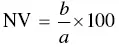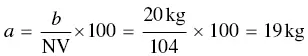2.2.2 Neutralizing Values
Neutralizing values (NV) of leavening acids are used to compare the available acidity of the acids and to calculate the amount of acid needed in a given formulation of chemically leavened product. Neutralizing value is defined as the weight of NaHCO 3that can be neutralized by 100 parts of the leavening acid. For example, if 100 g of an acid will neutralize 50 g of NaHCO 3, the neutralizing value of the acid is 50. NV are determined by titrating to a specific pH endpoint [6]. The formula for NV is:
(7) 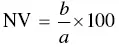
whereb = wt of NaHCO3 neutralized;
a = wt of acid required to neutralize b.
See Table 2.1for NV for some common leavening acids.
Table 2.1 Neutralizing values for some common leavening acids a.
| Leavening acid |
Molecular weight |
Neutralizing value |
Reaction rate b |
| Monocalcium phosphate monohydrate (MCP) |
252 |
80 |
Fast |
| Delayed anhydrous monocalcium phosphate (AMCP) |
234 |
82 |
Slow |
| Sodium acid pyrophosphate (SAPP) |
222 |
72 |
Slow |
| Sodium aluminum phosphate (SALP) |
950 |
100 |
Slow |
| Dicalcium phosphate dihydrate (DCP) |
172 |
33 |
Slow |
| Sodium aluminum sulfate (SAS) |
484 |
104 |
Slow |
| Fumaric acid |
116 |
145 |
Fast |
| Potassium acid tartrate |
188 |
50 |
Medium |
| Glucono‐delta‐lactone |
178 |
45 |
Slow |
aAdapted from [4] and [7].
bRate of reaction with NaHCO 3at room temperature.
NV are used to determine the amount of acid needed to neutralize a given amount of NaHCO 3. For example, if a formula specifies 20 kg of NaHCO 3, the amount of SAS (NV = 104) required would be:
(8) 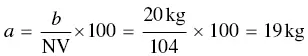
Baking powders and recipes are generally formulated to give baked products with near‐neutral pHs. There are, however, some exceptions to this:
1 Devil's Food Chocolate Cake. Excess sodium bicarbonate is added to give an alkaline pH in order for the chocolate to form the characteristic deep red color.
2 Buttermilk products. Excess acid (in the form of buttermilk) is added to give the characteristic flavor.
3 Pretzels and Gingerbread. Alkaline pHs are produced to accelerate nonenzymatic browning reactions which are important for the deep brown color.
The rates at which doughs are leavened are important determinants of the quality of baked products. Leavening rates in dough systems are influenced by a variety of factors including the types and concentrations of the leavening agents, temperature, the availability of water, and pressure [8].
Leavening rates may be measured by trapping evolved CO 2and plotting CO 2volume versus time. The amount of CO 2produced is usually expressed as a percentage of the total amount of CO 2that would be released if all of the sodium bicarbonate were converted to CO 2and H 2O. When measured in a dough system, leavening rates are called “dough reaction rates.” Typical dough reaction rates for some leavening acids are shown in Figure 2.1.
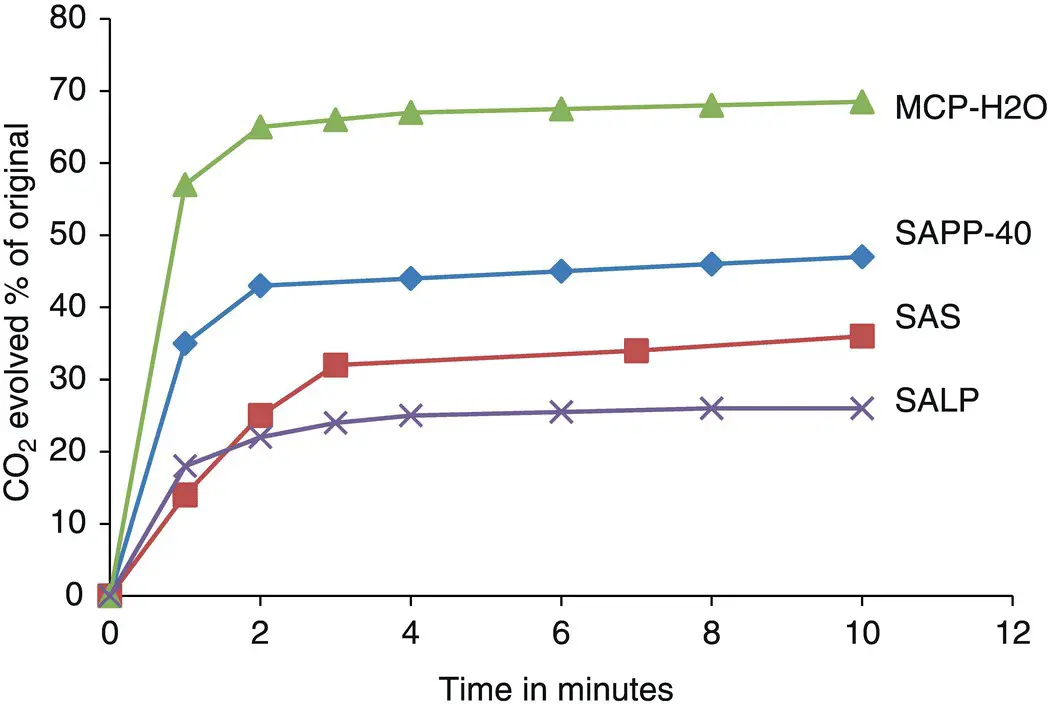
Figure 2.1 Rates of CO 2production from mixtures of SALP and MCP‐H 2O, SAPP‐40, or SAS. Evolved CO 2is expressed as a percentage of “available” CO 2in the NaHCO 3.
