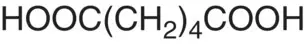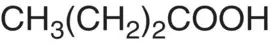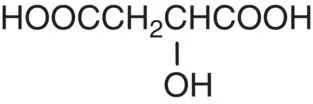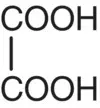Dennis D. Miller
Professor Emeritus
Department of Food Science, Cornell University
October 2021
Preface to the First Edition
Food chemistry is a broad discipline that draws on principles of physical, organic, and biological chemistry. Advances in food chemistry over the past century have had a dramatic impact on our understanding of all aspects of food science and technology and have played a major role in the improvement of the quality, quantity, and availability of the food supply.
This manual is designed for a one‐semester laboratory course in food chemistry. Emphasis is placed on understanding fundamental chemical principles that underlie relationships between the composition of foods and functional, nutritional, and organoleptic properties. In addition, many laboratory techniques that are common in basic and applied research in food chemistry are introduced.
Students should have a background in general, organic, and biochemistry as well as a concurrent lecture course in food chemistry. Each experiment is preceded by an introduction of the principles necessary for understanding and interpreting the data. Students are encouraged to supplement the introductory material by reading selected sections in a comprehensive food chemistry textbook in addition to the references cited at the end of each experiment.
Many students have successfully performed the experiments described in the manual. I have tried diligently to eliminate errors and provide clear instructions for students and instructors. Nevertheless, errors and unclear writing have a way of creeping in. I would appreciate hearing from students and instructors when they find errors.
Dennis D. Miller
Ithaca, New York
This manual is the product of the efforts of many people over a period of more than 40 years. First and foremost, we thank the students at Cornell University who enrolled in the Food Chemistry Laboratory course in the Department of Food Science over the 40 years it was offered as part of the Institute of Food Technologists‐approved curriculum for Food Science majors. Their enthusiasm, lab reports, presentations, questions, and gentle criticisms were an immense help in developing and refining the experiments in the manual. Another group of students who contributed enormously were the graduate and undergraduate teaching assistants who set up the laboratories, assisted students during the lab periods, graded lab reports, and offered suggestions for improving the exercises. In recent years, Aaron Jacobsen, Teaching Support Specialist at Cornell, has worked tirelessly to streamline the lab exercises and identify errors in the procedures. We also thank our colleagues at Cornell and Cal Poly for their comments, suggestions, and continual intellectual stimulation. We thank Drs. Alicia Orta Ramirez and Motoko Mukai who both took their turns in teaching the Food Chemistry Lab course at Cornell. We are especially grateful to Professors John Brady and Chang Lee for their friendship, encouragement, and support. Last but not least, we thank our editors at Wiley for their expert advice and support.
About the Companion Website
This book is accompanied by a companion website.
www.wiley.com/go/Miller/foodchemistry2 
This website includes:
Preparations and solutions for all chapters
1 Acids, Bases, and Buffers
1.1 Learning Outcomes
After completing this exercise, students will be able to:
1 Explain the roles of acids and bases in food products.
2 Measure the pH of selected food products.
3 Prepare and evaluate a buffer system.
4 Measure the buffering capacity of a common beverage.
Many food components may be classified as acids or bases due to their capacity to donate or accept protons (hydrogen ions). These components perform numerous important functions including flavor enhancement, control of microbial growth, inhibition of browning, alteration of texture, prevention of lipid oxidation, and pH control.
Acids and bases are key metabolites in living plant and animal organisms, for example as intermediates in the TCA cycle, and are mostly retained when the plant is harvested or the animal is slaughtered so they are naturally present in foods. They may also be added during processing or synthesized during fermentation to produce desired characteristics in the final food product.
The concentration and relative proportion of acids and bases determine the pH of a food, an extremely important characteristic. pH can affect the flavor, color, texture, stability, and behavior in food processing situations. For example, commercial sterilization of acid foods (pH less than 4.6) [1] can be achieved under milder processing conditions than in foods with a higher pH.
Acids serve a variety of functions in foods including flavor enhancement, control of microbial growth, protein coagulation, emulsification, control of browning, buffering action, and metal chelation (to control lipid oxidation). All acids have a sour taste but different acids produce distinctively different sour flavors. Thus, it is not enough to simply add any acid when attempting to produce a characteristic sour flavor in a food. Table 1.1gives structures and p K values of some common food acids.
Table 1.1 Acids common in foods: structures and p K avalues.
| Substance |
Structure |
p K a |
Food found in |
| Acetic acid |
 |
p K = 4.75 |
Vinegar, figs |
| Adipic acid |
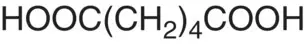 |
p K 1= 4.43 p K 2= 5.62 |
Beets |
| Butyric acid |
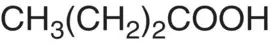 |
p K = 4.82 |
Cheese, butter |
| Citric acid |
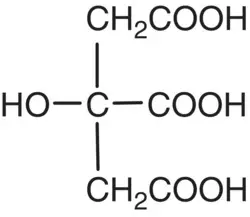 |
p K 1= 3.06 p K 2= 4.74 p K 3= 5.40 |
Oranges, lemons, apricots, tomatoes |
| Lactic acid |
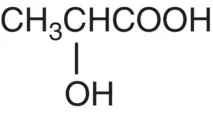 |
p K = 3.83 |
Yogurt, buttermilk, cheese, beer |
| Malic acid |
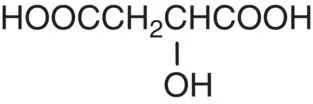 |
p K 1= 3.40 p K 2= 5.05 |
Apples, apricots, grapes, oranges, tomatoes |
| Oxalic acid |
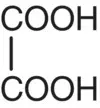 |
p K 1= 1.27 p K 2= 4.27 |
Spinach, potatoes, tomatoes |
| Phosphoric acid |
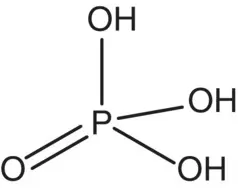 |
p K 1= 2.12 p K 2= 7.21 p K 3= 12.32 |
Tomatoes, acidulant used in soft drinks |
| Tartaric acid |
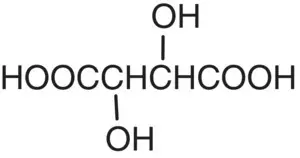 |
p K 1= 2.98 p K 2= 4.34 |
Grapes |
| Sodium hydrogen sulfate or sodium acid sulfate |
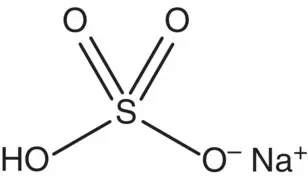 |
p K = 1.99 |
Acidulant. Lowers pH without imparting acid taste. May be added to process water to enhance chlorine activity |
Читать дальше