Zeolites appear to be promising supports for preparing SACs due to their well‐defined structures and high surface areas. Specifically, zeolites can provide effective voids to anchor individual metal atoms, thus maintaining the high dispersion of metals [42]. An atomically dispersed gold catalyst was prepared by the adsorption of an organogold precursor on zeolite NaY, and STEM images were used to determine the locations of isolated gold complexes in NaY [55]. However, this single‐atom Au(III)–O–NaY catalyst is unstable at 25 °C and 760 Torr in a flow reactor, losing ∼75% of their initial activity after 15 minutes in CO oxidation [55]. Flytzani‐Stephanopoulos and coworkers reported a successful approach to activate and stabilize atomic Au for the WGS reaction on zeolite and mesoporous silica ([Si]MCM‐41) materials with additional alkali ions [56].
Nitrides, carbons, and C 3N 4have also been used to fabricate SACs. For example, single‐atom Pt deposited on TiN was prepared using an incipient wetness impregnation method, and this catalyst was used for electrochemical oxygen reduction, formic acid oxidation, and methanol oxidation [57]. Chen et al. prepared an Au SAC supported on polymeric mesoporous graphitic C 3N 4by impregnation [58]. Wu and coworkers demonstrated an iced‐photochemical reduction strategy to construct atomically dispersed Pt species on mesoporous carbon [59]. Graphene can also be used as a support to obtain SACs. For instance, isolated Pt atoms were deposited on graphene nanosheets by atomic layer deposition (ALD), which involves graphite oxidation, thermal exfoliation, and chemical reduction [60]. As shown in Figure 6.6, firstly, the precursor MeCpPtMe 3was injected to react with the adsorbed oxygen on the surface of graphene nanosheets, forming CO 2, H 2O, and hydrocarbon fragments. The limited supply of surface oxygen provides the self‐limiting growth necessary for ALD, creating a Pt‐containing monolayer. The subsequent oxygen exposure forms a new adsorbed oxygen layer on the Pt surface. The two processes form a complete ALD cycle. The Pt deposition can be precisely controlled by tuning the number of ALD cycles [60].
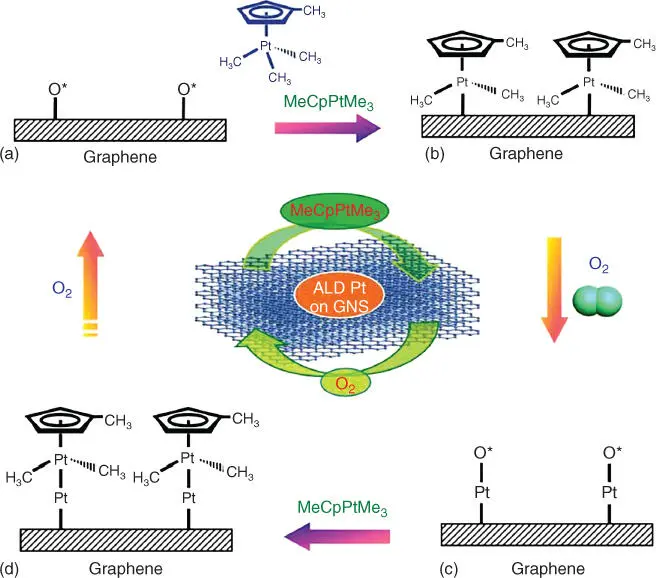
Figure 6.6 Schematic illustrations of Pt ALD mechanism on graphene nanosheets (GNSs).
Source: Sun et al. 2013 [60]. Reproduced with permission of Springer Nature.
(See online version for color figure).
The Galvanic Replacement MethodGalvanic replacement is an electrochemical process that involves the oxidation of one metal, termed as a sacrificial template, by the ions of another metal with a higher reduction potential. When they contact each other at a liquid/solid interface, the sacrificial template will be oxidized and dissolved into the solution, while the ions of the second metal will be reduced, and the second metal will be doped onto the template surface [61]. Galvanic replacement is highly effective and versatile for the generation of a wide variety of metal nanostructures, due to its abilities to control the size and shape and to tune the composition, internal structure, and morphology of the resultant nanostructures [50].
This method has been applied to synthesize SACs in recent years. Sykes and coworkers showed that low‐concentration isolated Pt atoms on Cu(111) surface can be prepared by galvanic replacement on pre‐reduced Cu NPs ( Figure 6.7a) [62]. They found that the isolated Pt atoms on Cu(111) surface can activate the dissociation and spillover of H to Cu. Sykes and coworkers also showed that such single‐atom Pt/Cu alloys can be used for C–H activation in methyl groups, methane, and butane [63]. An SAC consisting of isolated Rh atoms uniformly dispersed on the surface of VO 2nanorods can also be synthesized by this method. Simply, Na 3RhCl 6solution was injected into an aqueous solution containing VO 2nanorods, and the mixture was stirred at 300 rpm at room temperature for one hour. In this process, Rh 3+ions were reduced to Rh +by V 4+in the VO 2nanorods. Isolated Rh atoms were then uniformly dispersed on the surface of the VO 2nanorods, as seen by STEM ( Figure 6.7b) [36].
