Source: Based on [88].
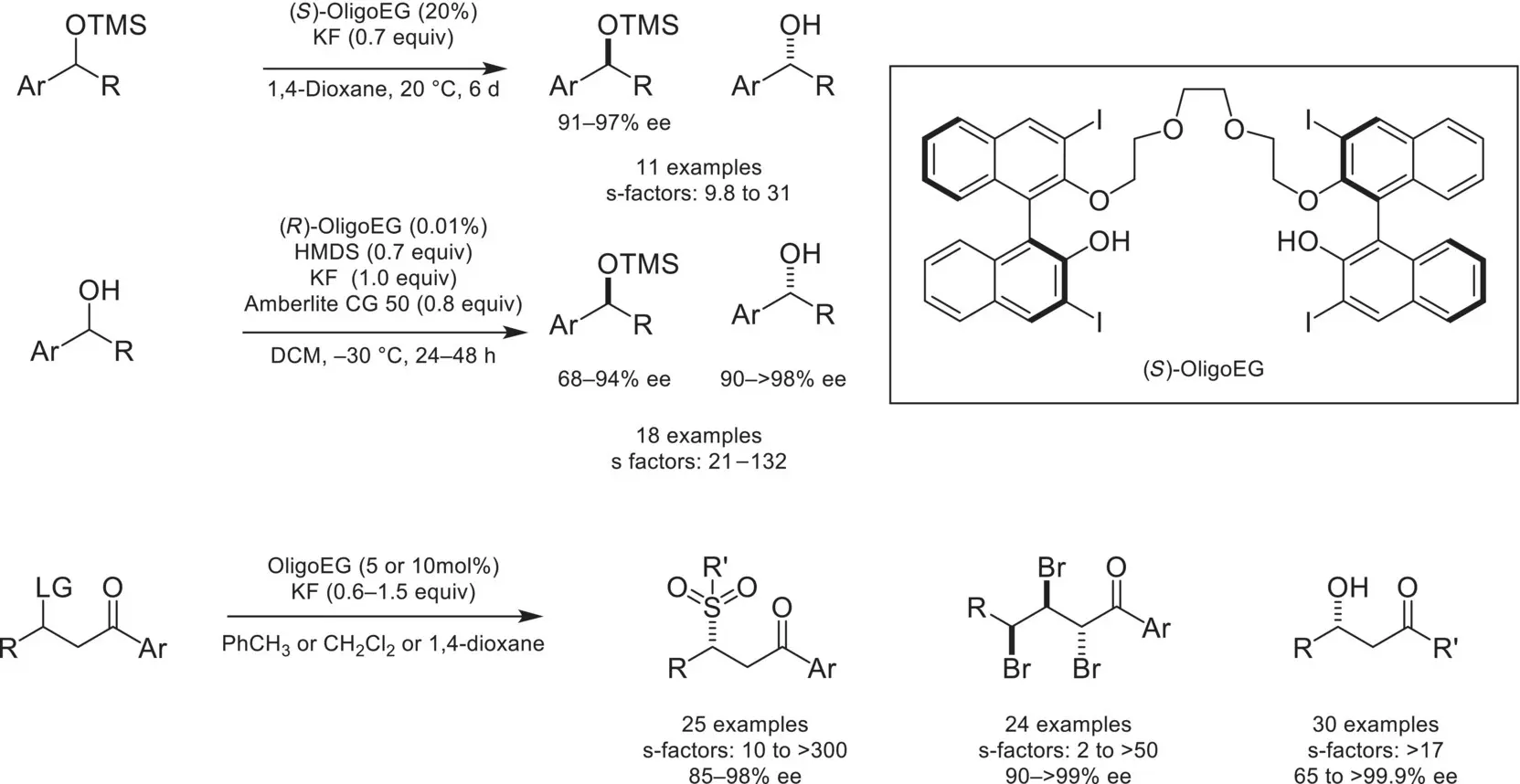
Scheme 4.29. Kinetic resolution of silyl‐protected alcohols.
Source: Based on [93].
