Dennis D. Miller - Food Chemistry
Здесь есть возможность читать онлайн «Dennis D. Miller - Food Chemistry» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Food Chemistry
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:3 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 60
- 1
- 2
- 3
- 4
- 5
Food Chemistry: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Food Chemistry»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
A manual designed for Food Chemistry Laboratory courses that meet Institute of Food Technologists undergraduate education standards for degrees in Food Science Food Chemistry: A Laboratory Manual
Food Chemistry: A Laboratory Manual
Food Chemistry — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Food Chemistry», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
In the food industry, food additives that have acidic properties are commonly known as food acidulants. There are many approved food acidulants, but only a few are in wide use. They include organic acids like acetic acid, citric acid, fumaric acid, lactic acid, malic acid, and tartaric acid as well as the mineral acids phosphoric acid and sodium hydrogen sulfate. (See [2] for guidance in selecting food acidulants.)
1.2.1.2 Reactions of Food Acids
Most naturally occurring food acids are carboxylic acids. Carboxylic acids are weak acids compared with mineral acids such as HCl and H 2SO 4. Important reactions of carboxylic acids include the following:
Ionization:
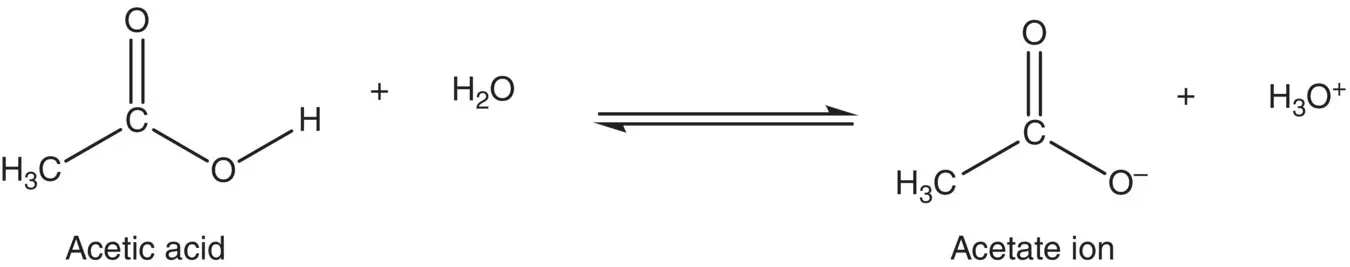
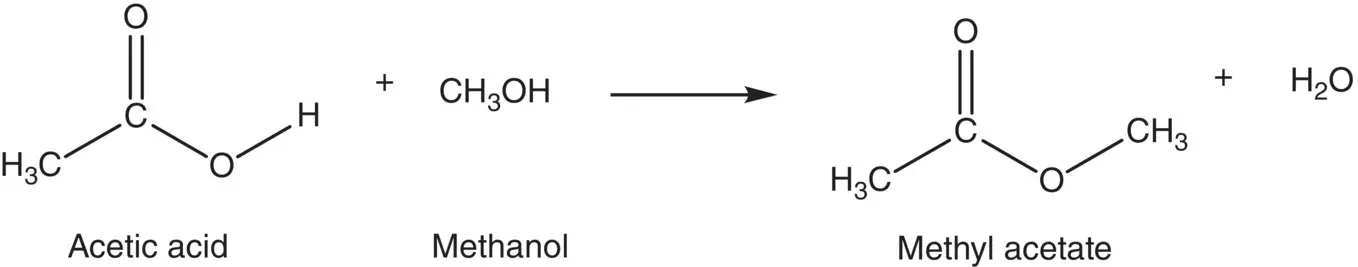
Reaction with alcohols to form esters:
1.2.2 Bases
Bases are also common food additives and are added for a variety of purposes. They may be added to modify the flavor, color, and texture, enhance browning, induce chemical peeling, and produce CO 2. Examples of bases used as food additives include dilute NaOH (to induce chemical peeling in fruits and vegetables, enhance browning, de‐bitter olives, solubilize proteins), phosphate salts (to prevent protein coagulation in evaporated and condensed milks, produce a smooth texture in processed cheese), and NaHCO 3(to give chocolate a darker color, produce CO 2in leavening systems).
1.2.3 Buffers
Buffers stabilize the pH in foods. They are also used to neutralize foods which are too acidic. By using the salt of the acid already present, acidity is reduced without adding neutralization flavors. Many buffers are present naturally in foods. Animal products are usually buffered by amino acids, proteins, and phosphate salts. In plants, organic acids (such as citric, malic, oxalic, and tartaric) in conjunction with phosphate salts are the primary buffers. Table 1.2shows the pHs of some common foods. Notice that most foods are buffered in the acidic range (pH < 7).
Table 1.2 Approximate pH values for some common foods a.
| Food | pH | Food | pH |
|---|---|---|---|
| Lime juice | 2.0 | Yogurt | 4.0–4.5 |
| Lemon juice | 2.2 | Cheddar cheese | 5.1–5.5 |
| Vinegar | 2.6 | Beef, fresh | 5.5–5.0 |
| Rhubarb | 3.0 | Pork, fresh | 5.6–6.9 |
| Grape juice | 3.1–3.2 | Turkey, fresh | 5.7–6.1 |
| Wines | 2.9–3.9 | Tuna | 6.0 |
| Apple juice | 3.5–3.9 | Carrots, fresh | 5.7–6.1 |
| Strawberries | 3.2–3.4 | Potatoes, fresh | 6.1 |
| Peaches | 3.8 | Green beans, fresh | 6.5–6.7 |
| Pears | 3.9 | Milk, fresh | 6.6 |
| Grapefruit juice | 4.0 | Sweet corn, fresh | 6.7 |
| Orange juice | 4.2 | Egg yolk | 6.0–6.9 |
| Tomato juice | 3.8–4.7 | Egg white (pH increases as egg ages) | 7.6–9.2 |
aModified from [3] and [4].
