69 69 Liu, Y., Tsunoyama, H., Akita, T., and Tsukuda, T. (2009). Preparation of ∼1 nm gold clusters confined within mesoporous silica and microwave‐assisted catalytic application for alcohol oxidation. J. Phys. Chem. C 113 (31): 13457–13461.
70 70 Liu, Y.M., Tsunoyama, H., Akita, T. et al. (2011). Aerobic oxidation of cyclohexane catalyzed by size‐controlled Au clusters on hydroxyapatite: size effect in the sub‐2 nm regime. ACS Catal. 1 (1): 2–6.
71 71 Azubel, M., Koh, A.L., Koyasu, K. et al. (2017). Structure determination of a water‐soluble 144‐gold atom particle at atomic resolution by aberration‐corrected electron microscopy. ACS Nano 11 (12): 11866–11871.
72 72 Al Qahtani, H.S., Kimoto, K., Bennett, T. et al. (2016). Atomically resolved structure of ligand‐protected Au9 clusters on TiO2 nanosheets using aberration‐corrected STEM. J. Chem. Phys. 144 (11): 114703.
73 73 Yamazoe, S., Yoskamtorn, T., Takano, S. et al. (2016). Controlled synthesis of carbon‐supported gold clusters for rational catalyst design. Chem. Rec. 16 (5): 2338–2348.
74 74 Donoeva, B.G., Ovoshchnikov, D.S., and Golovko, V.B. (2013). Establishing a Au nanoparticle size effect in the oxidation of cyclohexene using gradually changing Au catalysts. ACS Catal. 3 (12): 2986–2991.
75 75 Negishi, Y., Matsuura, Y., Tomizawa, R. et al. (2015). Controlled loading of small Aun clusters (n = 10–39) onto BaLa4Ti4O15 photocatalysts: toward an understanding of size effect of cocatalyst on water‐splitting photocatalytic activity. J. Phys. Chem. C 119 (20): 11224–11232.
76 76 Ovoshchnikov, D.S., Donoeva, B.G., Williamson, B.E., and Golovko, V.B. (2014). Tuning the selectivity of a supported gold catalyst in solvent‐ and radical initiator‐free aerobic oxidation of cyclohexene. Catal. Sci. Technol. 4 (3): 752–757.
77 77 Kurashige, W., Kumazawa, R., Ishii, D. et al. (2018). Au25‐loaded BaLa4Ti4O15 water‐splitting photocatalyst with enhanced activity and durability produced using new chromium oxide shell formation method. J. Phys. Chem. C 122 (25): 13669–13681.
78 78 Buchwalter, P., Rosé, J., and Braunstein, P. (2015). Multimetallic catalysis based on heterometallic complexes and clusters. Chem. Rev. 115 (1): 28–126.
79 79 Liu, L. and Corma, A. (2018). Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem. Rev. 118 (10): 4981–5079.
80 80 Zanella, R., Giorgio, S., Shin, C.‐H. et al. (2004). Characterization and reactivity in CO oxidation of gold nanoparticles supported on TiO2 prepared by deposition‐precipitation with NaOH and urea. J. Catal. 222 (2): 357–367.
81 81 Sachtler, W.M.H. and Zhang, C.Z. (2008). Metal clusters in zeolites. In: Handbook of Heterogeneous Catalysis, vol. 1 (eds. G. Ertl, H. Knözinger, F. Schüth and J. Weitkamp), 510–522. Wiley‐VCH.
82 82 Fortea‐pérez, F.R., Mon, M., Ferrando‐soria, J. et al. (2017). The MOF‐driven synthesis of supported palladium clusters with catalytic activity for carbene‐mediated chemistry. Nat. Mater. 16 (7): 760–766.
83 83 Grundner, S., Markovits, M.A.C., Li, G. et al. (2015). Single‐site trinuclear copper oxygen clusters in mordenite for selective conversion of methane to methanol. Nat. Commun. 6: 7546.
84 84 Vogiatzis, K.D., Li, G., Hensen, E.J.M. et al. (2017). Electronic structure of the [Cu3(μ‐O)3]2+ cluster in mordenite zeolite and its effects on the methane to methanol oxidation. J. Phys. Chem. C 121 (40): 22295–22302.
