Yu Lan - Computational Methods in Organometallic Catalysis
Здесь есть возможность читать онлайн «Yu Lan - Computational Methods in Organometallic Catalysis» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Computational Methods in Organometallic Catalysis
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:4 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 80
- 1
- 2
- 3
- 4
- 5
Computational Methods in Organometallic Catalysis: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Computational Methods in Organometallic Catalysis»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Computational Methods in Organometallic Catalysis — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Computational Methods in Organometallic Catalysis», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
1.2 Using Computational Tool to Study the Organometallic Chemistry Mechanism
Transition metal catalysis is one of the most powerful tools for the construction of new organic materials, whose development trend is more efficient as well as more complex. Therefore, studying the mechanism of organometallic catalysis has become even more essential, and has proved to be the basis for the design of new ligands, catalysts, and reactions.
1.2.1 Mechanism of Transition Metal Catalysis
Generally, reaction mechanism could be considered to be all elementary reactions used to describe a chemical change passing in a reaction. It is to decompose a complex reaction into several elementary reactions and then combine them according to certain rules, so as to expound the internal relations of complex reactions and the internal relations between total reactions and elementary reactions. The rate of chemical reaction is closely related to the specific pathways through which the reaction takes place.
To study the law of chemical reaction rate and find out the intrinsic causes of various chemical reaction rates, synthetic chemists must explore the reaction mechanism and find out the key to determine the reaction rate, so as to control the chemical reaction rate more effectively. As shown in Scheme 1.4, traditional research methods for reaction mechanism include: (i) determining the important intermediate or decisive step of a reaction by isotope tracing, (ii) determining the effect of different factors (e.g. reaction temperature, solvent, substituent effect, etc.) on reaction rate and selectivity by competitive test, (iii) studying the relationship between the reaction rate and the concentration of reactants and catalysts obtaining by kinetic experiments, and (iv) characterizing and tracking intermediates by instrumental analysis. However, these methods are often macroscopic observation of the average state of many molecules, which cannot watch a process of the transformation for one molecule from a micro‐perspective. Fortunately, theoretical calculations based on first principles have become one of the important means to study the reaction mechanism with the development of software and the improvement of hardware computing capability in recent several decades. Through theoretical calculation and simulation, the transformation of one molecule in reaction process can be “watched” more clearly from the microscopic point of view. Actually, theoretical calculation can be considered to be a special kind of microscope, which can see the geometrical structure, electronic structure, spectrum, and dynamic process at atomic level, and is helpful for chemists to understand the real reaction mechanism.
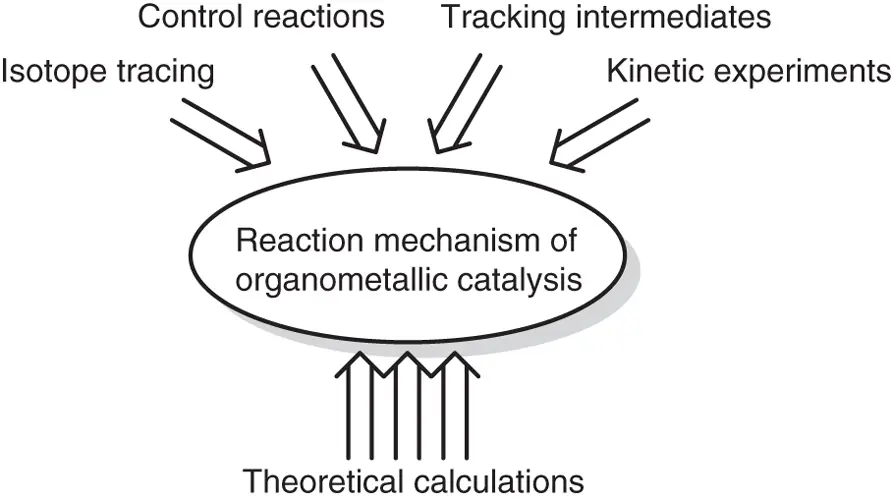
Scheme 1.4 Revealing the reaction mechanism of organometallic catalysis.
The combination of theoretical and experimental techniques could not only greatly improve the efficiency of reaction and yield of product, but also uncover the factors that control the selectivity of product more clearly. The promotion of theoretical study to experimental investigation could be summarized into “3D,” i.e. description, design, and direction. Based on the data obtained from experimental technique, detailed description for the mechanism of organometallic catalysis could be fulfilled using theoretical calculations. Based on the results of computations, the mechanisms could be verified by the designed experiment. To put in a nutshell, theoretical calculations could play a critical role in the direction of transition‐metal‐organic synthesis.