See Appendix IIIor your chemistry and biochemistry textbooks for a review of acid and base chemistry.
1.3 Apparatus and Instruments
1 pH meter equipped with a pH electrode
2 Analytical balance
3 Household blender
4 Centrifuge
5 Centrifuge tubes
6 Pipette and pipette bulb, 10 ml
7 Volumetric flask, 200 ml
8 Beakers, 150 ml
9 Burette, 25 or 50 ml
10 Burette holder and stand
11 Thermometer
12 Funnel
13 Graduated cylinder, 100 ml
14 Squeeze bottle for deionized water
15 Tissue
16 Weighing paper
17 Spatula
18 Stirring hot plate with stirring bars
1.4 Reagents and Materials
1 Citric acid, monohydrate. MW = 210 g mole−1
2 KOH, 0.5 N
3 HCl, 0.5 N
4 HCl, 0.001 N
5 Sprite® (Coca Cola Company) or comparable lemon‐flavored soda
6 Selected vegetables, e.g. fresh and canned tomatoes
7 Calibration buffers, pH 2 and 4
1.5 Procedures
1.5.1 Determining the pH of a Solid Food [5]
1 Cut a fresh tomato into small cubes and blend in a blender until a uniform slurry is formed, measure the temperature of the slurry.
2 Calibrate your pH meter.
3 Measure the pH of the slurry.
4 Centrifuge an aliquot of the slurry for 10 minutes at maximum speed.
5 Measure the pH of the supernatant.
6 Repeat Steps 1 through 5 using canned tomatoes.
1.5.2 Preparation of a Buffer and Determination of Buffer Capacity
1 Calculate the amounts of citric acid monohydrate and 0.5 N KOH required to prepare 200 ml of 0.05 M citrate buffer, pH 3. Note: Even though citric acid is a triprotic acid, calculations for this pH range are made using pKa = 3.06.
2 Prepare 200 ml of the buffer.
3 Measure the pH of your buffer. Is it 3.0? If not, can you explain why?
4 Determine the buffer capacity of your buffer in the alkaline direction by titrating a 100 ml aliquot with 0.5 N KOH. Express buffer capacity as the number of moles of OH− required to increase the pH of 1 l of the buffer by 1 pH unit.
5 Repeat Step 4 using 0.001 N HCl in place of the citrate buffer, i.e. determine the buffer capacity of 0.001 N HCl.
6 Determine the buffer capacity of your buffer and 0.001 N HCl by calculation. Compare your experimental results with your calculated answers. Explain any discrepancies between experimental and calculated values.
7 Determine the buffer capacity of Sprite® in the same way you did for your citrate buffer (Step 4 above).
1.6 Problem Set
1 The Ka for the weak acid HA is 4.0 × 10−6. What is the pH of a 0.01 M solution of this acid? What is its pKa?
2 How many grams of acetic acid and sodium acetate are required to prepare 1.0 l of 0.5 M acetate buffer, pH 4.5? The pKa for acetic acid is 4.75.
3 Explain carefully how to prepare 1.0 l of 0.05 M phosphate buffer, pH 7.0 from NaH2PO4 .H2O and 1.0 N NaOH or 1.0 N HCl. The molecular weight of sodium phosphate monohydrate monobasic is 138 g mole−1. The pKa2 for H3PO4 is 7.21. Hint: To determine whether you will need to add NaOH or HCl, you need to calculate the pH of a 0.05 M solution of NaH2PO4.
4 Preparation of buffers using published tables.Using Tables III.2a, III.2b, and III.2c in Appendix III, describe carefully how you would prepare 1 l of 0.1 M acetate buffer, pH 5.2 and 1 l of 1/15 M phosphate buffer, pH 7.6.Using the Henderson–Hasselbalch equation (shown below), calculate the theoretical pH of these 2 buffers. Explain why the calculated pHs are not exactly the same as the pHs shown in the Appendix table.
1.7 References
1 1 FDA (2019). Acidified & low‐acid canned foods guidance documents & regulatory information [Internet]. FDA [cited 2020 Mar 5]. http://www.fda.gov/food/guidance‐documents‐regulatory‐information‐topic‐food‐and‐dietary‐supplements/acidified‐low‐acid‐canned‐foods‐guidance‐documents‐regulatory‐information(accessed 5 March 2020).
2 2 Bartek Ingredients, Inc (2020). Self teaching guide for food acidulants ‐ Google search [Internet]. [cited 2020 Mar 5] p. 37. https://www.google.com/search?q=Self+teaching+guide+for+food+acidulants&rlz=1C1EJFC_enUS825US876&oq=Self+teaching+guide+for+food+acidulants&aqs=chrome..69i57j69i60.2887j0j4&sourceid=chrome&ie=UTF‐8(accessed 5 March 2020).
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Food Chemistry»
Представляем Вашему вниманию похожие книги на «Food Chemistry» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Food Chemistry» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