85 85 Tomkins, P., Mansouri, A., Bozbag, S.E. et al. (2016). Isothermal cyclic conversion of methane into methanol over copper‐exchanged zeolite at low temperature. Angew. Chem. Int. Ed. 55 (18): 5467–5471.
86 86 Narsimhan, K., Iyoki, K., Dinh, K., and Román‐Leshkov, Y. (2016). Catalytic oxidation of methane into methanol over copper‐exchanged zeolites with oxygen at low temperature. ACS Cent. Sci. 2 (6): 424–429.
87 87 Liu, L., Zakharov, D.N., Arenal, R. et al. (2018). Evolution and stabilization of subnanometric metal species in confined space by in situ TEM. Nat. Commun. 9 (1): 574.
88 88 Carvill, B.T., Lerner, B.A., Adelman, B.J. et al. (1993). Increased catalytic activity caused by local destruction of linear zeolite channels: effect of reduction temperature on heptane conversion over platinum supported in H‐mordenite. J. Catal. 144 (1): 1–8.
6 Single‐Atom Heterogeneous Catalysts
Yaxin Chen, Zhen Ma, and Xingfu Tang
Fudan University, Department of Environmental Science & Engineering, 2005 Songhu Rd., Shanghai, 200438, PR China
Supported noble metal catalysts have been widely used in the production of chemicals [1], the remediation of environmental pollution [2, 3], and the processing of fuels [4, 5]. However, it is still desirable to increase the efficiency of supported noble metal catalysts due to the high prices and limited supplies of noble metals. To achieve this goal, one may attempt to design better supported catalysts by decreasing the content of the noble metals while maintaining their high catalytic efficiency. Thus, the sustainability of related industries can be improved, and the costs of production can be minimized.
Liang et al. gave an example to illustrate the advantage of supported noble metal catalysts over bulk metals ( Figure 6.1) [6]. Assuming that the price of pure gold is US$ 38.1 g −1, a gold brick with the dimensions of 20 × 10 × 5 cm 3is worth approximately US$ 736 000. For comparison, if a common brick (20 cents) with the same dimensions is coated with an atomic ultrathin layer of gold (1 cent), the total value of the brick is only 21 cents (if the cost of coating processing is not counted), much lower than that of a gold brick. Thus, dispersing noble metals as ultrafine nanoparticles (NPs), clusters, or single atoms (SAs) on solid supports is effective for the efficient use of noble metals and the minimization of catalysts' costs. Then what is the effect of downsizing metal NPs on the catalytic activity of supported metal catalysts?
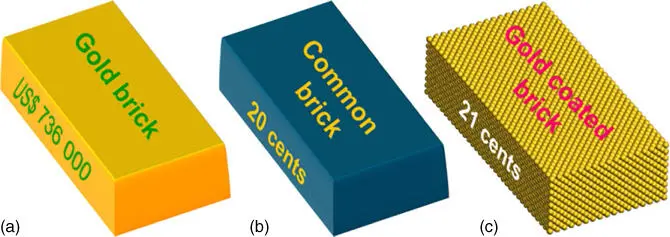
Figure 6.1 Prices of different bricks with a size of 20 × 10 × 5 cm 3. (a) A gold brick, (b) a common brick, and (c) a common brick coated with a single atomic layer of gold atoms.
Source: Liang et al. 2015 [6]. Reproduced with permission of John Wiley & Sons. (See online version for color figure).
Catalytically active sites (CASs) are the atoms where the chemical reaction actually occurs. This concept was introduced by Taylor [7]. Although the precise identification of CASs of supported metal catalysts is very difficult due to the quantum size effect [8–10] and the structure‐sensitive geometric effect [11, 12], CASs are generally the surface atoms in an unsaturated coordination environment [13–15]. When downsizing metal NPs, the number of surface atoms increases substantially, and the metal NPs expose more defects and active sites, thus leading to higher catalytic activity [13–15]. Forming SAs on the support is the most efficient approach to utilize metal atoms in a supported metal catalyst.
Single‐atom catalysts (SACs) are catalysts in which the active metal species either exist as isolated SAs stabilized by the support or exist by alloying with another metal [16–18]. Since the concept of SACs was proposed, research on SACs has progressed rapidly to obtain a better understanding of sample preparation and characterization, the role of support, the strong metal–support interactions, and the catalytic mechanisms. Herein, we review some of the recent research on SACs, focusing on various preparation methods. Future challenges and opportunities are also discussed.
Читать дальше