The developments in the field of chiral cation phase‐transfer and ion‐pairing catalyses have displayed immense advancements for the field of catalysis. Despite the long history of this mode of catalysis, the charged‐reversed version, chiral‐anion catalysis, has been a significantly more recent development. This mode of activation centers on utilizing chiral‐anions to impart enantioselectivity on cationic intermediates and reagents via ion‐pairing. One advantage of this strategy is the orthogonal reactivity that can be achieved by focusing on transformations that feature cationic intermediates from relatively unactivated starting materials under mild conditions.
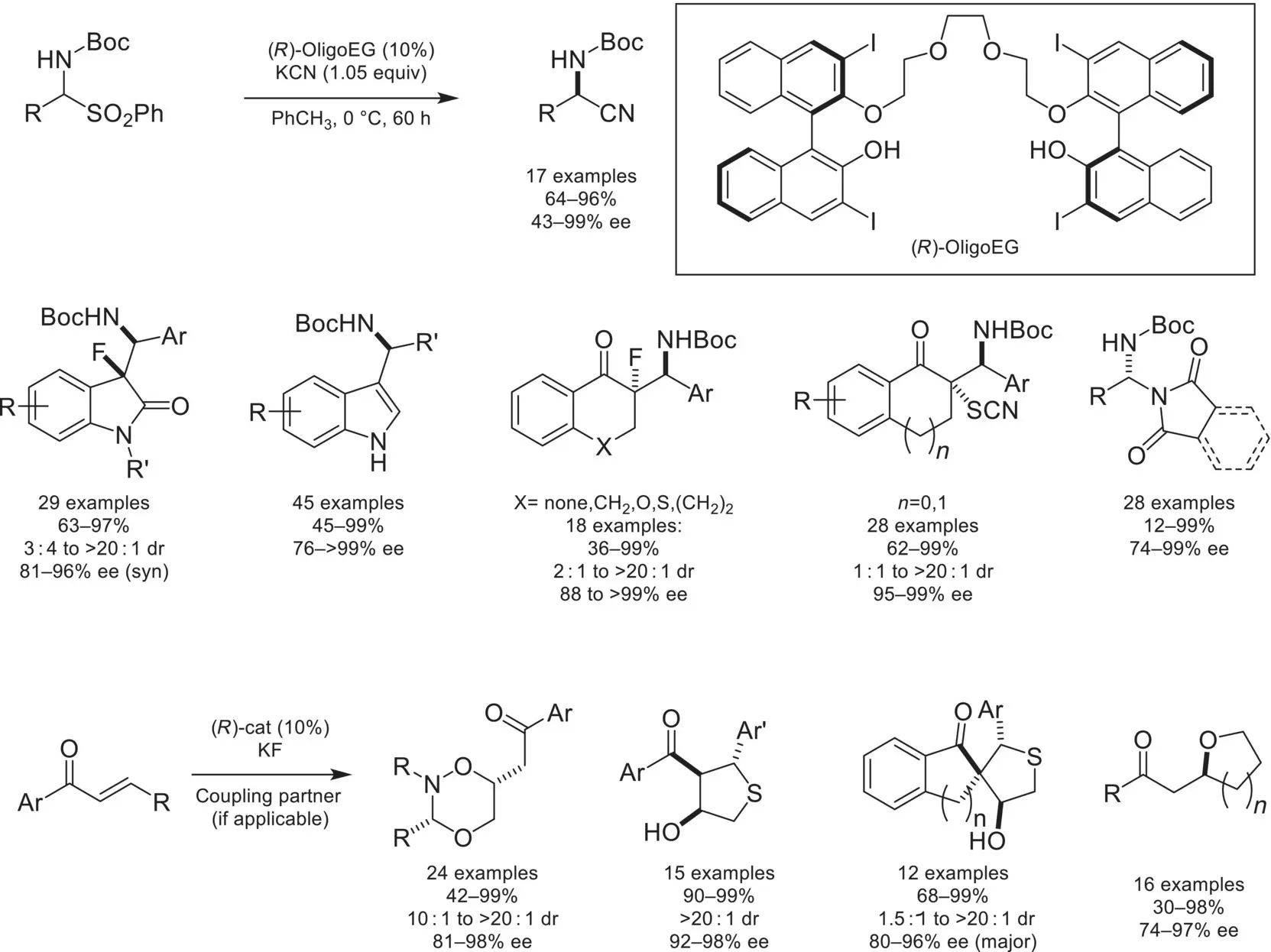
Scheme 4.30. Asymmetric Strecker reaction catalyzed by BINOL‐derived crown ether.