Source: Adapted from [9].
Measurement of leavening rates must be carefully standardized with respect to temperature, time, agitation, and ingredients so that comparisons between laboratories and experiments will be valid [10]. The examples of leavening rates given above serve to illustrate the fact that rates differ considerably among leavening systems. It should be remembered that leavening rates for a given leavening acid can be manipulated by applying coatings and changing particle size. Also, other ingredients in the batter or dough may affect leavening rates. Thus, when purchasing leavening acids from suppliers, it is important to specify the leavening rate needed for a particular application.
2.2.4 Effect of Leavening Acid on Dough Rheology
Cations and anions from chemical leaveners may alter the elastic and viscous properties of the dough and the texture and resiliency of the crumb. Calcium and aluminum ions can prevent coalescence of air bubbles into larger cells so that the structure of the finished product remains fine. Thus, two leavening acids with similar leavening rates may produce products with different textures.
2.3 Apparatus and Instruments
1 Filter flask, 125 ml
2 Rubber tubing
3 Rubber stopper
4 Graduated cylinders, 100 and 500 ml
5 Ring stand with holder
6 Magnetic stirrer and stir bars
7 Shallow pan
8 pH meter and pH standard buffers
9 Baking oven set at 218 °C (420 °F)
10 Water bath, 60–70 °C
11 Burettes
12 Electric mixer
13 Thermometer
2.4 Reagents and Materials
1 Sodium bicarbonate
2 Monocalcium phosphate monohydrate (MCP)
3 Sodium aluminum sulfate (SAS)
4 Commercial double‐acting baking powder
5 All‐purpose flour
6 Sugar
7 Salt
8 Skim milk
9 Vegetable oil
10 Millet seeds
2.5 Procedures
2.5.1 Determination of Leavening Rates
2.5.1.1 The Apparatus
Set up CO 2measuring apparatus as shown in Figure 2.2.
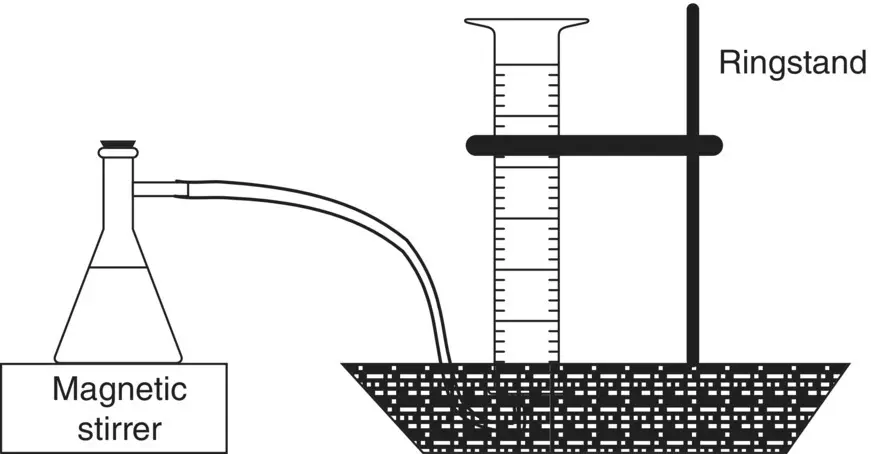
Figure 2.2 Apparatus for collecting and quantifying the volume of gas released.
2.5.1.2 Experimental Treatments and Controls
Note: When there is more than one ingredient, mix them together thoroughly before adding to the flask.
1 Control (no leavening agent)
2 Sodium bicarbonate (0.34 g)
3 Sodium bicarbonate (0.34 g) + MCP‐H2O (0.42 g)
4 Sodium bicarbonate (0.34 g) + SAS (0.33 g)
5 Baking powder (1.21 g). Note: this assumes that baking powder is 28% NaHCO3.
Note: This is a standard protocol for measuring leavening rates in a model system.
1 Add 60 ml distilled water and a magnetic stir bar to a filter flask.
2 Fill a 100 ml graduated cylinder with tap water and invert it in a pan of water as shown in Figure 2.2(take care to avoid allowing air to get into the cylinder).
3 Weigh out the specified amounts of each of the leavening agents (see Section 2.5.1.2).
4 With the stirrer running on low speed, transfer the leavening agent to the flask and stopper immediately. (Note: The rate of stirring will affect the rate of the reaction, so always use the same setting on the stirrer.)
5 Record the volume of displaced water in the cylinder every 30 seconds for five minutes.
6 Now, set the flask in a water bath held at 65 °C and continue recording the volume for an additional five minutes. (Note: Temperature can have a marked effect on CO2 generation, so regulate the temperature carefully.)
Читать дальше