrogen bond and halogen bonds [113] ( Figure 1.13). The pyridine derivative only polymerized when subjugated to higher pressures, whereas the nitrile derivative polymerizes spontaneously at room temperature ( Figure 1.13). Solid‐state reactivity can also occur by mechanochemistry or solvent‐assisted grinding. For example, halogen bond‐mediated cocrystals were produced with mechanochemistry using 1,4‐diiodotetrafluorobenzene and 1,4‐dibromotetrafluorobenzene halogen bond donors and analyzed through powder diffraction and single‐crystal analysis [114].
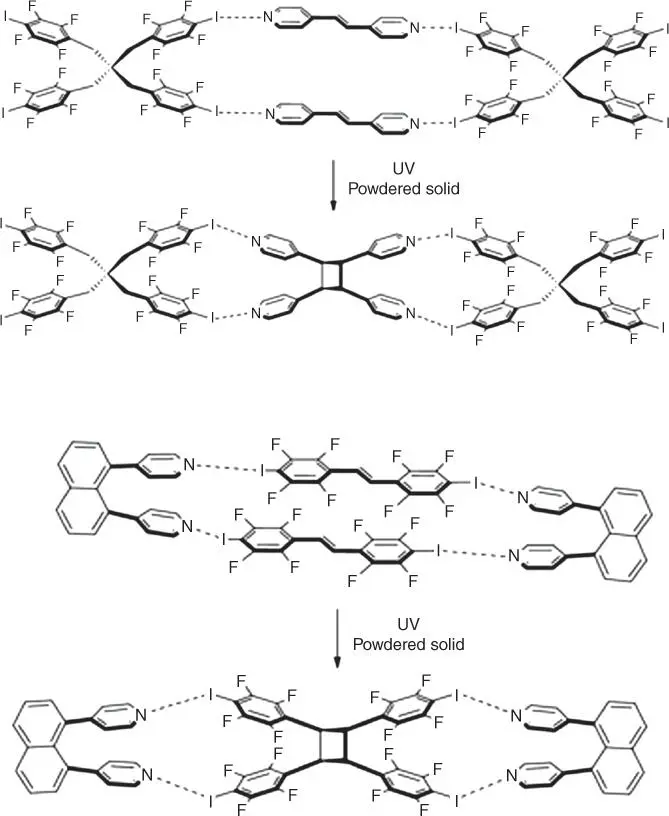
Figure 1.12Examples of halogen bond mediated [2 + 2] photodimerization of olefins in the solid state.
Source: From Sinnwell et al. [111]. Licensed under CC BY 2.0.
1.3.5 Crystallographic Studies Conclusion
Solid‐state evaluations of the halogen bond are vast, with numerous reviews written on the topic [7,58–65]. This section provided a topical survey highlighting some of the diversity within the field. However, one significant topic that was purposely omitted was halogen bonding to anions [115,116] as many of the later chapters include aspects of halogen bonding to anions in solution (e.g. quantification, receptors, transport, catalysis). Other solid‐state halogen bonding topics that have been omitted for brevity include solid‐state NMR [117,118], porous crystalline materials [119–121], crystalline rotors [122,123], polyhalides [124–126], cosublimation [127], energetic cocrystals [127], and intramolecular halogen bonding [128]. Looking forward, crystallography will continue to be an important research tool that complements studies of halogen bonding in solution.
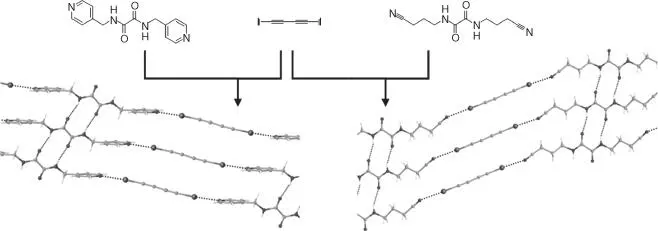
Figure 1.13Cocrystallization components and pre‐polymerization structures of 1,4‐diiodo‐1,3‐butadiyne with oxalamide derivatives. CCDC ref codes: WANNUV01 (left), CEKFUU (right).