1.2.2 Mechanistic Study of Transition Metal Catalysis by Theoretical Methods
Quantum chemical computation based on first principle provides a powerful tool for the mechanistic study of transition metal catalysis. Since the whole content of this book is to discuss the theoretical calculation‐based study for the mechanism of transition metal catalysis, we will give only a few examples to show how to study the reaction mechanism by theoretical calculations.
Generally, mechanism research of transition metal catalysis initially faces a series of studies involving the molecular structure and electronic states. As an example, (Xantphos)Pd(CH 2NBn 2) +is an important precursor for aminomethylation reactions, the geometric structure of which has been confirmed by X‐ray analysis [87]. However, why this complex could be formed and the electronic properties of this complex still remained unclear. As shown in Scheme 1.5, in resonance structure 1‐1, the Pd—C bond is a normal single bond, and Pd—N is a coordination bond. The formal positive charge is localized on palladium, and the formal oxidation state of palladium is +2. Alternatively, the iminium moiety acts as a monodentate ligand coordinated with Pd(0) in resonance structure 1‐2, and the formal positive charge is mainly localized on the iminium moiety. The real structure of this complex would be a mixture of resonance structures 1‐1and 1‐2. On the other hand, the bond orders of Pd—C, Pd—N, and C—N are determined to be 0.322, 0.135, and 0.965, respectively, which indicate that the Pd—C and Pd—N bonds are very weak. More importantly, these data support that the C—N is a double bond and 1‐2is most likely to be the main structure of this complex. Further frontier molecular orbital studies also supported this point.
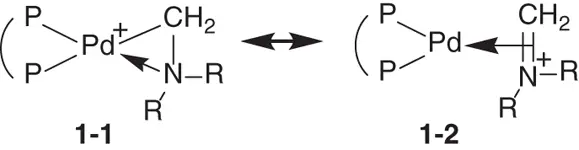
Scheme 1.5 The resonance structures of (Xantphos)Pd(CH 2NBn 2) +.
To summarize, computational organometallic chemistry focuses on some of the stationary points on potential energy surface for the corresponding reactions, which could be used to compare the possible elementary reactions. The lowest energy reaction pathway could be found by theoretical calculations, which is helpful for chemists to understand the reaction mechanism and design new reactions.
As an example, computational method was used to study the mechanism of rhodium‐catalyzed coupling reaction of quinoline N ‐oxide and acetylenes. As shown in Scheme 1.6, four possible pathways were taken into account (i.e. paths A–D) [88]. All these four pathways begin with the coordination of quinoline N ‐oxide to Rh(III) precursor 1‐6, which is followed by an N ‐oxide‐directed electrophilic deprotonation by acetate and coordination of acetylene substrate to give rhodacycle 1‐7. The subsequent insertion of the acetylene substrate into the Rh—C bond of intermediate 1‐7gives intermediate 1‐8, which is a common intermediate for both paths A and C. In path A, complex 1‐8is proposed to undergo a reductive elimination reaction to form a new C—O bond in π‐coordinated complex 1‐9. The N—O bond in complex 1‐9would then oxidatively add back to Rh(I) to generate Rh(III) enolate intermediate 1‐10. Protonolysis of intermediate 1‐10by acetic acid would release one molecule of product 1‐5and regenerate complex 1‐6. Product 1‐5could also be generated by the protonation of intermediate 1‐14, which is an isomer of intermediate 1‐10. In path C, the migration of the oxygen atom from complex 1‐8would lead to the formation of Rh(V)–oxo intermediate 1‐11. A subsequent oxygen insertion into Rh—C(vinyl) bond may give rise to intermediate 1‐10, which is the common intermediate in path A. Alternatively, the insertion of the acetylene substrate into the Rh—O bond of intermediate 1‐7would give rise to a common intermediate 1‐12, which could proceed along path B or path D. For path B, cleavage of the N—O bond would lead to the formation of α ‐oxo carbenoid species 1‐13, which could undergo carbene insertion to give intermediate 1‐14. This intermediate is either protonated directly or isomerizes to intermediate 1‐10, followed by protonolysis to afford product 1‐5together with the regeneration of active catalyst 1‐6. Intermediate 1‐12could also undergo reductive elimination through path D to give complex 1‐10. All four of these different pathways have been evaluated in the current study by using theoretical calculations. The computational results revealed that path A is the most favorable process for this reaction.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Computational Methods in Organometallic Catalysis»
Представляем Вашему вниманию похожие книги на «Computational Methods in Organometallic Catalysis» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Computational Methods in Organometallic Catalysis» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.