SACs have attracted much attention in the catalysis community due to the maximum atom use efficiency and unique catalytic properties. However, due to the excess surface free energy of SAs, SAs tend to aggregate into larger particles at elevated temperatures or during catalytic reactions [38, 43–45], resulting in a decrease or even complete loss of catalytic activity. Thus, it is challenging to fabricate SACs with high‐loading SAs dispersed finely and densely. Significant progress has been made in recent years to develop various methods for the synthesis of SACs. Here we summarize a few common methods that can be divided into two categories, i.e. physical and chemical methods. The chemical method can also be categorized into two types: bottom‐up and top‐down methods.
Physical synthesis of SACs can be realized through high vacuum physical deposition. For example, mass‐selected soft landing involves the use of a gas‐phase cluster as an ion source and a downstream mass spectrometer to mass‐select nanoclusters prior to their deposition onto a substrate ( Figure 6.4) [18, 47]. In this case, metal clusters with precisely defined number of atoms can be produced and “soft‐landed” onto the surface of a desired substrate. This approach allows for independent control of cluster size and coverage and, in principle, can be used for any combination of cluster and flat support. Thus, this method can provide excellent model catalysts for fundamental research of active sites, metal–support interactions, and cluster size effects [21, 46–49]. Anderson and coworkers deposited Pd clusters (Pd n, n = 1, 2, 4, 7, 10, 16, 20, and 25) on clean, vacuum‐annealed rutile TiO 2(110) to find out the correlation between the catalytic activity and the electronic structure of Pd clusters that varied with cluster size [48]. Supported size‐selected Pd nclusters were also tested in acetylene cyclotrimerization, and a single Pd atom adsorbed on MgO was found to be enough for the production of benzene at 300 K [21]. However, the disadvantage of this method is that the cost is high and the yield to desired catalysts is low, which makes it unsuitable for practical industrial applications. Moreover, this method is not suitable for making catalysts using high surface area or mesoporous supports, and it is difficult to achieve high metal loadings using this method. In all, this method is merely useful for making model catalysts for fundamental research.
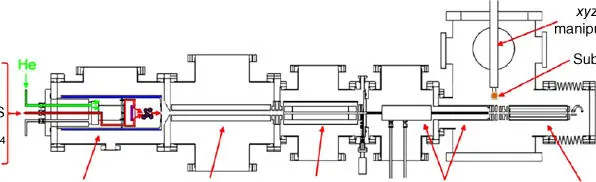
Figure 6.4 Schematic drawing of size‐selected cluster deposition apparatus at Brookhaven National Laboratory.
Source: Vajda and White 2015 [46]. Reproduced with permission of American Chemical Society.
Chemical methods are more common and can be routinely practiced. Chemical methods can be categorized according to how their components are integrated, namely, via bottom‐up or top‐down approaches. For bottom‐up strategy, single metal atom species (the metal precursors) are directly anchored to the support by a coordination effect between the metal complexes and the anchoring sites on the support surfaces [50]. For top‐down strategy, the metal NPs are directly introduced onto the support surface and then dispersed into SAs to form SACs.
6.3.2.1 Bottom‐Up Synthetic Methods
The bottom‐up strategy, including coprecipitation, adsorption, and galvanic replacement methods, is the most common strategy to synthesize SACs. Firstly, mononuclear metal precursors are introduced onto the support surface. Then the product was dried and calcinated to remove organic ligands of the metal complexes. Finally, SACs are produced by reduction or activation [19].
Coprecipitation MethodCoprecipitation seems to be the simplest method to prepare SACs. The precursors of the metal and the support should be soluble and could be coprecipitated by the precipitant at a certain pH value. The characteristics of the final catalysts, however, depend on many parameters including the order and the speed of the addition of the component solutions, the size of the droplets, efficient mixing, the temperature of the base solution, the pH value, and the aging time. Zhang and coworkers employed this method to fabricate single Pt atoms supported in iron oxide nanocrystallites (Pt 1/FeO x), as demonstrated by aberration‐corrected scanning transmission electron microscopy (AC‐STEM) and extended X‐ray absorption fine structure (EXAFS) spectra ( Figure 6.5) [16]. A series of SACs, M/TiO 2(M = Pt, Pd, Rh, or Ru), were also synthesized by this method [24]. The SACs exhibit high photocatalytic hydrogen evolution performance compared with metal NPs. However, the main disadvantage of this approach is that some metal atoms can be buried at the interfacial regions of the support agglomerates and within the bulk of the support crystallites, thus compromising the effectiveness and efficiency of the SACs.