Source: Based on [99].
The first proof of principal example of utilizing ion‐pairing catalysis with a chiral‐anion was demonstrated by the List group in 2006 ( Scheme 4.31) [108]. In this example, an ammonium/chiral phosphate salt would catalyze the enantioselective reduction of enals with Hantzsch ester. The key intermediate for imparting enantioselectivity is the iminium formed from condensation of the achiral ammonium and the enal, which remains ion‐paired with a (R)‐TRIP counteranion. This ion‐pair leads to preferential reduction of one face of the alkene by Hantzsch ester, leading to β‐chiral ketones in high yields and enantioselectivities. Following this, the List group utilized this key intermediate for the epoxidation of di‐ and tetrasubstituted enals by using t ‐butyl hydroperoxide [109]. It was found that the identity of the achiral ammonium had a profound effect on the observed enantioselectivities (24–90% ee), where dibenzylic ammoniums with electron deficient arenes provided the best enantioselectivities. This demonstrates the importance of tuning both components of the ion‐pair for cooperatively achieving high selectivities, and is a key feature in many examples of chiral‐anion catalysis.
In addition to ammonium/chiral phosphoric acid salts, a variety of catalyst architectures can be utilized for generating iminium intermediates that are amenable to chiral‐anion catalysis. In 2013, the Masson group demonstrated that an iminium intermediate could be generated from protonation and subsequent dehydration of γ‐hydroxy‐γ‐lactams by a chiral phosphoric acid ( Scheme 4.32) [110]. The resulting iminium intermediate underwent asymmetric C3‐arylation with indoles in high yields and enantioselectivities. Other catalysts not based on BINOL phosphoric acids have been demonstrated to be competent for chiral‐anion catalysis. Notable examples include a novel conjugate‐based stabilized carboxylic acid that catalyzes an enantioselective three‐component Povarov reaction[111], and an ammonium/chiral BOROX salt, which activates aldehydes toward an asymmetric three‐component Ugi reaction [112].