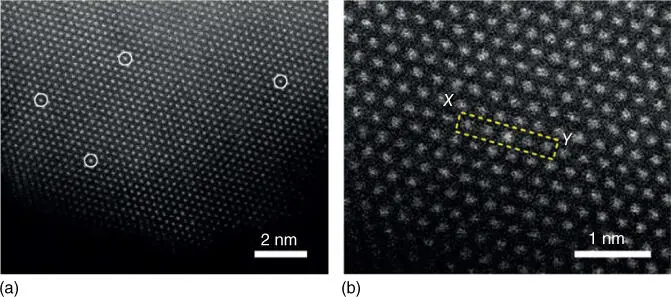
Figure 6.7 (a) HAADF‐STEM and (b) magnified HAADF‐STEM images of Rh 1/VO 2.
Source: Wang et al. 2017 [36]. Reproduced with permission of John Wiley & Sons.
6.3.2.2 Top‐Down Synthetic Methods
The top‐down strategy is based on turning ordered nanostructured into smaller pieces to give desirable properties and intriguing performance [50]. This strategy can also be used to synthesize SACs. A high‐temperature atomic migration method is a typical top‐down synthetic method to obtain SACs.
High temperatures are usually detrimental to catalysts' activities because SAs tend to move and agglomerate due to its high surface free energy. Ostwald ripening is one of the mechanisms to explain this process. The elemental steps of that process typically include the detachment of the metal atoms from smaller particles to form monomers, the diffusion of monomers on supports, and the attachment toward larger particles [64]. Tang and coworkers prepared single‐Ag‐atom catalysts, starting from Ag NPs supported on HMO [65]. Upon heating at 400 °C, the Ag NPs on HMO can be dispersed to form single Ag atoms, as evidenced by in situ transmission electron microscopy (TEM), in situ X‐ray diffraction (XRD), and ex situ X‐ray absorption near edge spectroscopy (XANES) ( Figure 6.8) [15, 65–67]. Datye and coworkers showed that at 800 °C in oxidizing ambient, Pt can transfer to ceria and be trapped there to form a sinter‐resistant, atomically dispersed catalyst [68]. Li and coworkers reported the successful reversion of sintering effects and the conversion of noble metal NPs into thermally stable and highly active SAs (Pd, Pt, Au) above 900 °C in an inert atmosphere by using nitrogen‐doped carbon derived from a metal–organic framework [69]. Such a high‐temperature atomic migration method requires a supply of mobile atoms and a support that can bind the mobile species. This method is potentially applicable for synthesizing high‐performance thermally stable SACs and reactivating sintered noble metal nanocatalysts.
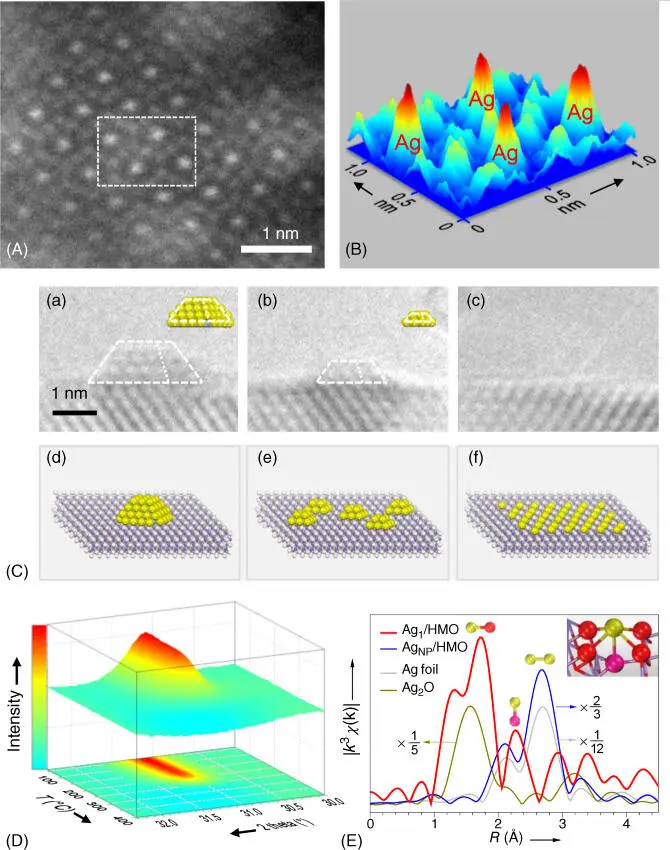
Figure 6.8 (A) HAADF‐STEM image of Ag 1/HMO. (B) Three‐dimensional projected image of the dash rectangle in (A). (C) The shrinking process of the supported Ag NPs heated by an electron beam. (D) Three‐dimensional and contour maps of in situ differential XRD patterns of Ag NP/HMO. (E) Ag K‐edge χ ( R ) k 3‐weighted FT EXAFS spectra of the samples.
Source: Adapted from Chen et al. 2015 [66] and Chen et al. 2016 [15].
(See online version for color figure).
6.4 Challenges and Perspective
SACs, as a kind of frontier catalysts, have attracted widespread attention and been extensively studied in recent years. Various methods have been developed to synthesize SACs, as summarized above. Specially, chemical methods are the most commonly used. Chemical methods can be categorized into two general approaches, i.e. bottom‐up and top‐down strategies. Although the top‐down strategy has provided a possibility to construct thermally stable SACs, it is still challenging to find practical methods for synthesizing stable SACs with high metal loadings.
Читать дальше