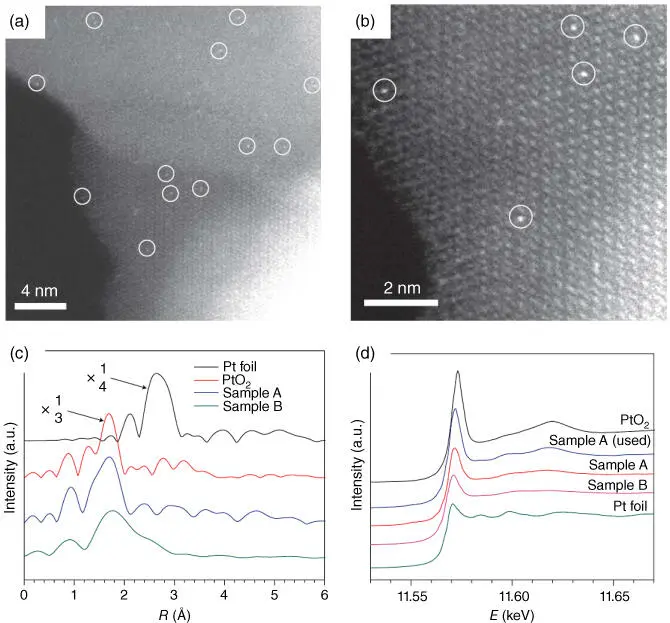
Figure 6.5 (a, b) HAADF‐STEM images of Pt 1/FeO x. (c) The k 3‐weighted Fourier transform spectra from EXAFS. (d) The normalized XANES spectra at the Pt L 3edge of samples. Sample A refers to Pt 1/FeO xwith a Pt loading of 0.17%. Sample B refers to a similar catalyst with a Pt loading of 2.5 wt%.
Source: Qiao et al. 2011 [16]. Reproduced with permission of Springer Nature.
(See online version for color figure).
Adsorption MethodThe adsorption method, including impregnation, electrostatic adsorption, and adsorption assisted by other techniques, can also be used to synthesize SACs [50]. Generally, after the metal precursors are adsorbed on the support, the residual solution is removed, and the samples are then dried and calcined. The final metal loading and dispersion depend strongly on the nature of the anchoring sites on the support surfaces. This point should be considered for sample preparation.
Oxide supports are commonly used for preparing catalysts. For example, single Pt atoms were supported on θ‐Al 2O 3(010) surface by a wet impregnation method using alumina powder and chloroplatinic acid [38]. An impregnation–reduction method involves the impregnation of metal cations on oxides followed by on‐site reduction of metal cations, as exemplified by the preparation of singly dispersed Rh atoms supported on Co 3O 4[51]. Gu et al. reported that single Pt 1and Au 1atoms can be stabilized by lattice oxygen on ZnO{1010} surface via an adsorption method [52]. High‐energy bottom‐up ball‐milling synthesis, a powerful method to break and reconstruct chemical bonds of materials with high efficiency, was used to synthesize a lattice‐confined single iron site catalyst embedded within a silica matrix [41, 53]. Liu et al. developed a room‐temperature photochemical strategy to fabricate stable atomically dispersed palladium–titanium oxide catalyst (Pd 1/TiO 2) with a Pd loading up to 1.5% [54]. Generally, the support, two‐atom‐thick TiO 2(B) nanosheets, was prepared by reacting TiCl 4with ethylene glycolate. H 2PdCl 4was added into a water dispersion of TiO 2(B) for the adsorption of Pd species. Then the mixture was irradiated by ultraviolet (UV) light to give a Pd 1/TiO 2catalyst. However, metal oxide supports are not ideal for making supported catalysts used in electrocatalysis because metal oxides are generally insulators or semiconductors with low electron conductivities and they are often unstable under corrosive electrochemical conditions.
Читать дальше