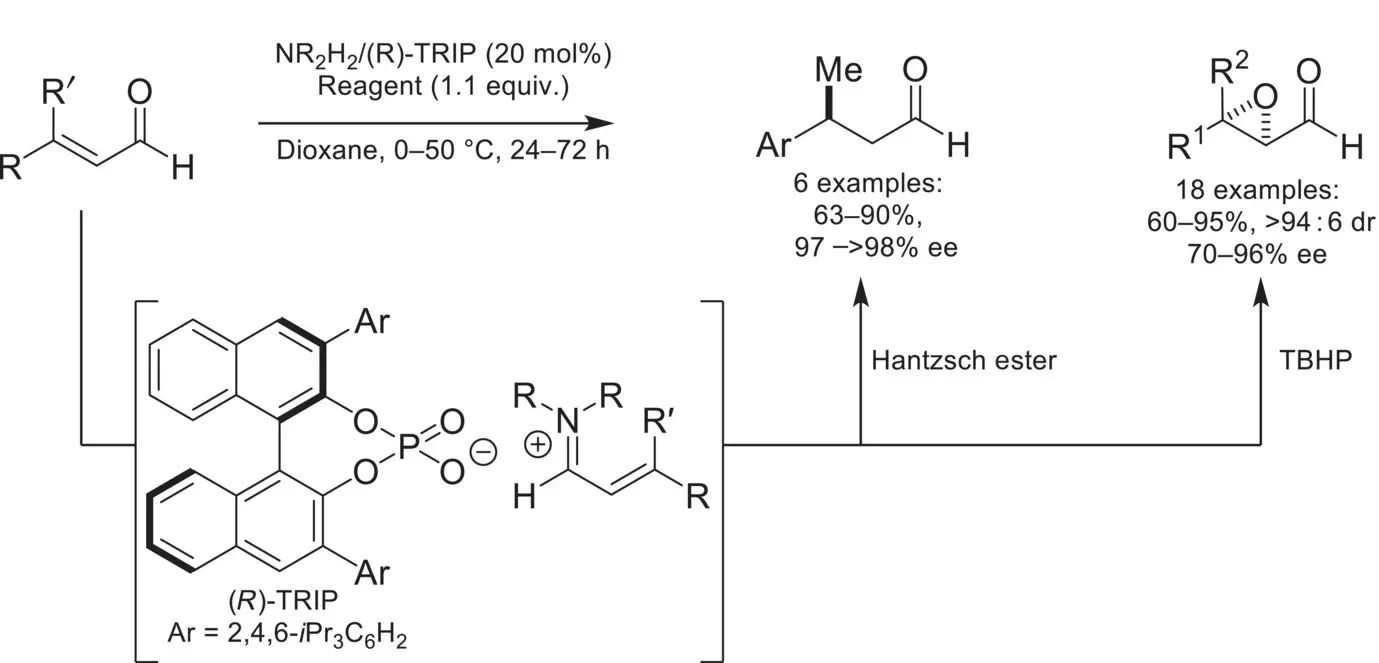
Scheme 4.31. First example of asymmetric counteranion‐directed catalysis.
Source: Based on [108].
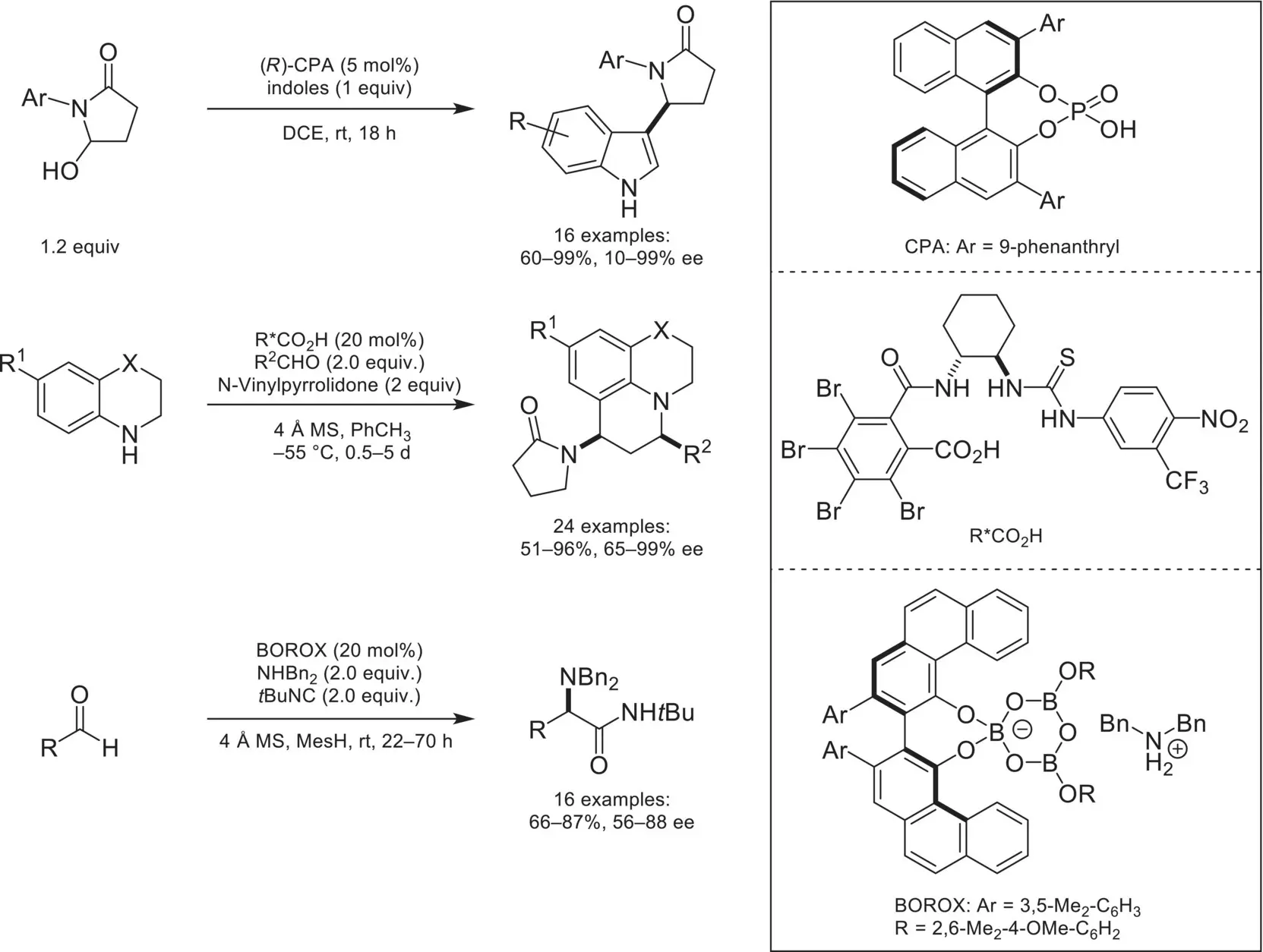
Scheme 4.32. Asymmetric counteranion‐directed catalysis involving iminium ion intermediates.
Source: Based on [110].
Following the successful demonstration of asymmetric iminium reactivity with chiral‐anions, there was an increased interest in expanding this reactivity to other cationic intermediates. In 2007, the List group expanded the scope of ion‐pairing catalysis to oxocarbenium intermediates in an asymmetric Mukaiyama aldol reaction by using a highly acidic disulfonimide catalyst ( Scheme 4.33) [113]. In a closely related follow‐up publication, the List group demonstrated vinylogous and doubly vinylogous silyl enol ethers as nucleophiles [114].
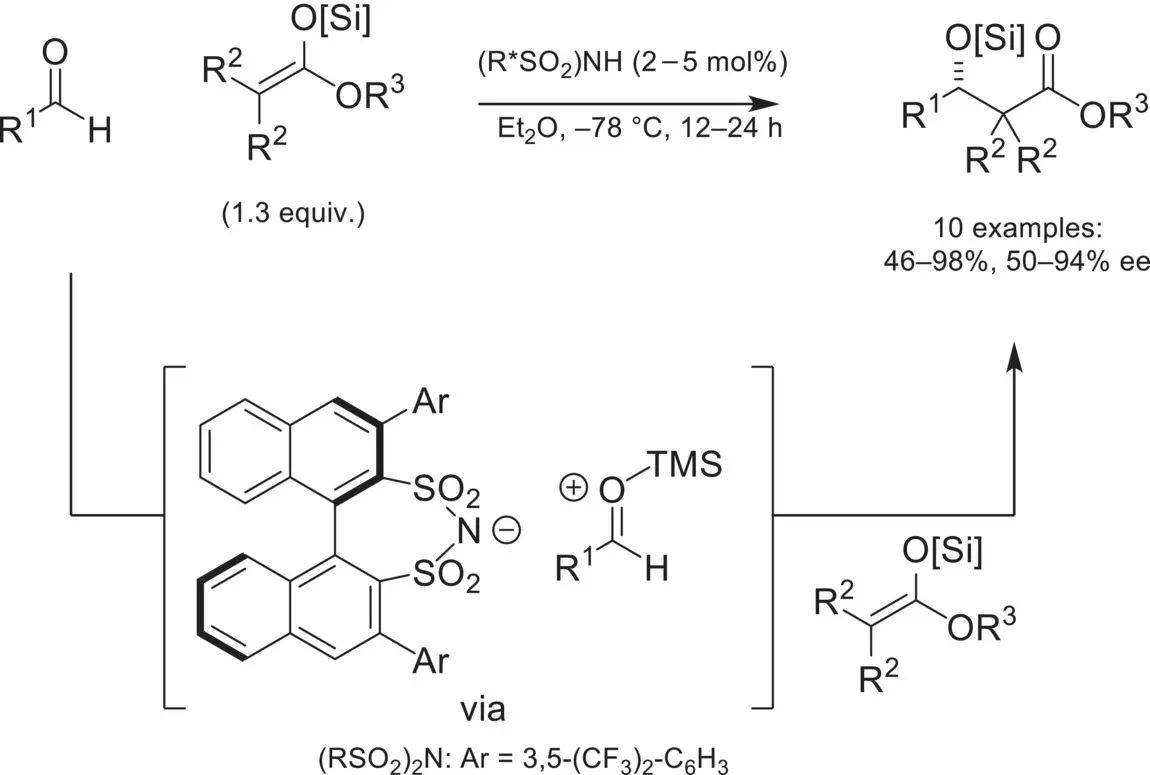
Scheme 4.33. Asymmetric Mukaiyama aldol reaction catalyzed by a highly acidic BINOL‐derived disulfonimide.