1.4 Computational Studies
1.4.1 Introduction
Computational chemistry has proven valuable to understanding the fundamental nature of the halogen bond and frequently complements observed experimental data. Computational studies have shown that different components (e.g. charge transfer, electrostatics, dispersion) contribute to the interaction and that the relative makeup depends on the nature of the halogen bond donor (e.g. inorganic, organic, neutral, charged assisted) and acceptor (e.g. neutral, charged, soft or hard Lewis base). In this section, the forces contributing to the halogen bond interaction and an overview of in silico methods used to study the halogen bond will be surveyed. For an in‐depth look, reviews on computational halogen bonding theory in small molecule [8,129,130] and biological [131] systems have been published. Additionally, techniques to study the halogen bond (and other σ‐hole interactions) in silico have been reviewed by Kozuch and Bickelhaupt [132] and Hobza [133].
1.4.2 Electrostatics of the Halogen Bond and the σ‐Hole
One description of the halogen bond is rooted in the electron distribution of an isolated molecule within a ground state. As a polarizable halogen forms a covalent bond with an electron‐withdrawing group, a rearrangement of electrons results in electron‐rich and electron‐poor regions within the newly formed species. Consequently, the halogen adopts a spheroid shape, with the radius of the halogen extending from the covalent bond to the outer surface being smaller than the radius measured normal to the covalent bond ( Figure 1.1b). The term “polar flattening” is sometimes used to describe the oblate shape of the electron cloud resulting from the depletion of electronic charge at the end of the halogen [134] and has been demonstrated in a CSD study [135] as well as by experimental charge density analysis [136,137]. Polar flattening is not limited to halogens, but instead applies to all atoms covalently bound to another atom. Computationally mapping this distortion of electronic density has become a routine task and is achieved by measuring the ESP surface of a molecule. To better understand what ESP maps are depicting, it is necessary to outline their construction:
Equation 1.1Electrostatic potential.
(1.1) 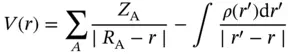
ESP is an application of Coulomb's law and is a physical property that can be determined experimentally by diffraction techniques or computationally [138]. Given an electron density function ρ ( r ′), V ( r ) is the ESP at any measured point r (Eq. (1.1)). Z Ais the charge on the nucleus that is located at R A. | R A− r | is the distance of the positive charge from r , and likewise | r ′ − r | is the distance of the electronic charge from r , where r ′ is the integration variable over all space. A positive V ( r ) indicates that effects by the nucleus are dominant or that the nucleus is not entirely shielded by the electron cloud. A negative V ( r ) indicates that the electron density, in the form of electron pairs, π‐bonds, etc., is dominant. ESP is frequently computed and viewed as a map covering the surface of a molecule. This surface is arbitrarily selected; however the most common surface to map is an outer contour of electron density, as it accurately encompasses lone pairs, strained bonds, and π‐electrons (Figures 1.2and 1.14). Typically, the ρ ( r ′) = 0.001 au (electrons/bohr 3) contour is used, but other similar contours at 0.0015 or 0.002 au will also achieve the same ends [138]. The ESP values along this surface are then set to a color gradient directly on the molecule in question, and the extremes are typically represented as blue and red (Figures 1.14and 1.2). While ρ ( r ′) and V ( r ) are in Eq. (1.1), there is a distinct difference between the values. ρ ( r ′) is dependent on only electrons, while V ( r ) incorporates contributions from all nuclei and electrons. As such, Politzer and Murray caution: It cannot be assumed that high (low) electronic densities correspond to negative (positive) electrostatic potentials. The potential in a given region is the net result of negative contributions from the electrons and positive ones from the nuclei of the entire molecule, their effects of course being greater as they are closer to the region in question [140]. In other words, ESP maps do not necessarily correlate with overall electron density.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Halogen Bonding in Solution»
Представляем Вашему вниманию похожие книги на «Halogen Bonding in Solution» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Halogen Bonding in Solution» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