Source: Based on [113].
A variety of stabilized oxocarbenium intermediates has been shown to be compatible with this reaction paradigm. In 2013, the Terada group demonstrated an enantioselective reduction of hydroxy‐pyrans via a pyrylium/TRIP chiral ion‐pair ( Scheme 4.34) [115]. A similar pyrylium intermediate was shown to undergo an enantioselective Diels‐Alder via ion‐pairing with a chiral phosphate, followed by rearrangement to an exo‐cyclic ketone [116]. Nonaromatic oxocarbeniums have also been accessed and utilized in chiral‐anion catalysis, as demonstrated by the Nagorny group. In this work, an allyl acetal underwent a Diels‐Alder, via the intermediacy of an allylic oxocarbenium ion‐paired with a chiral phosphate anion [117].
In order to access oxocarbenium intermediates without stabilizing moieties, more acidic chiral acids were required to favor ionization of the substrate. In 2016, the Thiel, De, and List groups developed an oxa‐Pictet‐Spengler reaction, catalyzed by a highly acidic imidodiphosphoric acid ( Scheme 4.35) [118]. In 2018, Scheidt demonstrated that a similar oxa‐Pictet‐Spengler reaction could be realized with a less acidic chiral catalyst [119], by introducing exogenous achiral urea to increase the acidity of the catalyst via hydrogen bonding of the phosphate anion. Similarly, a bifunctional catalyst featuring a thiourea moiety to greatly increase the acidity of an intramolecular carboxylic acid was shown to be competent for this reactivity [120].
A potential alternative to the use of stronger chiral acids to access unstabilized oxocarbeniums was demonstrated in 2015 by the Luo group ( Scheme 4.36) [121]. In this example, a trityl cation/chiral phosphate salt was shown to activate α‐ketoesters toward asymmetric reactivity with a variety of coupling partners. Numbers of transformations were compatible with this system, such as 1,4‐addition with indoles, Diels‐Alder with cyclopentadiene, and 1,2‐addition with α‐methyl styrene, which occurred in high yields and selectivities.
A variety of asymmetric intramolecular reactions was developed featuring stabilized carbocations as intermediates. In 2010, the Antilla group demonstrated that chiral phosphoric acids could catalyze the enantioselective semi‐pinacol rearrangement of an indolyl diol via intermediacy of the indolyl cation/chiral phosphate ion‐pair ( Scheme 4.37) [122]. A vinylogous variant of this reaction was reported in 2016 by the Zhu group, forming β,γ‐unsaturated ketones in high yields and enantioselectivities [123]. Other intramolecular reactivity was shown in 2018 by the Ariafard and Chan groups, with an enantioselective Nazarov‐type electrocyclization of allylic alcohols, forming chiral indenes and tricyclic compounds in high yields and enantioselectivities [124].
Читать дальше












